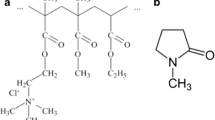
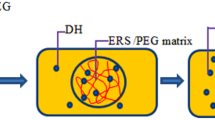
Abstract
Antibiotic-loaded in situ forming gels are particularly attractive for periodontitis treatment. They are in a sol form and gradually alter to a solid-like depot after administration into a periodontal pocket to deliver and maintain the effective high level of drug in the gingival crevicular fluid. Solvent-inducing in situ forming gel mostly exhibits the burst drug release owing to rapid diffusion of solvent. This study incorporated peppermint oil to modulate the drug release and the gel characteristics of doxycycline hyclate-loaded Eudragit RS in situ forming gel systems. Peppermint oil increased the viscosity and syringeability of the Eudragit RS solution comprising NMP as solvent and retarded the water penetration. Therefore the diminishment of burst liberation and the prolongation of drug release with an addition of peppermint oil were attained with concentration dependence mainly following Fickian diffusion mechanism. The drug release from the membrane-less diffusion method was apparently slower than that from the dialysis method because the rapid phase separation into solid-like matrix through a direct contact with dissolution medium generated a hard surrounding shell. These solvent exchange-inducing in situ forming gels comprising peppermint oil effectively inhibited Staphylococcus aureus, Escherichia coli, Streptococcus mutans and Porphyrommonas gingivalis; therefore, they exhibited the potential use as localized delivery systems for periodontitis treatment.
Graphical Abstract






Similar content being viewed by others
References
Agarwal RK, Robinson DH, Maze GI, Reinhardt RA (1993) Development and characterization of tetracycline-poly(lactide/glycolide) films for the treatment of periodontitis. J Control Release 23:137–146
Ahmed T (2015) Review: approaches to develop PLGA based in situ gelling system with low initial burst. Pak J Pharm Sci 28:657–665
Ahmed TA, Ibrahim HM, Samy AM, Mohammad AK, Nutan TH, Hussain MD (2014) Biodegradable injectable in situ implants and microparticles for sustained release of montelukast: in vitro release, pharmacokinetics, and stability. AAPS PharmSciTech 15:772–780. doi:10.1208/s12249-014-0101-3
Akhgari A, Farahmand F, Afrasiabi GH, Sadeghi F, Vandamme TF (2006) Permeability and swelling studies on free films containing inulin in combination with different polymethacrylates aimed for colonic drug delivery. Eur J Pharm Sci 28:307–314
Barrera-Necha LL, Garduno-Pizana C, Garcia-Barrera LJ (2009) In vitro antifungal activity of essential oils and their compounds on mycelial growth of Fusarium oxysporium f.sp. gladioli (Massey) Synder and Hansen. Plant Pathol J 8:17–21
Baumgartner S, Kristl J, Vrečer F, Vodopivec P, Zorko B (2000) Optimisation of floating matrix tablets and evaluation of their gastric residence time. Int J Pharm 195:125–135
Brodbeck KJ, DesNoyer JR, McHugh AJ (1999) Phase inversion dynamics of PLGA solutions related to drug delivery: Part II. The role of solution thermodynamics and bath-side mass transfer. J Control Release 62:333–344
Camargo JA, Sapin A, Nouvel C, Daloz D, Leonard M, Bonneaux F, Six JL, Maincent P (2013) Injectable PLA-based in situ forming implants for controlled release of ivermectin a BCS Class II drug: solvent selection based on physico-chemical characterization. Drug Dev Ind Pharm 39:146–155
Denyer SP, Hugo WB (1991) Biocide-induced damage to the bacterial cytoplasmic membranes. In: Mechanisms of action of chemical biocides. Blackwell Scientific Publications, Oxford
Do MP, Neut C, Delcourt E, Certo TS, Siepmann J, Siepmann F (2014) In-situ forming implants for periodontitis treatment with improved adhesive properties. Eur J Pharm Biopharm 88:342–350
Do MP, Neut C, Metz H, Delcourt E, Mäder K, Siepmann J, Siepmann F (2015) In-situ forming composite implants for periodontitis treatment: how the formulation determines system performance. Int J Pharm 486:38–51. doi:10.1016/j.ijpharm.2015.03.026
Duss CL, Lang NP, Cosyn J, Persson GR (2010) A randomized, controlled clinical trial on the clinical, microbiological, and staining effects of a novel 0.05% chlorhexidine/herbal extract and a 0.1% chlorhexidine mouthrinse adjunct to periodontal surgery. J Clin Periodontol 37:988–997. doi:10.1111/j.1600-051X.2010.01609.x
Engelhardt G, Fleig H (1993) Methyl-2-pyrrolidinone (NMP) does not induce structural and numerical chromosomal aberrations in vivo. Mutat Res Genet Toxicol 298:149–155
