Abstract
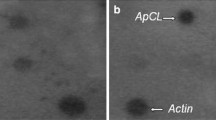
Plants of Arabidopsis thaliana pre-treated at 37 °C for 2 h can survive an otherwise lethal heat shock at 45 °C. Differential display reverse transcriptase-PCR (DDRT-PCR) was utilized to clone DNA fragments corresponding to mRNAs specifically expressed in conditions of induced thermotolerance or of expression of thermotolerance. One of these DDRT-PCR fragments enabled the isolation of a genomic clone pAt1.3EX, containing the sequence Athsp23.5, the gene for a low-molecular-weight (LMW) heat shock protein (HSP), AtHSP23.5. Athsp23.5 is low- or single-copy in the Arabidopsis genome and its open reading frame is interrupted by a 137 bp intron. Analysis of the sequence suggests AtHSP23.5 is targeted to the mitochondrion. The steady-state level of the AtHSP23.5 mRNA varied significantly according to the heat treatment, increasing on heat shock (transfer from 22 °C to 37 °C), with a further increase during expression of thermotolerance (transfer from 22 °C to 37 °C and then to 45 °C). Expression was low after an abrupt stress (from 22 °C to 45 °C). This behaviour was different from that observed for other LMW HSP mRNAs that were present at high level at 37 °C, but did not increase significantly in condition of expression of thermotolerance, and reached a considerable steady-state level also during the abrupt stress at 45 °C. The retrotranscription of AtHSP23.5 mRNA followed by amplification with two primers encompassing the intron allowed for the isolation of an almost full-length cDNA sequence. The sequence analysis of the two cDNAs obtained from condition 22 °C→37 °C and condition 22 °C→37 °C→45 °C suggested that in both cases the intron had been correctly spliced. The importance of correct intron splicing in survival at high temperatures and the role of mitochondrial HSP in induction and expression of thermotolerance are discussed.
Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ: Basic local alignment search tool. J Mol Biol 215: 403-410 (1990).
Amin J, Ananthan J, Voellmy R: Key features of heat shock regulatory elements. Mol Cell Biol 8: 3761-3769 (1988).
Bartling D, Bülter H, Liebeton K, Weiler EW: An Arabidopsis thalianacDNA clone encoding a 17.6 kDa class II heat shock protein. Plant Mol Biol 18: 1007-1008 (1992).
Dellavalle RP, Petersen R, Lindquist S: Preferential deadenylation of Hsp70mRNA plays a key role in regulating Hsp70 expression in Drosophila melanogaster. Mol Cell Biol 14: 3646-3659 (1994).
Dong JZ, Dunstan DI: Characterization of three heat-shockprotein genes and their developmental regulation during somatic embryogenesis in white spruce [Picea glauca(Moench) Voss]. Planta 200: 85-91 (1996).
Feinberg AP, Vogelstein B: Atechnique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132: 6-13 (1983).
Gavel Y, von Heijne G: Cleavage-site motifs in mitochondrial targeting peptides. Prot Eng 4: 33-37 (1990).
Gish W, States DJ: Identification of protein regions by database similarity search. Nat Genet 3: 266-272 (1993).
Helm KW, Schmeits J, Vierling E: An endomembranelocalized small heat-shock protein from Arabidopsis thaliana. Plant Physiol 107: 287-288 (1995).
Helm KW, Vierling E: An ArabidopsiscDNA clone encoding a low molecular weight heat shock protein. Nucl Acids Res 17: 7995 (1989).
Jäättelä M, Wissing D: Emerging role of heat shock proteins in biology and medicine. Ann Med 24: 249-258 (1992).
Jakob U, Buchner J: Assisting spontaneity: the role of HSP 90 and small Hsps as molecular chaperones. Trends Biochem Sci 19: 205-211 (1994).
Joshi CP: Putative polyadenylation signals in nuclear genes of higher plants. Nucl Acids Res 15: 9627-9640 (1987).
Joshi CP, Kumar S, Nguyen HT: Application of modified differential display technique for cloning and sequencing of the 3′ region from three putative members of wheat HSP70 gene family. Plant Mol Biol 30: 641-646 (1996).
Joshi CP, Nguyen HT: 5′ untranslated leader sequences of eukaryotic mRNAs encoding heat shock induced proteins. Nucl Acids Res 23: 541-549 (1995).
Joshi CP, Nguyen HT: Differential display-mediated rapid identification of different members of a multigene family,HSP16.9 in wheat. Plant Mol Biol 31: 575-584 (1996).
