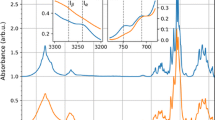
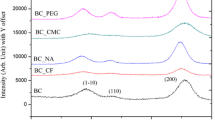
Abstract
Effects of polymer additives on the formation of microfibrils of bacterial cellulose have been examined by transmission electron microscopy. Among additives with different degrees of polymerization (DP) or substitution (DS), carboxymethyl cellulose sodium salt (CMC) with DP = 80 and DS = 0.57 is the most effective in producing separate, smaller-size microfibrils. By increasing the concentration of this CMC from 0.1 to 1.5%, the percentage of microfibrils measuring 3–7 nm wide is increased and levels off at around 1.0%. Other polymer additives such as xyloglucan are less effective than CMC in producing microfibrils with smaller sizes and the resulting microfibrils still tend to aggregate. The number of charged substituents and the molecular weight seem to be important factors in the production of highly separate smaller-size microfibrils. The reduction in average microfibril size is well correlated to the decrease in mass fraction of cellulose Iα in bacterial cellulose crystals. On the basis of these results, the mechanism of the crystallization of celluloses Iα and Iβ is discussed. The effect of colony types, smooth and rough, on the formation of microfibrils in the presence of CMC is also described.
Similar content being viewed by others
REFERENCES
Atalla, R. H. and VanderHart, D. L. (1984) Native cellulose: A composite of two distinct crystalline forms. Science 223, 283-285.
Ben-Hayim, G. and Ohad, I. (1965) Synthesis of cellulose by Acetobacter xylinum VIII. On the formation and orientation of bacterial cellulose fibrils in the presence of acidic polysaccharides. J. Cell Biol. 25, 191-207.
Brown, R. M. Jr., Willison, J. H. M. and Richardson, C. L. (1976) Cellulose biosynthesis in Acetobacter xylinum: Visualization of the site of synthesis and direct measurement of the in vivo process. Proc. Natl. Acad. Sci. U.S.A. 73, 4565-4569.
Haigler, C. H. (1991) Relationship between polymerization and crystallization in microfibril biogenesis. In Biosynthesis and Biodegradation of Cellulose (C. H. Haigler & P. J. Weimer eds). New York: Marcel Dekker, Inc. pp. 99-124.
Haigler, C. H. and Benziman, M. (1982) Biogenesis of cellulose I microfibrils occurs by cell-directed self-assembly in Acetobacter xylinum. In Cellulose and Other Natural Polymer Systems (R. M. Brown, Jr., ed). New York: Plenum Press. pp. 273-297.
Haigler, C. H. and Chanzy, H. (1988) Electron diffraction analysis of the altered cellulose synthesized by Acetobacter xylinum in the presence of fluorescent brightening agents and direct dyes. J. Ultrastruct. Mol. Struct. Res. 98, 299-311.
Haigler, C. H. and Chanzy, H. (1989) Electron diffraction analysis of altered cellulose: Implications for cellulose biogenesis. In Cellulose and Wood: Chemistry and Technology (C. Schuerch, ed.). New York: John Wiley and Sons. pp. 493-506.
Haigler, C. H., White, A. R., Brown, R. M. Jr. and Cooper, K. M. (1982) Alternation of in vivo cellulose ribbon assembly by carboxymethylcellulose and other cellulose derivatives. J. Cell Biol. 94, 64-69.
Hayashi, T., Marsden, M. P. E. and Delmer, D. P. (1987) Pea xyloglucan and cellulose. V. Xyloglucancellulose interactions in vitro and in vivo. Plant Physiol. 83, 384-389.
Hestrin, S. and Schramm, M. (1954) Synthesis of cellulose by Acetobacter xylinum. Biochem. J. 58, 345-352.
Hirai, A., Tsuji, M. and Horii, F. (1997) Culture conditions producing structure entities composed of Cellulose I and II in bacterial cellulose. Cellulose 4, 239-245.
Horii, F. (1998) Structure of cellulose: Recent developments in its characterization. Wood and Cellulosic Chemistry, (D. N.-S. Hon and N. Shiraishi, eds). New York: Marcel Dekker, Inc., in press.
Horii, F., Yamamoto, H. and Hirai, A. (1997) Microstructural analysis of microfibrils of bacterial cellulose. Macromol. Symp. 120, 197-205.
Saxena, I. M., Kudlicka, K., Okuda, K. and Brown, R. M., Jr. (1994) Characterization of genes in the cellulose-synthesizing operon (acs operon) of Acetobacter xylinum: Implications for cellulose crystallization. J. Bacteriol. 176, 5735-5752.
Sugiyama, J., Vuong, R. and Chanzy, H. (1991) Electron diffraction study on the two crystalline phases occurring in native cellulose from an algal cell wall. Macromolecules 24, 4168-4175.
Uhlin, K. I., Atalla, R. H. and Thompson, N. S. (1995) Influence of hemicelluloses on the aggregation patterns of bacterial cellulose. Cellulose 2, 129-144.
Whitney, S. E. C., Brigham, J. E., Darke, A. H., Grant Reid, J. S. and Gidley, M. J. (1995) In vitro assembly of cellulose/;xyloglucan networks: ultrastructural and molecular aspects. Plant J. 8, 491-504.
Yamamoto, H. and Horii, F. (1993) CP/;MAS 13C NMR analysis of the crystal transformation induced for Valonia cellulose by annealing at high temperatures. Macromolecules 26, 1313-1317.
Yamamoto, H. and Horii, F. (1994) In situ crystallization of bacterial cellulose I. Influences of polymeric additives, stirring and temperature on the formation celluloses Iα and Iβ as revealed by cross polarization/;magic angle spinning (CP/;MAS) 13C NMR spectroscopy. Cellulose 1, 57-66.
Yamamoto, H., Horii, F. and Hirai, A. (1996) In situ crystallization of bacterial cellulose II. Influences of polymeric additives with different molecular weights on the formation of celluloses Iα and Iβ at the early stage of incubation. Cellulose 3, 229-242.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hirai, A., Tsuji, M., Yamamoto, H. et al. In Situ Crystallization of Bacterial Cellulose III. Influences of Different Polymeric Additives on the Formation of Microfibrils as Revealed by Transmission Electron Microscopy. Cellulose 5, 201–213 (1998). https://doi.org/10.1023/A:1009233323237
Issue Date:
DOI: https://doi.org/10.1023/A:1009233323237