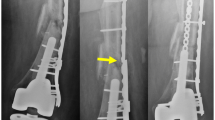
Abstract
Biomechanical analysis, numerical and experimental, has been extensively used for more than 3 decades to investigate the mechanical behaviour of bone and implant–bone structures in joint replacement. In this review article, a detailed overview of the state-of-the-art techniques used for the development and pre-clinical testing of orthopaedic implants has been presented, with special focus on the hip-joint and hip implants. The efficacies of biomechanical analysis in analysing failure mechanisms in joint replacement, its clinical relevance, challenges and limitations, and future directions have been highlighted. Finite element (FE) modelling and analysis have contributed immensely towards testing of clinical hypotheses and evaluation of implant designs. Over the last few decades, the size and sophistication of the FE models have increased considerably. A critical analysis of the currently available subject-specific FE modelling techniques has been presented including, development of computed tomography-scan-based FE models of bone and implant, assignment of heterogeneous bone material properties, loading and boundary conditions. The relationship between stress–strain analysis and implant failure needs careful interpretation for clinical relevance. Verification and validation of these models are essential for assessing the validity of the predicted results. The salient features of adaptive simulations including bone remodelling and tissue differentiation and its prominence in investigating potential failure mechanisms and implant design evaluations have been discussed. The key considerations for design and development of orthopaedic implants have been suggested. It is envisaged that the FE simulations would be more holistic in nature, incorporating the complexities and variabilities of the clinical problems.
Similar content being viewed by others
References
M.I.Z. Ridzwan, C. Sukjamsri, B. Pal, R.J. van Arkel, A. Bell, M. Khanna, A. Baskaradas, R. Abel, O. Boughton, J. Cobb, U.N. Hansen, Femoral fracture type can be predicted from femoral structure: a finite element study validated by digital volume correlation experiments. J. Orthop. Res. 36(3), 993–1001 (2018)
International Osteoporosis Foundation, facts and statistics: osteoporosis—incidence and burden. https://www.iofbonehealth.org/facts-statistics#category-14. Accessed 22 July 2020
World Health Organization, Chronic Rheumatic Conditions. http://www.who.int/chp/topics/rheumatic/en/. Accessed 22 July 2020
M. Taylor, P.J. Prendergast, Four decades of finite element analysis of orthopaedic devices: Where are we now and what are the opportunities? J. Biomech. 48, 767–778 (2015)
NJR, 16th Annual Report of National Joint Registry for England, Wales, Northern Ireland and the Isle of Man (2019). https://reports.njrcentre.org.uk/downloads. Accessed 22 July 2020
AOA 2019, Annual Report of Australian Orthopaedic Association (2018–19). https://www.aoa.org.au/about-aoa/communications/annual-reports. Accessed 22 July 2020
R. Huiskes, Failed innovation in total hip-replacement: diagnosis and proposals for a cure. Acta Orthop. Scand. 64(6), 699–716 (1993)
B. Pal, S. Gupta, A.M. New, A numerical study of failure mechanisms in the cemented resurfaced femur: effects of interface characteristics and bone remodelling. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 223, 471–484 (2009)
B. Pal, S. Gupta, A.M.R. New, Influence of the change in stem length on load transfer and bone remodelling for a cemented resurfaced femur. J. Biomech. 43, 2908–2914 (2010)
