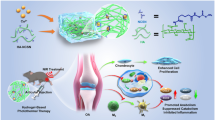
Abstract
Cell-based regenerative medicine strategies hold a great potential to revolutionize the treatment of a large number of injuries with limited regenerative potential. However, the effectiveness of the simple injection of a cell suspension in a target site/tissue of action is often limited by the dispersion of cells toward other tissues, hindering their therapeutic action. Nevertheless, the development of a custom-made cell carrier that can perfectly fit a patient’s defect and be ready on demand is still a challenging task. The present study proposes the development of an off-the-shelf injectable cell delivery system combining a photocross-linkable hyaluronic acid (HA) matrix enriched with platelet lysate (PL) and human adipose tissue-derived stem cells (hASCs), which can be stored using standard cryopreservation methods and used when required. The obtained results indicate that the mechanical and viscoelastic properties of the system are improved in the presence of cells and no significant differences were identified between cell-laden hydrogels produced before or after cryopreservation. In PL-enriched hydrogels, cells tend to better recover from cryopreservation maintaining the values of cell viability and DNA content. Moreover, viable cells laden in our system and expressing stemness markers were detected after 21 days in culture. Altogether, the results obtained in this work demonstrate the potential of the developed strategy as an injectable cell delivery system for ready-to-use applications or as a cryopreserved product to be available on demand for cell-based therapies.









Similar content being viewed by others
References
Snyder TN, Madhavan K, Intrator M, Dregalla RC, Park D. A fibrin/hyaluronic acid hydrogel for the delivery of mesenchymal stem cells and potential for articular cartilage repair. J Biol Eng. 2014;8:1. doi:10.1186/1754-1611-8-27.
Tobita M, Tajima S, Mizuno H. Adipose tissue-derived mesenchymal stem cells and platelet-rich plasma: stem cell transplantation methods that enhance stemness. Stem Cell Res Ther. 2015;6:215. doi:10.1186/s13287-015-0217-8.
Goncalves AI, Rodrigues MT, Lee SJ, Atala A, Yoo JJ, Reis RL, et al. Understanding the role of growth factors in modulating stem cell Tenogenesis. PLoS One. 2013;8(12):10. doi:10.1371/journal.pone.0083734.
Mihaila SM, Frias AM, Pirraco RP, Rada T, Reis RL, Gomes ME, et al. Human adipose tissue-derived SSEA-4 subpopulation multi-differentiation potential towards the endothelial and osteogenic lineages. Tissue Eng A. 2013;19(1–2):235–46. doi:10.1089/ten.tea.2012.0092.
Karp JM, Teol GSL. Mesenchymal stem cell homing: the devil is in the details. Cell Stem Cell. 2009;4(3):206–16. doi:10.1016/j.stem.2009.02.001.
Costa-Almeida R, Granja PL, Soares R, Guerreiro SG. Cellular strategies to promote vascularisation in tissue engineering applications. Eur Cells Mater. 2014;28:51–67.
Neves LS, Rodrigues MR, Reis RL, Gomes ME. Current approaches and future perspectives on strategies for the development of personalized tissue engineering therapies. Expert Rev Precis Med Drug Dev. 2016;1(1):93–108. doi:10.1080/23808993.2016.1140004.
Babo P, Santo VE, Duarte ARC, Correia C, Costa MHG. Platelet lysate membranes as new autologous templates for tissue engineering applications. Inflamm Regen. 2014;34:033–44.
Goei N, Liem IK, Pawitan JA, Mediana D. Effect of platelet rich plasma on post cryopreservation viability, morphology and proliferation of human umbilical cord stem cells. J Biol Sci. 2015;15(2):42–8. doi:10.3844/ojbsci.2015.42.48.
Balakrishnan B, Banerjee R. Biopolymer-based hydrogels for cartilage tissue engineering. Chem Rev. 2011;111(8):4453–74. doi:10.1021/cr100123h.