Esposito E, Sebben S, Cortesi R, Menegatti E, Nastruzzi C (1999) Preparation and characterization of cationic microspheres for gene delivery. Int J Pharm 189:29–41
Godavarthy SS, Yerramasetty KM, Neely BJ, Madihally SV, Robinson RL, Gasem KA (2009) Design of improved permeation enhancers for transdermal drug delivery. J Pharm Sci 98:4085–4099
Graham PD, Brodbeck KJ, McHugh AJ (1999) Phase inversion dynamics of PLGA solutions related to drug delivery. J Control Release 58:233–245
Heidari MR (2014) Reference module in biomedical sciences, Encyclopedia of Toxicology, 3rd edn. Elsevier, Amsterdam, pp 588–593
Huang SJ, Ho LH, Hong E, Kitchen O (1994) Hydrophilic-hydrophobic biodegradable polymers: release characteristics of hydrogen-bonded, ring-containing polymer matrices. Biomaterials 15:1243–1247
Ikeda Y, Kimura K, Hirayama F, Arima H, Uekama K (2000) Controlled release of a water-soluble drug, captopril, by a combination of hydrophilic and hydrophobic cyclodextrin derivatives. J Control Release 66:271–280
Jain RA, Rhodes CT, Railkar AM, Malick W, Shah NH (2000) Controlled release of drugs from injectable in situ formed biodegradable PLGA microspheres: effect of various formulation variables. Eur J Pharm Biopharm 50:257–262. doi:10.1016/S0939-6411(00)00062-X
Jain N, Jain GK, Javed S, Iqbal Z, Talegaonkar S, Ahmad FJ, Khar RK (2008) Recent approaches for the treatment of periodontitis. Drug Discov Today 13:932–943
Jeyakumar E, Lawrence R, Pal T (2011) Comparative evaluation in the efficacy of peppermint (Mentha piperita) oil with standards antibiotics against selected bacterial pathogens. Asian Pac J Trop Biomed 1:S253–S257
Khuathan N, Pongjanyakul T (2014) Modification of quaternary polymethacrylate films using sodium alginate: film characterization and drug permeability. Int J Pharm 460:63–72
Kogawa AC, Salgado HR (2012) Doxycycline hyclate: a review of properties, applications and analytical methods. Int J Life Sci Pharm Res 2:11–25
Lakhdar L, Hmamouchi M, Rida S, Ennibi O (2012) Antibacterial activity of essential oils against periodontal pathogens: a qualitative systematic review. Odontostomatol Trop 35:38–46
Lin WJ, Lee HK, Wamg DM (2004) The influence of plasticizers on the release of theophylline from microporous-controlled tablets. J Control Release 99:415–421
Liu H, Venkatraman SS (2012) Cosolvent effects on the drug release and depot swelling in injectable in situ depot-forming systems. J Pharm Sci 101:1783–1793
Liu Q, Zhang H, Zhou G, Xie S, Zou H, Yu Y, Li G, Sun D, Zhang G, Lu Y, Zhong Y (2010) In vitro and in vivo study of thymosin alpha1 biodegradable in situ forming poly(lactide-co-glycolide) implants. Int J Pharm 397:122–129
Lopedota A, Trapani A, Cutrignelli A, Chiarantini L, Pantucci E, Curci R, Manuali E, Trapani G (2009) The use of Eudragit® RS 100/cyclodextrin nanoparticles for the transmucosal administration of glutathione. Eur J Pharm Biopharm 72:509–520
Martin A (1993) Physical pharmacy. Lea and Febiger, Philadelphia, pp 393–476
MicroMath Scientist (1995) Handbook Rev. 7EEF. MicroMath, Salt Lake City
Mohsenzadeh M (2007) Evaluation of antibacterial activity of selected Iranian essential oils against Staphylococcus aureus and Escherichia coli in nutrient broth medium. Pak J Biol Sci 10:3693–3697
Nagarwal RC, Srinatha A, Pandit JK (2009) In situ forming formulation: Development, evaluation, and optimization using 33 factorial design. AAPS PharmSciTech 10:977–784
Phaechamud T, Mahadlek J (2015) Solvent exchange-induced in situ forming gel comprising ethyl cellulose-antimicrobial drugs. Int J Pharm 494:381–392. doi:10.1016/j.msec.2015.09.083
Phaechamud T, Mahadlek J, Charoenteeraboon J, Choopun S (2013a) Analysis for texture and topography of doxycycline hyclate thermosensitive systems comprising zinc oxide. Indian. J Pharm Sci 75:385–392
Phaechamud T, Mahadlek J, Charoenteeraboon J, Choopun S (2013b) Characterization and antimicrobial activity of N-methyl-2-pyrrolidone-loaded ethylene oxide-propylene oxide block copolymer thermosensitive gel. Indian J Pharm Sci 74:498–504
Phaechamud T, Jantadee T, Mahadlek J, Charoensuksai P, Pichayakorn W (2016a) Characterization of antimicrobial agent loaded eudragit RS solvent exchange-induced in situ forming gels for periodontitis treatment. AAPS PharmSciTech 18(2):494–508. doi:10.1208/s12249-016-0534-y
Phaechamud T, Mahadlek J, Chuenbarn T (2016b) In situ forming gel comprising bleached shellac loaded with antimicrobial drugs for periodontitis treatment. Mater Des 494:381–392. doi:10.1016/j.matdes.2015.09.138