B. Pal, S. Gupta, A.M.R. New, Design considerations of ceramic resurfaced femoral head: effect of interface characteristics on failure mechanism. Comput. Methods Biomech. Biomed. Eng. 13(2), 143–155 (2010)
M. Viceconti, S. Affatato, M. Baleani, B. Bordini, L. Cristofolini, F. Taddei, Pre-clinical validation of joint prostheses: a systematic approach. J. Mech. Behav. Biomed. Mater. 2(1), 120–127 (2009)
M. Taylor, R. Bryan, F. Galloway, Accounting for patient variability in finite element analysis of the intact and implanted hip and knee: a review. Int. J. Numer. Methods Biomed. Eng. 29(2), 273–292 (2013)
P. Pankaj, Patient-specific modelling of bone and bone–implant systems: the challenges. Int. J. Numer. Methods Biomed. Eng. 29(2), 233–249 (2013)
A. Erdemir, T.M. Guess, J. Halloran, S.C. Tadepalli, T.M. Morrison, Considerations for reporting finite element analysis studies in biomechanics. J. Biomech. 45(4), 625–633 (2012)
P.J. Laz, M. Browne, A review of probabilistic analysis in orthopaedic biomechanics. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 224(8), 927–943 (2010)
S. Poelert, E. Valstar, H. Weinans, A.A. Zadpoor, Patient-specific finite element modeling of bones. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 227(4), 464–478 (2012)
N. Byrne, M. VelascoForte, A. Tandon, I. Valverde, T. Hussain, A systematic review of image segmentation methodology, used in the additive manufacture of patient-specific 3D printed models of the cardiovascular system. JRSM Cardiovasc. Dis. (2016). https://doi.org/10.1177/2048004016645467
P. Dudek, J. Cieslik, Introduction to reverse engineering and rapid prototyping in medical applications, in Simulations in Medicine: Preclinical and Clinical Applications, ed. by I. Roterman-Konieczna (Walter de Gruyter GmbH & Co KG, Berlin, 2015)
M. Lengsfeld, J. Schmitt, P. Alter, J. Kaminsky, R. Leppek, Comparison of geometry-based and CT voxel-based finite element modelling and experimental validation. Med. Eng. Phys. 20, 515–522 (1998)
M. Viceconti, L. Bellingeri, L. Cristofolini, A. Toni, A comparative study on different methods of automatic mesh generation of human femurs. Med. Eng. Phys. 20, 1–10 (1998)
M. Viceconti, M. Davinelli, F. Taddei, A. Cappello, Automatic generation of accurate subject-specific bone finite element models to be used in clinical studies. J. Biomech. 37(10), 1597–1605 (2004)
F. Taddei, L. Cristofolini, S. Martelli, H.S. Gill, M. Viceconti, Subject-specific finite element models of long bones: an in vitro evaluation of the overall accuracy. J. Biomech. 39(13), 2457–2467 (2006)
A.E. Anderson, C.L. Peters, B.D. Tuttle, J.A. Weiss, Subject-specific finite element model of the pelvis: development, validation and sensitivity studies. J. Biomech. Eng. 127(3), 364–373 (2005)
F. Taddei, A. Pancanti, M. Viceconti, An Improved method for the automatic mapping of computed tomography numbers onto finite element models. Med. Eng. Phys. 26, 61–69 (2004)
J.Y. Rho, M.C. Hobatho, R.B. Ashman, Relations of mechanical properties to density and CT numbers in human bone. Med. Eng. Phys. 17(5), 347–355 (1995)
E.F. Morgan, H.H. Bayraktar, T.M. Keaveny, Trabecular bone modulus density relationships depend on anatomic site. J. Biomech. 36, 897–904 (2003)
E. Schileo, F. Taddei, A. Malandrino, L. Cristofolini, M. Viceconti, Subject-specific finite element models can accurately predict strain levels in long bones. J. Biomech. 40, 2982–2989 (2007)
