Abstract
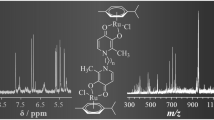
The 'caged NO' reagent, RuCl3NO(H2O)2, has been studied by n.m.r. and i.r. methods with imidazole, histidine, histamine, and N-methyliminodiacetate as complexing ligands. These ligands are representative of cellular donors that would be encountered as RuCl3NO(H2O)2 migrates through biological cells. [RuCl3NO(imH)(H2O)], [RuCl3(NO)(imH)2] and [RuCl2(NO)(imH)3]+ complexes (imH = imidazole) have been detected by 1H-n.m.r. and i.r. and electrospray mass spectrometry (e.s.i.–m.s.) methods. Based upon the effect of cis ligand addition on the ν(NO) frequency causing a decrease in frequency, the 1:1 and 1:2 complexes have the imidazole donors in the plane cis to the NO+ moiety, whereas the 1:3 species has the third imidazole trans to the NO+. The trans imidazole donor causes 'trans-strengthening' of the N–O bond of the {RuNO}6 chromophore. 1H-n.m.r. shows that the monodentate imidazole donor(s) is (are) in rapid exchange with free imidazole in solution for each of the n = 1–3 species. Histidine and histamine make kinetically more stable 1:1 complexes with the major isomer having an axially-coordinated histidine imidazole donor, but in-plane donation for histamine. The carboxylate of coordinated histidine remains pendant according to i.r. and 13C-n.m.r. data. From syntheses carried out at pH ca. 5, the amino donor is H-bonded to an in-plane H2O in the major species (ca. 75%) and coordinated with displacement of the in-plane H2O in the lesser isomer (25%). By contrast, the histamine ligand binds with an in-plane bound imidazole and a pendant protonated amino group (94%). The remaining 6% has an in-plane chelated histamine, analogous to the bis imidazole species and the known fac, cis-[RuCl3NO(en)] complex. N-Methyliminodiacetate is observed to form one main [RuCl(NO)(mida)(H2O)] complex (85%) with two chelated glycinato donor groups with RuCl3NO(H2O)2, one glycinato group chelated 'in-plane' with the central amine donor and one axial coordinated glycinato donor. A second [RuCl(NO)(mida)(H2O)] complex (the remaining 15%) has the amine donor trans to NO+ and chelated glycinato groups which coordinate in the RuClO2(OH2) plane, either cis or trans to each other, in a 60:40 split (ca. 9% and 6%). The presence of one Cl− and one H2O in the [RuCl(NO)(mida)(H2O)] complexes was established by e.s.i.–m.s. These results show that RuCl3NO(H2O)2 is likely to be freely mobile within a cellular environment, forming stable complexes via bidentate chelation with 'two-point' nitrogen donors (en, his, etc).
Similar content being viewed by others
References
N. Bettache, T. Carter, J.E. Corrie and D. Ogden, Methods in Enzymology, 268, 266 (1996).
T.D. Carter, N. Bettache and D. Ogden, Brit. J. Pharmacol, 122, 971 (1997).
P.C. Ford, J. Bourassa, K. Miranda, B. Lee, I. Lorkovic, S. Boggs, S. Kudo and L. Laverman, Coord. Chem. Rev., 171, 185 (1998).
G. Stochel, A. Wanat, E. Kulis and Z. Stasicka, Coord. Chem. Rev., 171, 203 (1998).
T. Ishiyama, T. Matsumura and Y. Honda, Radioisotopes, 30, 361 (1981).
H. Tomizawa, K. Harada, E. Miki, K. Mizumachi, T. Ishimori, A. Urushiyama and M. Nakahara, Bull. Chem. Soc. Jpn., 66, 1658 (1993).
W. Odenkirk, A.L. Rheingold and B. Bosnich, J. Am. Chem. Soc., 114, 6392 (1992).
A.A. Batista, C. Pereira, S.L. Queiroz, L.A.A. Olivera, R.H. de A. Santos and M.T. do P. Gambardella, Polyhedron, 16, 927 (1997).
C.T. Page and J.E. Fergusson, Aust. J. Chem., 36, 855 (1983).
S. Dhf, O.G. Teixeira and A.A. Batista, Polyhedron, 14, 1031 (1995).
A. Domenicano, A. Vaciago, L. Zambonelli, P.L. Loader and L.M. Venanzi, J. Chem. Soc., Chem. Commun., 476 (1966).
(a) H. Tomizawa, E. Miki, K. Mizumachi and T. Ishimori, Bull. Chem. Soc. Jpn., 67, 1809 (1994); (b) H. Tomizawa, E. Miki, K. Mizamachi and T. Ishimori, Bull. Chem. Soc. Jpn., 67, 1816 (1994).
G.A. Heath and R.L. Martin, Aust. J. Chem., 23, 2297 (1970).
J.V. Dubrawski and R.D. Feltham, Inorg. Chem., 19, 355 (1980).
J.M. Slocik, R.A. Kortes and R.E. Shepherd, Metal-Based Drugs, 7, 67 (2000).
D.R. Lang, J.A. Davis, L.G.F. Lopes, A.A. Ferro, L.C.G. Vaasconcellos, D.W. Franco, E. Tfouni, A. Wieraszko and M.J. Clarke, Inorg. Chem., 39, 2294 (2000).