Anseth KS, Metters AT, Bryant SJ, Martens PJ, Elisseeff JH, Bowman CN. In situ forming degradable networks and their application in tissue engineering and drug delivery. J Control Release. 2002;78(1–3):199–209. doi:10.1016/s0168-3659(01)00500-4.
Popa EG, Rodrigues MT, Coutinho DF, Oliveira MB, Mano JF, Reis RL, et al. Cryopreservation of cell laden natural origin hydrogels for cartilage regeneration strategies. Soft Matter. 2013;9(3):875–85. doi:10.1039/c2sm26846a.
Kawasaki K, Ochi M, Uchio Y, Adachi N, Matsusaki M. Hyaluronic acid enhances proliferation and chondroitin sulfate synthesis in cultured chondrocytes embedded in collagen gels. J Cell Physiol. 1999;179(2):142–8. doi:10.1002/(sici)1097-4652(199905)179:2%3C142::aid-jcp4%3E3.0.co;2-q.
Ialenti A, Di Rosa M. Hyaluronic-acid modulates acute and chronic inflammation. Agents and Actions. 1994;43(1–2):44–7. doi:10.1007/bf02005763.
Cai SS, Liu YC, Shu XZ, Prestwich GD. Injectable glycosaminoglycan hydrogels for controlled release of human basic fibroblast growth factor. Biomaterials. 2005;26(30):6054–67. doi:10.1016/j.biomaterials.2005.03.012.
Solchaga LA, Dennis JE, Goldberg VM, Caplan AI. Hyaluronic acid-based polymers as cell carriers for tissue-engineered repair of bone and cartilage. J Orthop Res. 1999;17(2):205–13. doi:10.1002/jor.1100170209.
Shu XZ, Liu YC, Palumbo FS, Lu Y, Prestwich GD. In situ crosslinkable hyaluronan hydrogels for tissue engineering. Biomaterials. 2004;25(7–8):1339–48. doi:10.1016/j.biomaterials.2003.08.014.
Noble PW. Hyaluronan and its catabolic products in tissue injury and repair. Matrix Biol. 2002;21(1):25–9. doi:10.1016/S0945-053x(01)00184-6.
Santos VE, Gomes ME, Mano JF. Chitosan–chondroitin sulphate nanoparticles for controlled delivery of platelet lysates in bone regenerative medicine. J Tissue Eng Regen Med. 2012;6(S3):S47–S59.
Smeds KA, Grinstaff MW. Photocrosslinkable polysaccharides for in situ hydrogel formation (vol 54, pg 115, 2000). J Biomed Mater Res. 2001;55(2):254–5. doi:10.1002/1097-4636(200105)55:2%3C254::aid-jbm1012%3E3.0.co;2-5.
Jeffery AF, Churchward MA, Mushahwar VK, Todd KG, Elias AL. Hyaluronic acid-based 3D culture model for in vitro testing of electrode biocompatibility. Biomacromolecules. 2014;15(6):2157–65. doi:10.1021/bm500318d.
Mihaila SM, Gaharwar AK, Reis RL, Marques AP, Gomes ME, Khademhosseini A. Photocrosslinkable kappa-carrageenan hydrogels for tissue engineering applications. Advanced Healthcare Materials. 2013;2(6):895–907. doi:10.1002/adhm.201200317.
Carvalho PP, Wu XY, Yu G, Dias IR, Gomes ME, Reis RL, et al. The effect of storage time on adipose-derived stem cell recovery from human Lipoaspirates. Cells Tissues Organs. 2011;194(6):494–500. doi:10.1159/000324892.
Rada T, Reis RL, Gomes ME. Distinct stem cells subpopulations isolated from human adipose tissue exhibit different chondrogenic and osteogenic differentiation potential. Stem Cell Rev Rep. 2011;7(1):64–76. doi:10.1007/s12015-010-9147-0.
Crespo-Diaz R, Behfar A, Butler GW, Padley DJ, Sarr MG, Bartunek J, et al. Platelet lysate consisting of a natural repair proteome supports human mesenchymal stem cell proliferation and chromosomal stability. Cell Transplant. 2011;20(6):797–811. doi:10.3727/096368910x543376.