H. Weinans, D.R. Sumner, R. Igloria, R.N. Natarajan, Sensitivity of periprosthetic stress-shielding to load and the bone density-modulus relationship in subject-specific finite element models. J. Biomech. 33(7), 809–817 (2000)
B. Helgason, E. Perilli, E. Schileo, F. Taddei, S. Brynjólfsson, M. Viceconti, Mathematical relationships between bone density and mechanical properties: a literature review. Clin. Biomech. 23(2), 135–146 (2008)
R. Ghosh, B. Pal, D. Ghosh, S. Gupta, Finite element analysis of a hemi-pelvis: the effect of inclusion of cartilage layer on acetabular stresses and strain. Comput. Methods Biomech. Biomed. Eng. 18(7), 697–710 (2015)
B. Mathai, S. Gupta, Numerical predictions of hip joint and muscle forces during daily activities: a comparison of musculoskeletal models. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 233(6), 636–647 (2019)
B. Mathai, S. Gupta, The influence of loading configurations on numerical evaluation of failure mechanisms in an uncemented femoral prosthesis. Int. J. Numer. Methods Biomed. Eng. (2020). https://doi.org/10.1002/cnm.3353
M. Heller, G. Bergmann, G. Deuretzbacher, L. Durselen, M. Pohl, L. Claes, G.N. Duda, Musculoskeletal loading conditions during walking and stair climbing. J. Biomech. 34, 883–893 (2001)
G. Bergmann, G. Deuretzbacher, M. Heller, F. Graichen, A. Rohlmann, J. Strauss, G. Duda, Hip contact forces and gait patterns from routine activities. J. Biomech. 34, 859–871 (2001)
OrthoLoad, Loading of orthopaedic implants. https://orthoload.com/database/. Accessed 22 July 2020
S. Gupta, F.C.T. van der Helm, J.C. Sterk, F. vanKeulen, Bl Kaptein, Development and experimental validation of a three-dimensional finite element model of the human scapula. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 218, 127–142 (2004)
R. Ghosh, S. Gupta, Bone remodelling around cementless composite acetabular components: the effects of implant geometry and implant–bone interfacial conditions. J. Mech. Behav. Biomed. Mater. 32, 257–269 (2014)
I.A.J. Radcliffe, M. Taylor, Investigation into the affect of cementing techniques on load transfer in the resurfaced femoral head: a multi-femur finite element analysis. Clin. Biomech. 22, 422–430 (2007)
I.A.J. Radcliffe, M. Taylor, Investigation into the effect of varus-valgus orientation on load transfer in the resurfaced femoral head: a multi-femur finite element analysis. Clin. Biomech. 22, 780–786 (2007)
R. Bryan, P. SuryaMohan, F. Galloway, M. Taylor, P.B. Nair, Statistical modelling of the whole human femur incorporating geometric and material properties. Med. Eng. Phys. 32(1), 57–65 (2010)
F. Galloway, M. Kahnt, H. Ramm, P. Worsley, S. Zachow, P. Nair, M. Taylor, A large scale finite element study of a cementless osseointegrated tibial tray. J. Biomech. 46(11), 1900–1906 (2013)
M. Viceconti, S. Olsen, L. Nolte, K. Burton, Extracting clinically relevant data from finite element simulations. Clin. Biomech. 20(5), 451–454 (2005)
A.E. Anderson, B.J. Ellis, J.A. Weiss, Verification, validation and sensitivity studies in computational biomechanics. Comput. Methods Biomech. Biomed. Eng. 10(3), 171–184 (2007)
R. Zdero, S. Shah, P. Goshulak, Surface strain gage testing of whole bone and implants: Chapter 3, in Experimental Methods in Orthopaedic Biomechanics, ed. by R. Zdero (Academic Press, London, 2017)
L. Cristofolini, S.A. Teutonico, L. Monti, A. Cappello, A. Toni, Comparative in vitro study on the long term performance of cemented hip stem: validation of protocol to discriminate between ‘good’ and ‘bad’ design. J. Biomech. 36, 1603–1615 (2003)
