Abstract
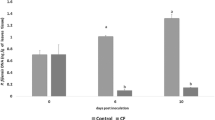
To study whether the immunoreactions of banana contribute the pathogenic differences between physiological races 1 (Foc1) and 4 (Foc4) of Fusarium oxysporum f. sp. cubense (Foc), we monitored the resistance reaction of Brazilian banana plants treated respectively by inoculation with Foc1 and Foc4, coculture in the medium with pure oligogalacturonides (OGs), and pretreatment with OGs before the inoculation with Foc4 (OGs-Foc4). Our results showed that all four treatments could induce the up-regulated expression of the defense-related genes and increase the callose and flavonoid contents. Moreover, the range of the increase induced by Foc1 was higher than that induced by Foc4, and the resistance against Foc4 was stronger in plants pretreated with OGs than in plants that were not pretreated with OGs. We also found that both Foc1 and Foc4 could induce the H2O2 outburst, including the two outbursts of Foc4 and one outburst of Foc1. Although no significant change of H2O2 was induced by OGs alone, the contents of H2O2 production in plants treated with OGs-Foc4 were lower than in plants treated with Foc4 alone. This result indicates that the pretreatment with OGs can inhibit the H2O2 production during the Foc4 infection, resulting in a reduction in the occurrence of necrotic hypersensitive reaction. These results suggest that the immune reactions of banana are involved in the regulation of different pathogenicities of Foc1 and Foc4. Moreover, OGs may play an important intermediate role in inducing the immune responses.





Similar content being viewed by others
References
Altamura MM, Zaghi D, Salvi G, De Lorenzo G, Bellincampi D (1998) Oligogalacturonides stimulate pericycle cell wall thickening and cell divisions leading to stoma formation in tobacco leaf explants. Planta 204:429–436
Aziz A, Heyraud A, Lambert B (2004) Oligogalacturonide signal transduction, induction of defense-related responses and protection of grapevine against Botrytis cinerea. Planta 218:767–774
Bai TT, Xie WB, Zhou PP, Wu ZL, Xiao WC, Zhou L, Sun J, Ruan RL, Li HP (2013) Transcriptome and expression profile analysis of highly resistant and susceptible banana roots challenged with Fusarium oxysporum f. sp. cubense tropical race 4. PLoS One 8(9):e73945. https://doi.org/10.1371/journal.pone.0073945
Baxter A, Mittler R, Suzuki N (2014) ROS as key players in plant stress signaling. J Exp Bot 65:1229–1240
Bellincampi D, Cervone F, Lionetti V (2014) Plant cell wall dynamics and wall-related susceptibility in plant-pathogen interactions. Front Plant Sci 5. https://doi.org/10.3389/fpls.2014.00228
Bellincampi D, Dipierro N, Salvi G, Cervone F, De Lorenzo G (2000) Extracellular H2O2 induced by oligogalacturonides is not involved in the inhibition of the auxin-regulated rolB gene expression in tobacco leaf explants. Plant Physiol 122:1379–1386
Benedetti M, Pontiggia D, Raggi S, Cheng Z, Scaloni F, Ferrari S, Ausubel FM, Cervone F, De Lorenzo G (2015) Plant immunity triggered by engineered in vivo release of oligogalacturonides, damage-associated molecular patterns. Proc Natl Acad Sci 112:5533–5538
Boller T, Felix G (2009) A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu Rev Plant Biol 60:379–406
Bonnin E, Garnier C, Ralet M (2014) Pectin-modifying enzymes and pectin-derived materials: applications and impacts. Appl Microbiol Biot 98:519–532
Bosch M, Hepler PK (2005) Pectin methylesterases and pectin dynamics in pollen tubes. Plant Cell 17:3219–3226
Camejo D, Martí MC, Jiménez A, Cabrera JC, Olmos E, Sevilla F (2011) Effect of oligogalacturonides on root length, extracellular alkalinization and O2−-accumulation in alfalfa. J Plant Physiol 168:566–575