Y. Suzuki, H. Tomizawa and E. Miki, Inorg. Chim. Acta, 290, 36 (1999).
(a) W. Petri, T. Pieper, M. Sommer, B.K. Keppler and G. Geister, Eur. J. Inorg. Chem., 9, 155 (1999). (b) B.K. Keppler, W. Rupp, U.W. Juhl, H. Endres, R. Niebel and W. Balzer, Inorg. Chem., 26, 4366 (1987). (c) B.K. Keppler and W. Rupp, J. Cancer Res. Clin. Oncol., 111, 166 (1986). (d) F.T. Garzon, M.R. Berger, B.K. Keppler and D. Schmal, 19, 347 (1987). (e) A. Galeano, M.R. Berger and B.K. Keppler, Arzneimittel-Forsch, 42, 821 (1992).
(a) L. Trynda-Lemiesz, B.K. Keppler and H. Kozlowski, J. Inorg. Biochem., 73, 123 (1999). (b) F. Kratz, B.K. Keppler, L. Messori, C. Smith and E.N. Baker, Metal-Based Drugs, 1, 169 (1994). (c) F. Frantz, M. Hartmann, B. Keppler and L. Messori, J. Biol. Chem., 269, 2581 (1994). (d) C.A. Smith, A.J. Sutherland-Smith, B.K. Keppler, F. Kratz and E.N. Baker, J. Biol. Inorg. Chem., 1, 424 (1996).
F.G. Vilchez, R. Vilaplana, G. Balasco and L. Messori, J. Inorg. Biochem., 71, 45 (1998).
T.A. Balakaeva, A.V. Vhurakov, M.G. Erzernitskaya, L.G. Kux'mina, B.V. Lokshin and E. Ehimenko, Russ. J. Coord. Chem., 25, 579 (1999).
J. Slocik and R.E. Shepherd, Inorg. Chim. Acta, (2000), accepted for publication.
(a) G.E. Kirvan and D.W. Margerum, Inorg. Chem., 24, 3017 (1985). (b) J.C. Cooper, L.F. Wong and D.W. Margerum, Inorg. Chem., 17, 261 (1978). (c) B.E. Schwederski, H.P.D. Lee and D.W. Margerum, Inorg. Chem., 29, 3569 (1990). (d) E.J. Billo, G.F. Smith and D.W. Margerum, J. Am. Chem. Soc., 93, 2635 (1971).
P. Tsiveriotis, N. Hadjiladis and G. Stavropoulos, Inorg. Chim. Acta, 261, 83 (1997).
M.K. Kim and A.E. Martell, J. Am. Chem. Soc., 89, 5138 (1967).
S.P. Fricker, E. Slade, N.A. Powell, O.J. Vaughn, G. Henderson, S.A. Murrer, I.C. Megson, S.K. Bisland and F.W. Flitney, Br. J. Pharmacol., 122, 1441 (1997).
N. Davies, M.T. Wilson, E. Slade, S.P. Fricker, B.A. Murrer, N.A. Powell and G.R. Henderson, J. Chem. Soc., Chem. Commun., 47 (1997).
(a) Z. Guo and P.J. Sadler, Adv. Inorg. Chem., 49, 183 (2000); (b) M.J. Clarke, F.C. Zhu and D.R. Frasca, Chem. Rev., 99, 2511 (1999).
Y. Chen and R.E. Shepherd, J. Inorg. Biochem., 68, 183 (1997).
Y. Chen, F.-T. Lin and R.E. Shepherd, Inorg. Chem., 38, 973 (1999).
J.M. Fletcher, I.L. Jenkins and F.M. Lever, J. Inorg. Nucl. Chem., 1, 378 (1955).
Y. Chen, F.-T. Lin and R.E. Shepherd, Inorg. Chem., 36, 818 (1997).
Y. Chen, F.-T. Lin and R.E. Shepherd, Inorg. Chim. Acta, 268, 287 (1998).
Y. Chen and R.E. Shepherd, Inorg. Chim. Acta, 293, 123 (1999).
Y. Chen, M.A. Sweetland and R.E. Shepherd, Inorg. Chim. Acta, 260, 163 (1997).
M.S. Ward and R.E. Shepherd, Inorg. Chim. Acta, 286, 197 (1999).
M.S. Ward, G. Borisenko and R.E. Shepherd, Transition Met. Chem., 24, 224 (1999).
J.B. Godwin and T.J. Meyer, Inorg. Chem., 10, 471 (1971).
H. Nagao, H. Nishimura, H. Funato, Y. Ichikawa, F.S. Howell, M. Mukaida and H. Kakihana, Inorg. Chem., 28, 3955 (1989).
(a) R.E. Shepherd and H. Taube, Inorg. Chem., 12, 1392 (1973). (b) R.J. Sundberg, R.E. Shepherd and H. Taube, J. Am. Chem. Soc., 94, 6558 (1972).
(a) M.F. Hoq, C.R. Johnson, S. Paden and R.E. Shepherd, Inorg. Chem., 22, 2693 (1983). (b) W.W. Henderson, R.E. Shepherd and A.J. Abola, Inorg. Chem., 25, 1911 (1986).
R.A. Sanchez-Delgado, M. Navarro, K. Lazardi, R. Atencio, M. Capparelli, F. Vargas, J.A. Urbina, A. Bouillez, A.F. Noels and D. Masi, Inorg. Chim. Acta, 276, 528 (1998).
T. Ishiyama and T. Matsumura, Bull. Chem. Soc. Jpn., 52, 619 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Slocik, J.M., Ward, M.S., Somayajula, K.V. et al. Coordination of RuCl3(NO)(H2O)2 by imidazole, histidine and iminodiacetate ligands: a study of complexation of 'Caged NO' by simple bio-cellular donors. Transition Metal Chemistry 26, 351–364 (2001). https://doi.org/10.1023/A:1007194314107
Issue Date:
DOI: https://doi.org/10.1023/A:1007194314107