J. Eng, L. Cristofolini, E. Varini, M. Viceconti, In vitro method for assessing femoral implant: bone micromotions in resurfacing hip implants under different loading conditions. Proc. Inst. Mech. Eng. Part H. Med. 221, 943–950 (2007)
M. Papini, R. Zdero, E.H. Schemitsch, P. Zalzal, The biomechanics of human femurs in axial and torsional loading: comparison of finite element analysis, human cadaveric femurs and synthetic femurs. J. Biomech. Eng. 129(1), 12–19 (2007)
B. Pal, S. Gupta, A.M.R. New, M. Browne, Strain and micromotion in intact and resurfaced composite femurs: experimental and numerical investigations. J. Biomech. 43, 1923–1930 (2010)
R. Ghosh, S. Gupta, A. Dickinson, M. Browne, Experimental validation of numerically predicted strain and micromotion in intact and implanted composite hemi-pelvises. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 227(2), 162–174 (2012)
J. Eng, M. Tarala, D. Janssen, A. Telka, D. Waanders, N. Verdonschot, Experimental versus computational analysis of micromotions at the implant– bone interface. Proc. Inst. Mech. Eng. Part H Med. 225, 8–15 (2011)
J.W. Dally, W.F. Railey, Experimental Stress Analysis, 3rd edn. (McGraw-Hill College, Boston, 1991)
R. Ghosh, S. Gupta, A. Dickinson, M. Browne, Experimental validation of finite element models of intact and implanted composite hemipelvises using digital image correlation. J. Biomech. Eng. (2012). https://doi.org/10.1115/1.4007173
H. Everitt, S.L. Evans, C.A. Holt, R. Bigsby, I. Khan, Acetabular component deformation under rim loading using digital image correlation and finite element methods. Appl. Mech. Mater. 24–25, 275–280 (2010)
E. Tayton, S. Evans, D. O’Doherty, Mapping the strain distribution on the proximal femur with titanium and flexible stemmed implants using digital image correlation. J. Bone Jt. Surg. Br. 92(8), 1176–1181 (2010)
A.S. Dickinson, A.C. Taylor, H. Ozturk, M. Browne, Experimental validation of a finite element model of the proximal femur using digital image correlation and a composite bone model. J. Biomech. Eng. 133(1), 1–6 (2011)
R. Tiossi, L. Lin, R.C.S. Rodrigues, Y.C. Heo, H.J. Conrad, M.G.C. Mattos, R.F. Ribeiro, A.S.L. Fok, Digital image correlation analysis of the load transfer by implant-supported restorations. J. Biomech. 44(6), 1008–1013 (2011)
A.S. Dickinson, A.C. Taylor, M. Browne, The influence of acetabular cup material on pelvis cortex surface strains, measured using digital image correlation. J. Biomech. 45(4), 719–723 (2012)
T.A. Correa, B. Pal, R.J. van Arkel, F. Vanacore, A. Amis, Reduced tibial strain-shielding with extraosseous total knee arthroplasty revision system. Med. Eng. Phys. 62, 22–28 (2018)
S. Chanda, A. Dickinson, S. Gupta, M. Browne, Full-field in vitro measurements and in silico predictions of strain shielding in the implanted femur after total hip arthroplasty. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 229(8), 549–559 (2015)
K. Madi, G. Tozzi, Q.H. Zhang, J. Tong, A. Cossey, A. Au, D. Hollis, F. Hild, Computation of full-field displacements in a scaffold implant using digital volume correlation and finite element analysis. Med. Eng. Phys. 35, 1298–1312 (2013)
M. Peña-Fernández, A. Barber, G.W. Blunn, G. Tozzi, Optimization of digital volume correlation computation in SR-microCT images of trabecular bone and bone-biomaterials system. J. Microsc. 272(3), 213–228 (2018)
