Abstract
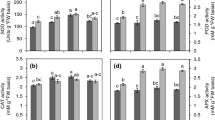
Seedlings of three wheat cultivars viz. PBW 343, C 306 (heat tolerant) and WH 542 (heat susceptible) were subjected to a brief heat shock at 45 °C, followed by transfer to the normal temperature (25 °C) for three days in dark with and without Ca2+ (5 mM CaCl2). Heat shock treatment caused reduction in activities of enzymes, viz., soluble acid invertase, amylases and seedling growth with increase in total free sugar content. This effect was more pronounced in susceptible cv. WH 542 compared with tolerant cvs. PBW 343, C 306. With Ca2+ pretreatment a substantial increase in total sugars and reducing sugars as well as activities of both α and β amylases was observed in both control and stressed seedlings. Under normal temperature Ca2+ had non-significant effect on seedling growth and acid invertase activity, whereas, under stress conditions it improved the shoot and root length with enhancement in acid invertase activity. Thus, an appreciable level of thermo-protection was induced by 5 mM Ca2+ under stress conditions, indicating that Ca2+ level required for growth under heat stress exceeds that required for growth under normal conditions. Elevated Ca2+ concentration might be having a role in alleviating the inhibition of sucrose and starch metabolism, thereby, increasing their utilization for seedling growth under heat stress.





Similar content being viewed by others
References
Aloni, B., Peet, M., Pharr, M., & Karni, L. (2001). The effect of high temperature and high atmospheric CO2 on carbohydrate changes in bell pepper (Capsicum annum) pollen in relation to its germination. Physiologia Plantarum, 112, 505–512.
Asthir, B., Basra, A. S., & Batta, S. K. (1999). Differential response of carbon and nitrogen metabolism to fluoride application in fruiting structures of chickpea (Cicer arietinum). Acta Physiologia Plantarum, 21, 67–73.
Asthir, B., & Singh, R. (1995). Fluoride-induced changes in the activities of sucrose metabolizing enzymes in relation to starch accumulation in sorghum caryopsis, raised through liquid culture. Plant Physiology and Biochemistry, 33, 219–223.
Berenguer, C. L., Alcaraz, C. F., & Carlos, M. (2004). Involvement of sugars in the response of pepper plants to salinity: Effect of calcium application. Asian Journal Plant Science, 3, 455–462.
Bewley, J. D., & Black, M. (1994). Seed physiology of development and germination. New York: Plenum Press.
Bhatia, S., & Singh, R. (2000). Calcium-mediated conversion of sucrose to starch in relation to the activities of amylases and sucrose-metabolizing enzymes in sorghum grains raised through liquid culture. Indian Journal Biochemistry and Biophysics, 37, 135–139.
Bhattacharjee, S. (2008). Calcium-dependent signaling pathway in the heat induced oxidative injury in Amaranthus lividus. Biologia Plantarum, 52, 137–140.
Bhattacharjee, S., & Mukherjee, A. K. (2006). Heat and salinity induced oxidative stress and changes in protein profile in Amaranthus lividus L. Indian Journal Plant Physiology, 11, 41–47.
Blum, A., & Sinmena, B. (1994). Wheat seed endosperm utilization under heat stress and its relation to thermotolerance in autotrophic plant. Field Crop Research, 37, 185–191.
Bohnert, H. J., Nelson, D. E., & Jensen, R. G. (1995). Adaptations to environmental stresses. Plant Cell, 7, 1099–1111.
Cousson, A. (2007). Two calcium mobilizing pathways implicated within abscisic acid- induced stomatal closing in Arabidopsis thaliana. Biologia Plantarum, 51, 285–291.
Duffus, C., & Rosie, R. (1973). Starch hydrolyzing enzyme in developing barley grain. Planta, 109, 153–160.