Knack G, Kloppstech K: cDNA sequence of a heat-inducible protein of Chenopodiumsharing little homology with other heat-shock proteins. Nucl Acids Res 17: 5380 (1989).
Knack G, Liu Z, Kloppstech K: Low molecular mass heatshock proteins of a light-resistant photoautotrophic cell culture. Eur J Cell Biol 59: 166-175 (1992).
Kyte J, Doolittle RF: Asimplemethod for displaying the hydropathic character of a protein. J Mol Biol 157: 105-132 (1982).
LaFayette PR, Nagao RT, O'Grady K, Vierling E, Key JL: Molecular characterization of cDNAs encoding lowmolecular-weight heat shock proteins of soybean. Plant Mol Biol 30: 159-169 (1996).
Laszlo A, Li GC: Heat resistant variants of Chinese hamster fibroblasts altered in expression of heat shock protein. Proc Natl Acad Sci USA 82: 8029-8033 (1985).
Lee JH, Hüber A, Schöffl F: Derepression of the activity of genetically engineered heat-shock factor causes constitutive synthesis of heat-shock proteins and increased thermotolerance in transgenic Arabidopsis. Plant J 8: 603-612 (1995).
Lee JH, Schöffl F:AnHSP70 antisense gene affects the expression of HSP70/HSC70, the regulation of HSF, and the acquisition of thermotolerance in transgenic Arabidopsis thaliana. Mol Gen Genet 252: 11-19 (1996).
Lenne C, Block MA, Garin J, Douce R: Sequence and expression of the mRNA encoding HSP22, the mitochondrial small heat-shock protein in pea leaves. Biochem J 311: 805-813 (1995).
Lenne C, Douce R: A low molecular mass heat-shock protein is localized to higher plant mitochondria. Plant Physiol 105: 1255-1261 (1994).
Liang P, Averboukh L, Pardee AB: Distribution and cloning of eukaryotic mRNAs by means of differential display: refinements and optimization. Nucl Acids Res 21: 3269-3275 (1993).
Liang P, Pardee AB: Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 257: 967-971 (1992).
Lindquist S: The heat-shock response. Annu Rev Biochem 55: 1151-1191 (1986).
Lindquist S, Craig EA: The heat shock proteins. Annu Rev Genet 22: 631-677 (1988).
Loomis WF, Wheeler SA: Chromatin-associated heat shock proteins in Dictyostelium. Devel Biol 90: 412-418 (1982).
Marmiroli N, Maestri E, Terzi V, Gulli M, Pavesi A, Raho G, Lupotto E, Di Cola G, Sinibaldi R, Perrotta C: Genetic and molecular evidences of the regulation of gene expression during heat shock in plants. In: Cherry JH (ed) Biochemical and Cellular Mechanisms of Stress Tolerance in Plants. NATOASI Series, Series H: Cell Biology, vol. 86, pp. 157-190. Springer-Verlag, Berlin (1993).
Morimoto RI, Tissiéres A, Georgopoulos C: The stress response, function of the proteins, and perspectives. In: Morimoto RI, Tissiéres A, Georgopoulos C (eds) Stress Proteins in Biology and Medicine, pp. 1-36. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY (1990).
Mount SM: A catalogue of splice junction sequences. Nucl Acids Res 10: 1406-1410 (1981).
Murashige T, Skoog F: A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473-497 (1962).
Nover L: Gene technology and functional analyses of heat shock genes. In: Nover L (ed), Heat-Shock Response, pp. 167-220. CRC Press, Boca Raton, FL (1991).
Nover L: Induced thermotolerance. In: Nover L (ed), Heat-Shock Response, pp. 409-452. CRC Press, Boca Raton, FL (1991).
Osteryoung KW, Sundberg H, Vierling E: Poly(A) tail length of a heat-shock protein RNA is increased by severe heat-stress, but intron splicing is unaffected. Mol Gen Genet 239: 323-333 (1993).
Reiter RS, Young RM, Scolnik PA: Genetic linkage of the Arabidopsisgenome: methods for mapping with recombinant inbreds and random amplified polymorphic DNAs (RAPDs). In: Koncz C, Chua NH, Schell J (eds) Methods in Arabidopsis Research, pp. 170-190.World Scientific Publishing, Singapore (1992).
Ritossa FM: A new puffing pattern induced by a temperature shock and DNP in Drosophila. Experientia 18: 571-573 (1962).