F. Gillard, R. Boardman, M. Mavrogordato, D. Hollis, I. Sinclair, F. Pierron, M. Browne, The application of digital volume correlation (DVC) to study the microstructural behaviour of trabecular bone during compression. J. Mech. Behav. Biomed. Mater. 29, 480–499 (2014)
M. Palanca, L. Cristofolini, E. DallAra, M. Curto, F. Innocente, V. Danesi, G. Tozzi, Digital volume correlation can be used to estimate local strains in natural and augmented vertebrae: an organ-level study. J. Biomech. 49, 3882–3890 (2016)
G. Tozzi, V. Danesi, M. Palanca, L. Cristofolini, Elastic full-field strain analysis and microdamage progression in the vertebral body from digital volume correlation. Strain 52, 446–455 (2016)
E. DallAra, M. Peña-Fernández, M. Palanca, M. Giorgi, L. Cristofolini, G. Tozzi, Precision of digital volume correlation approaches for strain analysis in bone imaged with micro-computed tomography at different dimensional levels. Front. Mater. (2017). https://doi.org/10.3389/fmats.2017.00031
S. Rapagna, S. Berahmani, C.E. Wyers, J.P.W. van den Bergh, K.J. Reynolds, G. Tozzi, D. Janssen, E. Perilli, Quantification of human bone microarchitecture damage in press-fit femoral knee implantation using HR-pQCT and digital volume correlation. J. Mech. Behav. Biomed. Mater. 97, 278–287 (2019)
R. Zdero, Experimental Methods in Orthopaedic Biomechanics (Academic Press, London, 2017)
R. Huiskes, H. Weinans, H.J. Grootenboer, M. Dalstra, B. Fudala, T.J. Sloof, Adaptive bone remodeling theory applied to prosthetic design analysis. J. Biomech. 20, 1135–1150 (1987)
R.T. Hart, D.T. Davy, Theories of bone modelling and remodelling, in Bone Mechanics, ed. by S.C. Cowin (CRC Press, Boca Reton, 1989), pp. 253–277
J.M. García, M. Doblaré, J. Cegoñino, Bone remodelling simulation: a tool for implant design. Comput. Mater. Sci. 25, 100–114 (2002)
D. Carter, T.E. Orr, D. Fyhrie, Relationships between loading history and femoral cancellous bone architecture. J. Biomech. 22(3), 231–244 (1989)
H. Weinans, R. Huiskes, B. van Rietbergen, D.R. Sumner, T.M. Turner, J.O. Galante, Adaptive bone remodelling around bonded noncemented total hip arthroplasty: a comparison between animal experiments and computer simulation. J. Orthop. Res. 11, 500–513 (1993)
L.M. McNamara, P.J. Prendergast, Bone remodelling algorithms incorporating both strain and microdamage stimuli. J. Biomech. 40, 1381–1391 (2007)
K. Mukherjee, S. Gupta, The effects of musculoskeletal loading regimes on numerical evaluations of acetabular component. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 230(10), 918–929 (2016)
D.R. Suarez, H. Weinans, F. van Keulen, Bone remodelling around a cementless glenoid component. Biomech. Model Mechnobiol. 11(6), 903–913 (2012)
S. Rothstock, A. Uhlenbrock, N. Bishop, L. Laird, R. Nassutt, M. Morlock, Influence of interface condition and implant design on bone remodelling and failure risk for the resurfaced femoral head. J. Biomech. 44(9), 1646–1653 (2011)
I.T. Haider, A.D. Speirs, P.E. Beaulé, H. Frei, Influence of ingrowth regions on bone remodelling around a cementless hip resurfacing femoral implant. Comput. Methods Biomech. Biomed. Eng. 18(12), 1349–1357 (2015)
B. Pal, S. Gupta, The effect of primary stability on load transfer and bone remodelling within the uncemented resurfaced femur. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 225, 549–561 (2011)
R. Ghosh, K. Mukherjee, S. Gupta, Bone remodelling around uncemented metallic and ceramic acetabular components. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 227(5), 490–502 (2013)