Rachakonda VK, Terramsetty KM, Madihally SV, Robinson RL, Gasem KA (2008) Screening of chemical penetration enhancers for transdermal drug delivery using electrical resistance of skin. Pharm Res 25:2697–2704
Rongthong T, Sungthongjeen S, Siepmann F, Siepmann J, Pongjanyakul T (2015) Quaternary polymethacrylate–magnesium aluminum silicate films: water uptake kinetics and film permeability. Int J Pharm 490:165–172
Rowe RC, Sheskey PJ, Quinn EM (2009) Handbook of pharmaceutical excipients, 6th edn. Pharmaceutical Press and American Pharmaceutical Association, Washington, DC
Sánchez-Lafuente C, Faucci MT, Fernández-Arévalo M, Álvarez-Fuentes J, Rabasco AM, Mura P (2002) Development of sustained release matrix tablets of didanosine containing methacrylic and ethylcellulose polymers. Int J Pharm 234:213–221
Sanghvi R, Narazaki R, Machatha SG, Yalkowsky SH (2008) Solubility improvement of drugs using N-methyl pyrrolidone. AAPS PharmSciTech 9:366–376
Sartoratto A, Machado MA, Delarmelina C, Figueira GM, Duarte MC, Rehder VL (2004) Composition and antimicrobial activity of essential oils from aromatic plants used in Brazil. Braz J Microbiol 35:275–280
Schelz Z, Molnar J, Hohmann J (2006) Antimicrobial and antiplasmid activities of essential oils. Fitoterapia 77:279–285
Schuhmacher A, Reichling J, Schnitzler P (2003) Virucidal effect of peppermint oil on the enveloped viruses herpes simplex virus type 1 and type 2 in vitro. Phytomedicine 10:504–510
Schwach AK, Vivien CN, Gurny R (2000) Local delivery of antimicrobial agents for the treatment of periodontal diseases. Eur J Pharm Biopharm 50:83–99
Semdé R, Amighi K, Pierre D, Devleeschouwer MJ, Moës AJ (1998) Leaching of pectin from mixed pectin/insoluble polymer films intended for colonic delivery. Int J Pharm 174:233–241
Shapiro S, Meier A, Guggenheim B (1994) The antimicrobial activity of essential oils and essential oil components towards oral bacteria. Oral Microbiol Immunol 9:202–208
Siepmann J, Peppas NA (2001) Modelling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Adv Drug Deliv Rev 48:139–157
Sikkema J, de Bont JA, Poolman B (1994) Interaction of cyclic hydrocarbons with biological membranes. J Biol Chem 269:8022–8028
Solorio L, Olear AM, Hamilton JI, Patel RB, Beiswenger AC, Wallace JE, Zho H, Exner AA (2012) Noninvasive characterization of the effect of varying PLGA molecular weight blends on in situ forming implant behavior using ultrasound imaging. Theranostics 2:1064–1077. doi:10.7150/thno.4181
Thakur RR, McMillan HL, Jones DS (2014) Solvent induced phase inversion-based in situ forming controlled release drug delivery implants. J Control Release 176:8–23. doi:10.1016/j.jconrel.2013.12.020
Thosar N, Basak S, Bahadure RN, Rajurkar M (2013) Antimicrobial efficacy of five essential oils against oral pathogens: an in vitro study. Eur J Dent 7:S71–S77. doi:10.4103/1305-7456.119078
Wang L, Venkatraman S, Kleiner L (2004) Drug release from injectable depots: two different in vitro mechanisms. J Control Release 99:207–216
Xiong W, Gao X, Zhao Y, Xu H, Yang X (2011) The dual temperature/pH-sensitive multiphase behavior of poly(N-isopropylacrylamide-co-acrylic acid) microgels for potential application in in situ gelling system. Colloids Surf B 84:103–110
Yamamoto S, Saeki T, Inoshita T (2002) Drying of gelled sugar solutions-water diffusion behavior. Chem Eng J 86:179–184
You C, Liang X, Sun J, Sun L, Wang Y, Fan T, Zheng Y (2014) Blends of hydrophobic and swelling agents in the swelling layer in the preparation of delayed-release pellets of a hydrophilic drug with low MW: physicochemical characterizations and in-vivo evaluations. Asian J Pharm Sci 9:199–207
Acknowledgements
This research work was grateful for the Research and Development Institute, Silpakorn University (Grant No. SURDI 57/01/42). This research work was also facilitated by the Faculty of Pharmacy, Silpakorn University, Thailand. We also thank Dr. Kamonpan Boonkit for valuable comments and help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest in this work.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Phaechamud, T., Mahadlek, J. & Tuntarawongsa, S. Peppermint oil/doxycycline hyclate-loaded Eudragit RS in situ forming gel for periodontitis treatment. J. Pharm. Investig. 48, 451–464 (2018). https://doi.org/10.1007/s40005-017-0340-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-017-0340-x