Gibeaut, D. M., Karuppiah, N., Chang, S. R., Brock, T. G., Vadlamudi, B., Kim, D., et al. (1990). Cell wall and enzyme changes during gravi response of leaf sheath pulvinus of oat (Avena sativa). Plant Physiology, 94, 411–416.
Jones, R. L., & Carbonell, J. (1984). Regulation of the synthesis of barley aleurone α; amylase by gibberellic acid and calcium ions. Plant Physiology, 76, 213–218.
Kaur, K., Gupta, A. K., & Kaur, N. (2004). A comparative study on amylases from seedlings of tall and double dwarf wheat. Journal of Plant Biology, 31, 111–116.
Khanna, A., & Chopra, K. (1995). Influence of temperature on germination and seedling growth and its relationship with amylase activity and respiration in wheat cultivars differing in temperature tolerance. Indian Journal Experimental Biology, 33, 775–779.
Kleinhenz, M. D., & Palta, J. P. (2002). Root zone calcium modulates the response of potato plants to heat stress. Physiologia Plantarum, 115, 111–118.
Knight, H., & Knight, M. R. (1993). Calcium signaling in plants responding to stress. In M. F. Small Ward, C. M. Calvert, & D. J. Bowles (Eds.), Plant responds to environmental stress (pp. 4–9). Oxford: BIOS Scientific Publishers.
Koc, M., Barutcular, C., & Tiryakioglu, M. (2008). Possible heat-tolerant wheat cultivar improvement through the use of flag leaf gas exchange traits in a Mediterranean environment. Journal Science Food Agriculture, 88, 1638–1647.
Kuznetsov, V. V., Rakitin, V. Y., & Zholkevich, V. N. (1999). Effects of preliminary heat shock treatment on accumulation of osmolytes and drought resistance in cotton plants during water deficiency. Physiologia Plantarum, 107, 399–406.
Lauriere, C., Doyen, C., Thevenot, C., & Daussant, J. (1992). β amylase in cereals: A study of the maize beta- amylase system. Plant Physiology, 100, 887–893.
Marina, S. T., Igor, M. A., & Vladimir, V. K. (1999). Calcium is involved in regulation of the synthesis of HSPs in suspension: Cultured sugarbeet cell under hyperthermia. Physiologia Plantarum, 105, 67–73.
Nayyar, H. (2003). Calcium as environmental sensor in plant. Current Science, 84, 893–902.
Paulsen, G. M. (1994). High temperature responses of crop plants. In K. J. Boote, J. M. Bennett, T. Sinclair, & G. M. Paulsen (Eds.), Physiology and determination of crop yield (pp. 365–389). Madison: American Society of Agronomy.
Singh, R., & Asthir, B. (1988). Import of sucrose and its transformation to starch in the developing sorghum caryopsis. Physiologia Plantarum, 74, 58–65.
Sturm, A. (1999). Invertases Primary structures, functions and roles in plant development and sucrose partitioning. Plant Physiology, 121, 1–7.
Sumesh, K. V., Sharma-Natu, P., & Ghildiyal, M. C. (2008). Starch synthase activity and heat shock protein in relation to thermal tolerance of developing wheat grains. Biologia Plantarum, 52, 749–753.
Tsai, C. Y. (1974). Sucrose–UDP glucosyl transferase of Zea mays endosperm. Phytochemistry, 13, 865–891.
Wang, J. B., & Li, R. Q. (1999). Changes of Ca2+ distribution in mesophyll cells of pepper under heat stress. Acta Horticulture Sinica, 26, 57–58.
Zhao, H. J., & Tan, J. F. (2005). Role of calcium ion in protection against heat and high irradiance stress-induced oxidative damage to photosynthesis of wheat leaves. Photosynthetica, 43, 473–476.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhatia, S., Asthir, B. Calcium mitigates heat stress effect in wheat seeding growth by altering carbohydrate metabolism. Ind J Plant Physiol. 19, 138–143 (2014). https://doi.org/10.1007/s40502-014-0087-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-014-0087-6