M. Viceconti, L. Monti, R. Muccini, M. Bernakiewicz, A. Toni, Even a thin layer of soft tissue may compromise the primary stability of cementless hip stems. Clin. Biomech. 16(9), 765–775 (2001)
R.B. Ruben, J. Folgado, P.R. Fernandes, On the optimal shape of hip implants. J. Biomech. 45(2), 239–246 (2012)
S.A. Khanoki, D. Pasini, Multiscale design and multiobjective optimization of orthopedic hip implants with functionally graded cellular material. J. Biomech. Eng. (2012). https://doi.org/10.1115/1.4006115
S. Chanda, S. Gupta, D.K. Pratihar, A genetic algorithm based multi-objective shape optimization scheme for cementless femoral implant. J. Biomech. Eng. 137, 034502 (2015)
H. Katoozian, D.T. Davy, Effects of loading conditions and objective function on three-dimensional shape optimization of femoral components of hip endoprostheses. Med. Eng. Phys. 22(4), 243–251 (2000)
R.B. Ruben, J. Folgado, P.R. Fernandes, Three-dimensional shape optimization of a hip prosthesis using a multicriteria formulation. Struct. Multidisc. Optim. 34(4), 261–275 (2007)
J.E. Davies, Understanding peri-implant endosseous healing. J. Dent. Edu. 67(8), 932–949 (2003)
F.P. Barry, Biology and clinical applications of mesenchymal stem cells. Birth Defects Res. (Part C) 69, 250–256 (2003)
D.R. Carter, P.R. Blenman, G.S. Beaupré, Correlations between mechanical stress history and tissue differentiation in initial fracture healing. J. Orthop. Res. 6, 736–748 (1988)
A.D. Speirs, M.A. Slomczykowski, T.E. Orr, K. Siebenrock, L.P. Nolte, Three-dimensional measurement of cemented femoral stem stability: an in vitro cadaver study. Clin. Biomech. 15, 248–255 (2000)
L.E. Claes, C.A. Heigele, Magnitudes of local stress and strain along bony surfaces predict the course and type of fracture healing. J. Biomech. 32(3), 255–266 (1999)
P.J. Prendergast, R. Huiskes, K. Søballe, Biophysical stimuli on cells during tissue differentiation at implant interfaces. J. Biomech. 30(6), 539–548 (1997)
P.J. Prendergast, R. Huiskes, Finite element analysis of fibrous tissue morphogenesis: a study of the osteogenic index with a biphasic approach. Mech. Compos. Mater. 32, 144–150 (1996)
T.N. Gardner, S. Mishra, L. Marks, The role of osteogenic index, octahedral shear stress and dilatational stress in the ossification of a fracture callus. Med. Eng. Phys. 26, 493–501 (2004)
D. Lacroix, P.J. Prendergast, A mechano-regulation model for tissue differentiation during fracture healing: analysis of gap size and loading. J. Biomech. 35(9), 1163–1171 (2002)
D. Lacroix, P.J. Prendergast, G. Li, D. Marsh, Biomechanical model to simulate tissue differentiation and bone regeneration: application to fracture healing. Med. Biol. Eng. Comput. 40(1), 14–21 (2002)
H. Isaksson, W. Wilson, C.C. van Donkelaar, R. Huiskes, K. Ito, Comparison of biophysical stimuli for mechanoregulation of tissue differentiation during fracture healing. J. Biomech. 39(8), 1507–1516 (2006)
D.J. Kelly, P.J. Prendergast, Mechano-regulation of stem cell differentiation and tissue regeneration in osteochondral defects. J. Biomech. 38, 1413–1422 (2005)
H.Y. Chou, S. Müftü, Simulation of peri-implant bone healing due to immediate loading in dental implant treatments. J. Biomech. 46(14), 871–878 (2013)
A. Dickinson, A. Taylor, M. Browne, Implant–bone interface healing and adaptation in resurfacing hip replacement. Comput. Methods Biomech. Biomed. Eng. 15(9), 935–947 (2012)
K. Mukherjee, S. Gupta, Bone ingrowth around porous coated acetabular implant: a three-dimensional finite element study using mechanoregulatory algorithm. Biomech. Model. Mechanobiol. 15(2), 389–403 (2016)