Sambrook J, Fritsch EF, Maniatis T: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY (1989).
Sanchez Y, Lindquist SL: HSP104 required for induced thermotolerance. Science 248: 1112-1115 (1990).
Sanger F, Nicklen S, Coulson AR: DNA sequencing with chainterminating inhibitors. Proc Natl Acad Sci USA 74: 5463 (1977).
Schöffl F, Bauman G, Raschke E: The expression of heat shock genes-a model for environmental stress response. In: Verma DPS, Goldberg RB (eds) Temporal and Spatial Regulation of Plant Genes, pp. 253-273. Springer-Verlag, Wien (1988).
Sharma YK, Davis KR: Isolation of a novel Arabidopsisozoneinduced cDNA by differential display. Plant Mol Biol 29: 91-98 (1995).
Sinibaldi RM, Mettler I: Intron splicing and intron-mediated enhanced expression in monocots. Progr Nucl Acids Res Mol Biol 42: 229-257 (1992).
Sinibaldi RM, Turpen T: Aheat shock protein is encodedwithin mitochondria of higher plants. J Biol Chem 260: 15382-15385 (1985).
Sullivan ML, Green PJ: Post-transcriptional regulation of nuclear-encoded genes in higher plants: the roles of mRNA stability and translation. PlantMol Biol 23: 1091-1104 (1993).
Takahashi T, Komeda Y: Characterization of two genes encoding small heat-shock proteins in Arabidopsis thaliana. Mol Gen Genet 219: 365-372 (1989).
Takahashi T, Naito S, Komeda Y: Isolation and analysis of the expression of two genes for the 81-kilodalton heat-shock proteins from Arabidopsis. Plant Physiol 99: 383-390 (1992).
van Tunen AJ, Koes RE, Spelt CE, van der Krol AR, Stuitje AR, Moe JNM: Cloning of two chalcone flavanone isomerase genes from Petunia hybrida: coordinate, light regulated and differential expression of flavonoid gene. EMBO J 7: 1257-1263 (1988).
Vierling E: The role of heat shock proteins in plants. Annu Rev Plant Physiol Plant Mol Biol 42: 579-620 (1991).
Vierling E, Nagao RT, DeRocher AE, Harris L: A heat shock protein localized to chloroplast is a member of a eukaryotic superfamily of heat shock proteins. EMBO J 7: 575-581 (1988).
Visioli G, Rossi P, Maestri E, Marmiroli N: Molecular and genetic analysis of thermotolerance. In: Leigh RA, Blake-Kalff MMAM(eds) Proceedings of the second STRESSNET conference, pp. 77-81. European Commission, Directorate General VI (1995).
Vogel JL, Parsell DA, Lindquist S: Heat-shock proteinsHsp104 and Hsp70 reactivate mRNA splicing after heat inactivation. Curr Biol 5: 306-317 (1995).
Waters ER: The molecular evolution of the small heat-shock proteins in plants. Genetics 141: 785-795 (1995).
Welte MA, Tetrault JM, Dellavalle RP, Lindquist SL: A new method for manipulating transgenes: engineering heat tolerance in a complex, multicellular organism. Curr Biol 3: 805-915 (1993).
Winter J, Wright R, Duck N, Gasser C, Fraley R, Shah D: The inhibition of petunia hsp70 mRNA processing during CdCl2 stress. Mol Gen Genet 211: 315-319 (1988).
Xiao C-M, Mascarenhas JP: High temperature-induced thermotolerance in pollen tubes of Tradescantiaand heat-shock proteins. Plant Physiol 78: 887-890 (1985).
Yahara I, Iida H, Koyasu S: A heat shock-resistant variant of Chinese hamster cell line constitutively expressing heat shock protein of Mr 90 000 at high level. Cell Struct Funct 11: 65-73 (1986).
Yost HJ, Lindquist S: Heat shock proteins affect RNA processing during the heat shock response of Saccharomyces cerevisiae. Mol Cell Biol 11: 1062-1068 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Visioli, G., Maestri, E. & Marmiroli, N. Differential display-mediated isolation of a genomic sequence for a putative mitochondrial LMW HSP specifically expressed in condition of induced thermotolerance in Arabidopsis thaliana (L.) Heynh.. Plant Mol Biol 34, 517–527 (1997). https://doi.org/10.1023/A:1005824314022
Issue Date:
DOI: https://doi.org/10.1023/A:1005824314022