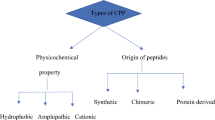
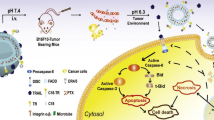
Abstract
With the rapid development of biotechnology, various macromolecules as therapeutic agents have made drug delivery an important field of research. However, these are being commercialized as injection form. Due to low patient compliance, various non-invasive routes emerge as a promising strategy. Cell penetrating peptides (CPPs) have shown to assist in efficient and non-toxic manner. They provide ample evidence to deliver of many cargoes ranging from small molecules to proteins and even nanocarriers for various applications. This review briefly discusses about introduction of CPPs, history, cellular uptake mechanisms and various possible alternative routes for CPP-conjugated drug delivery system. It also aims to give a perspective on present status of CPP-mediated research, clinical development, possible obstacles as well as future opportunities. Thus, development of novel CPPs that are safe, tissue-specific and highly efficient will be exemplified and become ideal vehicles for therapeutic delivery in near future.





Similar content being viewed by others
References
Abes R, Moulton HM, Clair P, Yang ST, Abes S, Melikov K, Prevot P, Youngblood DS, Iversen PL, Chernomordik LV (2008) Delivery of steric block morpholino oligomers by (RXR) 4 peptides: structure–activity studies. Nucleic Acids Res 36:6343–6354
Bae H, Lee K (2013) On employing a translationally controlled tumor protein-derived protein transduction domain analog for transmucosal delivery of drugs. J Controlled Release 170:358–364
Baoum A, Ovcharenko D, Berkland C (2012) Calcium condensed cell penetrating peptide complexes offer highly efficient, low toxicity gene silencing. Int J Pharm 427:134–142
Bashyal S, Lee S (2015) Delivery of biopharmaceuticals using combination of liposome and iontophoresis: a review. J Pharm Investig 45:611–624
Bleifuss E, Kammertoens T, Hutloff A, Quarcoo D, Dorner M, Straub P, Uckert W, Hildt E (2006) The translocation motif of hepatitis B virus improves protein vaccination. Cell Mol Life Sci 63:627–635
Cohen-Avrahami M, Aserin A, Garti N (2010) H II mesophase and peptide cell-penetrating enhancers for improved transdermal delivery of sodium diclofenac. Colloids Surf, B 77:131–138
Cohen-Avrahami M, Libster D, Aserin A, Garti N (2012) Penetratin-induced transdermal delivery from H II mesophases of sodium diclofenac. J Controlled Release 159:419–428
Derossi D, Joliot AH, Chassaing G, Prochiantz A (1994) The third helix of the Antennapedia homeodomain translocates through biological membranes. J Biol Chem 269:10444–10450
Derossi D, Calvet S, Trembleau A, Brunissen A, Chassaing G, Prochiantz A (1996) Cell internalization of the third helix of the Antennapedia homeodomain is receptor-independent. J Biol Chem 271:18188–18193
Deshayes S, Plenat T, Aldrian-Herrada G, Divita G, Le Grimellec C, Heitz F (2004) Primary amphipathic cell-penetrating peptides: structural requirements and interactions with model membranes. Biochemistry 43:7698–7706
Deshayes S, Morris M, Divita G, Heitz F (2005) Cell-penetrating peptides: tools for intracellular delivery of therapeutics. Cell Mol Life Sci CMLS 62:1839–1849
Duan X, Mao S (2010) New strategies to improve the intranasal absorption of insulin. Drug Discov Today 15:416–427
Duchardt F, Fotin-Mleczek M, Schwarz H, Fischer R, Brock R (2007) A comprehensive model for the cellular uptake of cationic cell-penetrating peptides. Traffic 8:848–866
El-Andaloussi S, Holm T, Langel U (2005) Cell-penetrating peptides: mechanisms and applications. Curr Pharm Des 11:3597–3611
El-Andaloussi S, Johansson HJ, Holm T, Langel U (2007) A novel cell-penetrating peptide, M918, for efficient delivery of proteins and peptide nucleic acids. Mol Ther 15:1820–1826
Elliott GO, Hare P (1997) Intercellular trafficking and protein delivery by a herpesvirus structural protein. Cell 88:223–233
Elmquist A, Lindgren M, Bartfai T, Langel U (2001) VE-cadherin-derived cell-penetrating peptide, pVEC, with carrier functions. Exp Cell Res 269:237–244
El-Sayed A, Futaki S, Harashima H (2009) Delivery of macromolecules using arginine-rich cell-penetrating peptides: ways to overcome endosomal entrapment. AAPS J 11:13–22
Eum WS, Jang SH, Kim DW, Choi HS, Choi SH, Kim SY, An JJ, Lee SH, Han K, Kang JH (2005) Enhanced transduction of Cu, Zn-superoxide dismutase with HIV-1 Tat protein transduction domains at both termini. Mol Cells 19:191–197
Foerg C, Merkle HP (2008) On the biomedical promise of cell penetrating peptides: limits versus prospects. J Pharm Sci 97:144–162
Frankel AD, Pabo CO (1988) Cellular uptake of the tat protein from human immunodeficiency virus. Cell 55:1189–1193
