Abstract
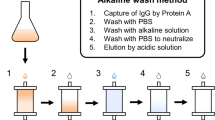
Hydrophobic charge induction chromatography is a recently developed method for protein separation based on the use of dual-mode ligands. They are designed in such a way so as to combine a molecular interaction supported by a mild hydrophobic association effect in the absence of salts. When environmental pH is changed, the ligand becomes ionically charged resulting into the desorption of the protein. This method is applied to the separation of antibodies from ascite fluids and culture supernatants from hybridomas cultured in the presence of fetal bovine serum or in protein free environment. Typically adsorption from cell culture supernatants is accomplished without any pH or ionic strength adjustment; the column is then washed with a typical buffer to eliminate protein impurities. Antibodies are then desorbed using acetate buffer, pH 4. Antibody binding capacity is in the range of 30 mg per ml of resin at 10% breakthrough. Antibody purity varies according to the initial feed stock and can reach values higher than 90% in a single pass. One example of antibody purification process involving hydrophobic charge induction chromatography as a capture step followed by a polishing phase with DEAE Ceramic HyperD is described. Longevity and ligand leakage are compatible with large-scale applications.
Similar content being viewed by others
References
Boschetti E and Jungbauer A (2000) Separation of antibodies by liquid chromatography. Handbook of Bioseparation, Academic Press, S. Ahuja ed., vol. 2 pp 533-632.
Bruck C, Portelle D, Glineur C and Bollen A (1982) One-step purification of mouse monoclonal antibodies from ascites fluid by DEAE Affigel Blue chromatography. J. Immunol. Meth. 53: 313-319.
Burton SC and Harding DRK (1998) Hydrophobic charge induction chromatography: salt independent protein adsorption and facile elution with aqueous buffers. J. Chromatogr. A. 814: 71-81.
Duffy S, Moellering BJ, Prior GM, Doyle KR and Prior CP (1989) Recovery of therapeutic-grade of antibodies: Protein A and ion exchange chromatography. BioPharm. 6: 35-47.
Finger UB, Brummer W, Knieps E, Thommes J and Kula MR (1996) Investigations on the specificity of thiophilic interaction for monoclonal antibodies of different subclasses. J. Chromatogr. 675: 197-204.
Gooding DL, Schmuck MN, Nowlan MP and Gooding KM (1986) Optimization of preparative hydrophobic interaction chromatography purification methods. J. Chromatogr. 359: 331-337.
Kent UM (1994) Purification of antibodies using ion exchange chromatography. Methods Mol. Biol. 34: 23-27.
Li R, Dowd V, Stewart DJ, Burton SJ and Lowe CR (1998) Design, synthesis and application of a Protein A mimetic. Nature Biotechnology 16: 190-195.
Manzke O, Tesch H, Diehl V and Bohlen H (1997) Single-step purification of biospecific monoclonal antibodies for immunotherapeutic use by hydrophobic interaction chromatography. J. Immunol. Meth. 208: 65-73.
Necina R, Amatschek K and Jungbauer A (1998) Capture of human monoclonal antibodies from cell culture supernatant by ion exchange media exhibiting high charge density. Biotechnol. Bioeng. 60: 679-698.
Ngo TT (1994) Rapid purification of immunoglobulin G using aza-arenophilic chromatography: novel mode of solid phase interaction. J. Chromatogr. 662: 351-356.
Nopper B, Kohen F and Wilchek M (1989) A thiophilic adsorbent for the one-step high-performance liquid chromatography purification of monoclonal antibodies. Anal. Biochem. 180: 66-71.
Oscarson S. and Porath J (1990) Protein chromatography with pyridine-and alkyl-thioether-based agarose adsorbents. J. Chromatogr. 499: 235-247.
Ostlund C, Borwell P and Malm B (1987) Process-scale purification from cell culture supernatants: monoclonal antibodies. Dev. Biol. Stand. 66: 367-375.
Palombo G, Verdoliva A and Fassina G (1998) Affinity purification of immunoglobulin M using a novel synthetic ligand. J. Chromatogr. 715: 137-145.
Pilcher JB, Tsang VCW, Zhou W, Black CM and Sidnam C (1991) Optimization of binding capacity and specificity of protein G on various solid phase matrices for immunoglobulins. J. Immunol. Meth 136: 279-286.
Rapoport EM, Zhigis LS, Vlasova EV, Piskaref VE, Bovin NV and Zubov VP 1995) Purification of monoclonal antibodies to Le(y) and Le(d) carbohydrate antigens by ion exchange and thiophilic adsorption chromatography. Bioseparation 5: 141-146.
Scholz GH, Vieweg S, Leistner S, Seissler J, Scherbaum WA, Huse K (1998) A simplified procedure for the isolation of immunoglobulins from human serum using a novel type of thiophilic gel at low salt concentration. J. Immunol. Meth. 219: 109-118.
Scholz GH, Wippich P, Leistner S and Huse K (1998) Salt-independent binding of antibodies from human serum to thiophilic heterocyclic ligands. J. Chromatogr. 709: 189-196.
Schwarz A (1996) Five-membered mercaptoheterocyclic ligands for thiophilic adsorption chromatography. J. Mol. Recognit. 6: 672-674.
Sene C, Santambien P and Pouradier Duteil X (1990) Chemistry Today 3: 15-21.
Sulk B, Birkenmeier G and Kopperschlager G (1992) Application of phase partitioning and thiophilic adsorption chromatography to the purification of monoclonal antibodies from cell culture fluids. J. Immunol. Meth. 149: 165-171.
Thommes J, Bader A, Halfar M, Karau A and Kula MR (1996) Isolation of monoclonal antibodies from cell containing hybridoma broth using a Protein A coated adsorbent in expanded beds. J. Chromatogr. 752: 111-122.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guerrier, L., Girot, P., Schwartz, W. et al. New method for the selective capture of antibodies under physiolgical conditions. Bioseparation 9, 211–221 (2000). https://doi.org/10.1023/A:1008170226665
Issue Date:
DOI: https://doi.org/10.1023/A:1008170226665