J.A. Hanzlik, J.S. Day, Bone ingrowth in well-fixed retrieved porous tantalum implants. J. Arthrop. 28, 922–927 (2013)
K. Mukherjee, S. Gupta, Mechanobiological simulations of peri-acetabular bone ingrowth: a comparative analysis of cell-phenotype specific and phenomenological algorithms. Med. Biol. Eng. Comput. 55(3), 449–465 (2017)
K. Mukherjee, S. Gupta, Influence of implant surface texture design on peri-acetabular bone ingrowth: a mechanobiology based finite element analysis. J. Biomech. Eng. 139(3), 031006 (2017)
V.V. Ruiwale, R.U. Sambhe, A review on design process of orthopedic implants. IOSR J. Mech. Civ. Eng. 12(6), 76–82 (2015)
M. Kutz, Standard Handbook of Biomedical Engineering and Design (McGraw-Hill, New York, 2003)
K. Alexander, P.J. Clarkson, Good design practice for medical devices and equipment, part I: a review of current literature. J. Med. Eng. Technol. 24, 5–13 (2000)
BS EN ISO 21535:2009 + A1:2016, Non-active surgical implants. Joint replacement implants. Specific requirements for hip-joint replacement implants (British Standards Institute, London, 2009)
BS EN ISO 21536:2009 + A1:2014, Non-active surgical implants. Joint replacement implants. Specific requirements for knee-joint replacement implants (British Standards Institute, London, 2009)
K. Alexander, P.J. Clarkson, Good design practice for medical devices and equipment, part II: design for validation. J. Med. Eng. Technol. 24, 53–62 (2000)
BS EN ISO 14971:2019, Medical devices: application of risk management to medical devices (British Standards Institute, London, 2019)
BS EN IEC 60812:2018, Analysis techniques for system reliability. Procedure for failure mode and effects analysis (FMEA) (British Standards Institute, London, 2018)
BS EN 61025: 2007 Fault tree analysis (FTA) (British Standards Institute, London, 2007)
J.R. Meakin, D.E.T. Shepherd, D.W.L. Hukins, Fused deposition models from CT scans. Br. J. Radiol. 77, 504–507 (2004)
E. Murr, S.M. Gaytan, E. Martinez, F. Medina, R.B. Wicker, Next generation orthopaedic implants by additive manufacturing using electron beam melting. Int. J. Biomater. (2012). https://doi.org/10.1155/2012/245727
Q. Yan, H. Dong, J. Su, J. Han, B. Song, Q. Wei, Y. Shi, A review of 3d printing technology for medical applications. Engineering 4(5), 729–742 (2018)
BS 7251-12:1995, ISO 7206-8:1995, Orthopedic joint prostheses. Specification for endurance of stemmed femoral components with application of torsion (British Standards Institute, London, 1995)
R. Mundi, H. Chaudhry, S. Mundi, K. Godin, M. Bhandari, Design and execution of clinical trials in orthopaedic surgery. Bone Joint Res. 3(5), 161–168 (2014)
N. Kelly, D.T. Cawley, F.J. Shannon, J.P. McGarry, An investigation of the inelastic behaviour of trabecular bone during the press-fit implantation of a tibial component in total knee arthroplasty. Med. Eng. Phys. 35(11), 1599–1606 (2013)
J.H. Marangalou, K. Ito, M. Cataldi, F. Taddei, B. van Rietbergen, A novel approach to estimate trabecular bone anisotropy using a database approach. J. Biomech. 46(14), 2356–2362 (2013)
D. Janssen, R.E. Zwartelé, H.C. Doets, N. Verdonschot, Computational assessment of press-fit acetabular implant fixation: the effect of implant design, interference fit, bone quality, and frictional properties. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 224, 67–75 (2010)