Camejo D, Martí MC, Olmos E, Torres W, Sevilla F, Jiménez A (2012) Oligogalacturonides stimulate antioxidant system in alfalfa roots. Biol Plant 56:537–544
Chen JC, Jiang CZ, Gookin TE, Hunter DA, Clark DG, Reid MS (2004) Chalcone synthase as a reporter in virus-induced gene silencing studies of flower senescence. Plant Mol Biol 55:521–530
Chen JY, Wen PF, Kong WF, Pan QH, Zhan JC, Li JM, Wan SB, Huang WD (2006) Effect of salicylic acid on phenylpropanoids and phenylalanine ammonia-lyase in harvested grape berries. Postharvest Biol Tec 40:64–72
Chen L, Zhong H, Kuang J, Li J, Lu W, Chen J (2011) Validation of reference genes for RT-qPCR studies of gene expression in banana fruit under different experimental conditions. Planta 234:377–390
Davidsson P, Broberg M, Kariola T, Sipari N, Pirhonen M, Palva ET (2017) Short oligogalacturonides induce pathogen resistance-associated gene expression in Arabidopsis thaliana. BMC Plant Biol 17. https://doi.org/10.1186/s12870-016-0959-1
De Lorenzo G, Brutus A, Savatin DV, Sicilia F, Cervone F (2011) Engineering plant resistance by constructing chimeric receptors that recognize damage-associated molecular patterns (DAMPs). FEBS Lett 585:1521–1528
Denoux C, Galletti R, Mammarella N, Gopalan S, Werck D, De Lorenzo G, Ferrari S, Ausubel FM, Dewdney J (2008) Activation of defense response pathways by OGs and Flg22 elicitors in Arabidopsis seedlings. Mol Plant 1:423–445
Fan H, Dong H, Xu C, Liu J, Hu B, Ye J, Mai G, Li H (2017) Pectin methylesterases contribute the pathogenic differences between races 1 and 4 of Fusarium oxysporum f. Sp. cubense. Sci. Rep-UK:7. https://doi.org/10.1038/s41598-017-13625-4
Ferrari S (2013) Oligogalacturonides: plant damage-associated molecular patterns and regulators of growth and development. Front Plant Sci 4. https://doi.org/10.3389/fpls.2013.00049
Ferrari S, Galletti R, Denoux C, De Lorenzo G, Ausubel FM, Dewdney J (2007) Resistance to Botrytis cinerea induced in Arabidopsis by elicitors is independent of salicylic acid, ethylene, or jasmonate signaling but requires phytoalexin deficient3. Plant Physiol 144:367–379
Galletti R, Denoux C, Gambetta S, Dewdney J, Ausubel FM, De Lorenzo G, Ferrari S (2008) The Atrboh D-mediated oxidative burst elicited by oligogalacturonidesin Arabidopsisis dispensable for the activation of defense responses effective against Botrytis cinerea. Plant Physiol 148:1695–1706
Galletti R, Ferrari S, De Lorenzo G (2011) Arabidopsis MPK3 and MPK6 play different roles in basal and oligogalacturonide- or flagellin-induced resistance against Botrytis cinerea. Plant Physiol 157:804–814
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophicand necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Guo L, Yang L, Liang C, Wang G, Dai Q, Huang J (2015) Differential colonization patterns of bananas (Musa spp.) by physiological race 1 and Rrace 4 isolates of Fusarium oxysporum f. Sp. cubense. J Phytopathol 163:807–817
Hwang S, Ko W (2004) Cavendish banana cultivars resistant to Fusarium wilt acquired through somaclonal variation in Taiwan. Plant Dis 88:580–588
Jia Z, Tang M, Wu J (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 4:555–559
Jin DF, West CA (1984) Characteristics of galacturonicacid oligomers as elicitors of casbene synthesase activity in castor bean seedlings. Plant Physiol 74:989–992
Kohle H, Jeblick W, Poten F, Blaschek W, Kauss H (1985) Chitosan-elicited callose synthesis in soybean cells as a ca-dependent process. Plant Physiol 77:544–551
Kushalappa AC, Yogendra KN, Karre S (2016) Plant innate immune response: qualitative and quantitative resistance. Crit Rev Plant Sci 35:38–55