Fekete N, Gadelorge M, Furst D, Maurer C, Dausend J, Fleury-Cappellesso S, et al. Platelet lysate from whole blood-derived pooled platelet concentrates and apheresis-derived platelet concentrates for the isolation and expansion of human bone marrow mesenchymal stromal cells: production process, content and identification of active components. Cytotherapy. 2012;14(5):540–54. doi:10.3109/14653249.2012.655420.
Coutinho DF, Sant SV, Shin H, Oliveira JT, Gomes ME, Neves NM, et al. Modified Gellan gum hydrogels with tunable physical and mechanical properties. Biomaterials. 2010;31(29):7494–502. doi:10.1016/j.biomaterials.2010.06.035.
Costa AMS, Mano JF. Highly robust hydrogels via a fast, simple and cytocompatible dual crosslinking-based process. Chem Commun. 2015;51(86):15673–6. doi:10.1039/c5cc05564d.
Lefebvre CA, Schuster GS. Biocompatibility of visible-light-cured resin in prosthodontics. J Prosthet Dent. 1994;71(2):178–85. doi:10.1016/0022-3913(94)90028-0.
Yalug S, Yazicioglu H. An alternative approach to fabricating a meatus obturator prosthesis. J Oral Sci. 2003;45:43–5.
Domingues RMA, Silva M, Gershovich P, Betta S, Babo P, Caridade SG, et al. Development of injectable hyaluronic acid/cellulose nanocrystals bionanocomposite hydrogels for tissue engineering applications. Bioconjug Chem. 2015;26(8):1571–81. doi:10.1021/acs.bioconjchem.5b00209.
Babo P, Pires R, Santos L, Franco A, Rodrigues F, Leonor I et al. Platelet lysate-loaded photocrosslinkable hyaluronic acid hydrogels exhibit Mitogenic and anti-microbial properties. ACS Biomaterials Science and Engineering. 2016. p. 1–11.
Popa EG. Chondrogenic potential of injectable κ-carrageenan hydrogel with encapsulated adipose stem cells for cartilage tissue-engineering applications. J Tissue Eng Regen Med. 2015;9(5):550–63. doi:10.1002/term.1683.
Nettles DL, Vail TP, Morgan MT, Grinstaff MW, Setton LA. Photocrosslinkable hyaluronan as a scaffold for articular cartilage repair. Ann Biomed Eng. 2004;32(3):391–7. doi:10.1023/b:abme.0000017552.65260.94.
Becher J, Moller S, Schnabelrauch M. Phase transfer-catalyzed synthesis of highly acrylated hyaluronan. Carbohydr Polym. 2013;93(2):438–41. doi:10.1016/j.carbpol.2012.12.056.
Bahney CS, Lujan TJ, Hsu CW, Bottlang M, West JL, Johnstone B. Visible light photoinitiation of mesenchymal stem cell-laden bioresponsive hydrogels. Eur Cells Mater. 2011;22:43–55.
Afrimzon E, Zurgil N, Shafran Y, Ehrhart F, Namer Y, Moshkov S, et al. The individual-cell-based cryo-chip for the cryopreservation, manipulation and observation of spatially identifiable cells. II: Functional activity of cryopreserved cells. BMC Cell Biology. 2010;11:13. doi:10.1186/1471-2121-11-83.
Atashi F, Jaconi MEE, Pittet-Cuenod B, Modarressi A. Autologous platelet-rich plasma: a biological supplement to enhance adipose-derived mesenchymal stem cell expansion. Tissue Engineering Part C-Methods. 2015;21(3):253–62. doi:10.1089/ten.tec.2014.0206.
Warnke PH, Humpe A, Strunk D, Stephens S, Warnke F, Wiltfang J, et al. A clinically-feasible protocol for using human platelet lysate and mesenchymal stem cells in regenerative therapies. J Cranio-Maxillofac Surg. 2013;41(2):153–61. doi:10.1016/j.jcms.2012.07.003.