E. Verhulp, B. van Rietbergen, R. Huiskes, Comparison of micro-level and continuum-level voxel models of the proximal femur. J. Biomech. 39(16), 2951–2957 (2006)
P.M. Cattaneo, M. Dalstra, L.H. Frich, A three-dimensional finite element model from computed tomography data: a semi-automated method. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 215(2), 203–213 (2001)
D.M. Geraldes, A. Phillips, A comparative study of orthotropic and isotropic bone adaptation in the femur. Int. J. Numer. Meth. Biomed. Eng. 30(9), 873–889 (2014)
M. Taylor, D.S. Barrett, D. Deffenbaugh, Influence of loading and activity on the primary stability of cementless tibial trays. J. Orthop. Res. 30(9), 1362–1368 (2012)
F. Taddei, S. Martelli, H.S. Gill, L. Cristofolini, M. Viceconti, Finite element modeling of resurfacing hip prosthesis: estimation of accuracy through experimental validation. J. Biomech. Eng. 132(2), 021002 (2010)
S. Gupta, B. Pal, A.M.R. New, The effects of interfacial conditions and stem length on potential failure mechanisms in the uncemented resurfaced femur. Ann. Biomed. Eng. 38(6), 2107–2120 (2010)
J. Shi, M. Browne, M. Strickland, G. Flivik, M. Taylor, Sensitivity analysis of a cemented hip stem to implant position and cement mantle thickness. Comput. Methods Biomech. Biomed. Eng. 17(15), 1671–1684 (2014)
A. Roques, M. Browne, A. Taylor, A. New, D. Baker, Quantitative measurement of the stresses induced during polymerisation of bone cement. Biomaterials 25(18), 4415–4424 (2004)
A. Ramos, M.W. Schiller, I. Abe, P.A. Lopes, J.A. Simões, Experimental measurement and numerical validation of bone cement mantle strains of an in vitro hip replacement using optical FBG sensors. Exp. Mech. 52(9), 1267–1274 (2012)
A.B. Lennon, P.J. Prendergast, Residual stress due to curing can initiate damage in porous bone cement: experimental and theoretical evidence. J. Biomech. 35(3), 311–321 (2002)
C. Li, Y. Wang, J. Mason, The effects of curing history on residual stresses in bone cement during hip arthroplasty. J. Biomed. Mater. Res. B Appl. Biomater. 70(1), 30–36 (2004)
J.R.T. Jeffers, M. Browne, M. Taylor, Damage accumulation, fatigue and creep behaviour of vacuum mixed bone cement. Biomaterials 26(27), 5532–5541 (2005)
J.R.T. Jeffers, M. Browne, A.B. Lennon, P.J. Prendergast, M. Taylor, Cement mantle fatigue failure in total hip replacement: experimental and computational testing. J. Biomech. 40(7), 1525–1533 (2007)
M. Pérez, J. Palacios, Comparative finite element analysis of the debonding process in different concepts of cemented hip implants. Ann. Biomed. Eng. 38(6), 2093–2106 (2010)
D. Waanders, D. Janssen, K.A. Mann, N. Verdonschot, The behaviour of the micro-mechanical cement–bone interface affects the cement failure in total hip replacement. J. Biomech. 44(2), 228–234 (2011)
Q.H. Zhang, G. Tozzi, J. Tong, Micro-mechanical damage of trabecular bone–cement interface under selected loading conditions: a finite element study. Comput. Methods Biomech. Biomed. Eng. (2012). https://doi.org/10.1080/10255842.2012.675057
Acknowledgements
The authors wish to thank the current and former research scholars of the Biomechanics Laboratory, Department of Mechanical Engineering, IIT Kharagpur, for their scientific contributions in this research area.
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pal, B., Gupta, S. The Relevance of Biomechanical Analysis in Joint Replacements: A Review. J. Inst. Eng. India Ser. C 101, 913–927 (2020). https://doi.org/10.1007/s40032-020-00611-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40032-020-00611-5