Li C, Shao J, Wang Y, Li W, Guo D, Yan B, Xia Y, Peng M (2013b) Analysis of banana transcriptome and global gene expression profiles in banana roots in response to infection by race 1 and tropical race 4 of Fusarium oxysporum f. Sp. cubense. BMC Genomics 14:851. https://doi.org/10.1186/1471-2164-14-851
Li XS, Bai TT, Li YF, Ruan XL, Li HP (2013a) Proteomic analysis of Fusarium oxysporum f. Sp. cubense tropical race 4-inoculated response to fusarium wilts in the banana root cells. Proteome Sci 11:41. https://doi.org/10.1186/1477-5956-11-41
Ma L, Jiang S, Lin G, Cai J, Ye X, Chen H, Li M, Li H, Takac T, Samaj J, Xu C (2013) Wound-induced pectin methylesterases enhance banana (Musa spp. AAA) susceptibility to Fusarium oxysporum f. sp. cubense. J Exp Bot 64:2219–2229
Moscatiello R, Baldan B, Squartini A, Mariani P, Navazio L (2012) Oligogalacturonides: novel signaling molecules in rhizobium-legume communications. Mol Plant Microbe Interact 25:1387–1395
Oh Y, Baldwin IT, Galis I (2013) A Jasmonate ZIM-domain protein NaJAZ dregulates floral jasmonic acid levels and counteracts flower abscission in Nicotiana attenuata plants. PLoS One 8. https://doi.org/10.1371/journal.pone.0057868
Patterson BD, MacRae EA, Ferguson IB (1984) Estimation of hydrogen peroxide in plant extracts using titanium(IV). Anal Biochem 139:487
Ploetz RC (2006) Fusarium wilt of banana is caused by several pathogens referred to as Fusarium oxysporum f. Sp. cubense. Phytopathology 96:653–656
Schijlen EGWM, de Vos CHR, Martens S, Jonker HH, Rosin FM, Molthoff JW, Tikunov YM, Angenent GC, van Tunen AJ, Bovy AG (2007) RNA interference silencing of chalconesynthase, the first step in the flavonoid biosynthesis pathway, leads to parthenocarpic tomato fruits. Plant Physiol 144:1520–1530
Sivankalyani V, Feygenberg O, Diskin S, Wright B, Alkan N (2016) Increased anthocyanin and flavonoids in mango fruit peel are associated with cold and pathogen resistance. Postharvest Biol. Tec. 111:132–139
Sutanto A, Sukma D, Hermanto C, Sudarsono S (2014) Isolation and characterization of resistance gene analogue (RGA) from Fusarium resistant banana cultivars. Emir J Food Agr 26:508–518
Treml J, Šmejkal K (2016) Flavonoids as potent scavengers of hydroxyl radicals. Compr Rev Food Sci F 15:720–738
Wagner U, Edwards R, Dixon DP, Mauch F (2002) Probing the diversity of the Arabidopsis glutathione s-transferase gene family. Plant Mol Biol 49:515–532
Wiesel L, Newton AC, Elliott I, Booty D, Gilroy EM, Birch PRJ, Hein I (2014) Molecular effects of resistance elicitors from biological origin and their potential for crop protection. Front Plant Sci 5. https://doi.org/10.3389/fpls.2014.00655
Wirthmueller L, Maqbool A, Banfield MJ (2013) On the front line: structural insights into plant–pathogen interactions. Nat Rev Microbiol 11:761–776
Xie B, Deng Y, Kanaoka MM, Okada K, Hong Z (2012) Expression of Arabidopsis callose synthase 5 results in callose accumulation and cell wall permeability alteration. Plant Sci 183:1–8
Yang C, Sathyapriya H, Wong M (2016) Characterisation of pathogenesis-related genes and resistance gene candidates in banana (Musa acuminata) and their expression during host-pathogen interaction. Pertanika J Trop Agric Sci 39:55–72
Acknowledgements
The study was supported by the earmarked fund for Modern Agro-industry Technology Research System (CARS-31-09) in China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, H., Lei, Z., Dong, H. et al. Immune responses in Brazilian banana determining the pathogenic differences between the physiological races 1 and 4 of Fusarium oxysporum f. sp. cubense. J Plant Pathol 101, 225–234 (2019). https://doi.org/10.1007/s42161-018-0165-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-018-0165-0