Doucet C, Ernou I, Zhang YZ, Llense JR, Begot L, Holy X, et al. Platelet lysates promote mesenchymal stem cell expansion: a safety substitute for animal serum in cell-based therapy applications. J Cell Physiol. 2005;205(2):228–36. doi:10.1002/jcp.20391.
Diaz-Gomez L, Alvarez-Lorenzo C, Concheiro A, Silva M, Dominguez F, Sheikh FA, et al. Biodegradable electrospun nanofibers coated with platelet-rich plasma for cell adhesion and proliferation. Materials Science & Engineering C-Materials for Biological Applications. 2014;40:180–8. doi:10.1016/j.msec.2014.03.065.
Yong KW, Pingguan-Murphy B, Xu F, Abas WAW, Choi JR, Omar SZ, et al. Phenotypic and functional characterization of long-term cryopreserved human adipose-derived stem cells. Scientific Reports. 2015;5:10. doi:10.1038/srep09596.
Yoo KH, Jang IK, Lee MW. Comparison of immunomodulatory properties of mesenchymal stem cells derived from adult human tissues. 2009; 259(2):150–6. doi:10.1016/j.cellimm.2009.06.010.
Ivanova-Todorova E, Bochev I, Mourdjeva M, Dimitrov R, Bukarev D, Kyurkchiev S, et al. Adipose tissue-derived mesenchymal stem cells are more potent suppressors of dendritic cells differentiation compared to bone marrow-derived mesenchymal stem cells. Immunol Lett. 2009;126(1–2):37–42. doi:10.1016/j.imlet.2009.07.010.
Zavan B, Vindigni V, Vezzu K, Zorzato G, Luni C, Abatangelo G, et al. Hyaluronan based porous nano-particles enriched with growth factors for the treatment of ulcers: a placebo-controlled study. J Mater Sci Mater Med. 2009;20(1):235–47. doi:10.1007/s10856-008-3566-3.
Fan HH, Vitharana SN, Chen T, O’Keefe D, Middaugh CR. Effects of pH and polyanions on the thermal stability of fibroblast growth factor 20. Mol Pharm. 2007;4(2):232–40. doi:10.1021/mp060097h.
Maity H, Karkaria C, Davagnino J. Effects of pH and arginine on the solubility and stability of a therapeutic protein (fibroblast growth factor 20): relationship between solubility and stability. Curr Pharm Biotechnol. 2009;10(6):609–25.
Acknowledgements
The authors wish to acknowledge the financial support from the Portuguese Foundation for Science and Technology (FCT) for the PhD grant of R.C-A (SFRH/BD/96593/2013), post-doctoral grant of M.T.R (SFRH/BPD/111729/2015), grant of M.E.G. (IF/00685/2012) and Recognize project (UTAP-ICDT/CTM-BIO/0023/2014), RL3-TECT -NORTE-07-0124-FEDER-000020 project co-financed by ON.2 (NSRF) through ERD. The authors also acknowledge the financial support from FCT/MCTES (Fundação para a Ciência e a Tecnologia/Ministério da Ciência, Tecnologia, e Ensino Superior) and the Fundo Social Europeu through Programa Operacional do Capital Humano (FSE/POCH) PD/59/2013 for the PhD grant of A.I.G (PD/BD/113802/2015), and LA ICVS-3Bs (UID/Multi/50026/2013). The authors would also thank to Serviço de Imuno-Hemoterapia, Centro Hospitalar São João, EPE (Porto, Portugal) for providing human platelet concentrate samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors declare no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 134 kb)
Rights and permissions
About this article
Cite this article
Neves, L.S., Babo, P.S., Gonçalves, A.I. et al. Injectable Hyaluronic Acid Hydrogels Enriched with Platelet Lysate as a Cryostable Off-the-Shelf System for Cell-Based Therapies. Regen. Eng. Transl. Med. 3, 53–69 (2017). https://doi.org/10.1007/s40883-017-0029-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40883-017-0029-8