Futaki S, Suzuki T, Ohashi W, Yagami T, Tanaka S, Ueda K, Sugiura Y (2001) Arginine-rich peptides An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery. J Biol Chem 276:5836–5840
Gazit E, Lee WJ, Brey PT, Shai Y (1994) Mode of action of the antibacterial cecropin B2: a spectrofluorometric study. Biochemistry 33:10681–10692
Goldberg M, Gomez-Orellana I (2003) Challenges for the oral delivery of macromolecules. Nat Rev Drug Discovery 2:289–295
Green M, Loewenstein PM (1988) Autonomous functional domains of chemically synthesized human immunodeficiency virus tat trans-activator protein. Cell 55:1179–1188
Hoogstraate JA, Wertz PW (1998) Drug delivery via the buccal mucosa. Pharm Sci Technol Today 1:309–316
Hou YW, Chan MH, Hsu HR, Liu BR, Chen CP, Chen HH, Lee HJ (2007) Transdermal delivery of proteins mediated by non-covalently associated arginine-rich intracellular delivery peptides. Exp Dermatol 16:999–1006
Jarver P, Langel U (2006) Cell-penetrating peptides-a brief introduction. Biochim Biophys Acta (BBA)-Biomembranes 1758:260–263
Jeong EJ, Choi M, Lee J, Rhim T, Lee KY (2015) The spacer arm length in cell-penetrating peptides influences chitosan/siRNA nanoparticle delivery for pulmonary inflammation treatment. Nanoscale 7:20095–20104
Jin LH, Bahn JH, Eum WS, Kwon HY, Jang SH, Han KH, Kang TC, Won MH, Kang JH, Cho SW (2001) Transduction of human catalase mediated by an HIV-1 TAT protein basic domain and arginine-rich peptides into mammalian cells. Free Radic Biol Med 31:1509–1519
Johansson HJ, El-Andaloussi S, Holm T, Mae M, Janes J, Maimets T, Langel U (2008) Characterization of a novel cytotoxic cell & hyphen; penetrating peptide derived from p14ARF protein. Mol Ther 16:115–123
Joliot A, Pernelle C, Deagostini-Bazin H, Prochiantz A (1991) Antennapedia homeobox peptide regulates neural morphogenesis. Proc Natl Acad Sci 88:1864–1868
Jones AT, Sayers EJ (2012) Cell entry of cell penetrating peptides: tales of tails wagging dogs. J Controlled Release 161:582–591
Juliano R, Alam MR, Dixit V, Kang H (2008) Mechanisms and strategies for effective delivery of antisense and siRNA oligonucleotides. Nucleic Acids Res 36:4158–4171
Kale AA, Torchilin VP (2007a) Enhanced transfection of tumor cells in vivo using “Smart” pH-sensitive TAT-modified pegylated liposomes. J Drug Target 15:538–545
Kale AA, Torchilin VP (2007b) “Smart” drug carriers: PEGylated TATp-modified pH-sensitive liposomes. J Liposome Res 17:197–203
Kamei N, Takeda-Morishita M (2015) Brain delivery of insulin boosted by intranasal coadministration with cell-penetrating peptides. J Controlled Release 197:105–110
Kamei N, Morishita M, Eda Y, Ida N, Nishio R, Takayama K (2008) Usefulness of cell-penetrating peptides to improve intestinal insulin absorption. J Controlled Release 132:21–25
Kamei N, Nielsen EJB, Khafagy ES, Takeda-Morishita M (2013) Noninvasive insulin delivery: the great potential of cell-penetrating peptides. Ther Deliv 4:315–326
Kang MH, Park MJ, Yoo HJ, Lee SG, Kim SR, Yeom DW, Kang MJ, Choi YW (2014) RIPL peptide (IPLVVPLRRRRRRRRC)-conjugated liposomes for enhanced intracellular drug delivery to hepsin-expressing cancer cells. Eur J Pharm Biopharm 87:489–499
Kang MH, Yoo HJ, Kwon YH, Yoon HY, Lee SG, Kim SR, Yeom DW, Kang MJ, Choi YW (2015) Design of multifunctional liposomal nanocarriers for folate receptor-specific intracellular drug delivery. Mol Pharm 12:4200–4213
Kersemans V, Kersemans K, Cornelissen B (2008) Cell penetrating peptides for in vivo molecular imaging applications. Curr Pharm Des 14:2415–2427
Khafagy ES, Morishita M, Onuki Y, Takayama K (2007) Current challenges in non-invasive insulin delivery systems: a comparative review. Adv Drug Deliv Rev 59:1521–1546
Khafagy ES, Morishita M, Isowa K, Imai J, Takayama K (2009) Effect of cell-penetrating peptides on the nasal absorption of insulin. J Controlled Release 133:103–108
Khafagy ES, Morishita M, Ida N, Nishio R, Isowa K, Takayama K (2010) Structural requirements of penetratin absorption enhancement efficiency for insulin delivery. J Controlled Release 143:302–310
Khafagy ES, Kamei N, Nielsen EJB, Nishio R, Takeda-Morishita M (2013) One-month subchronic toxicity study of cell-penetrating peptides for insulin nasal delivery in rats. Eur J Pharm Biopharm 85:736–743
Kim DW, Eum WS, Jang SH, Yoon CS, Choi HS, Choi SH, Kim YH, Kim SY, Lee ES, Nl Baek (2003) Ginsenosides enhance the transduction of tat-superoxide dismutase into mammalian cells and skin. Mol Cells 16:402–406
Koren E, Torchilin VP (2012) Cell-penetrating peptides: breaking through to the other side. Trends Mol Med 18:385–393
Kristensen M, Franzyk H, Klausen MT, Iversen A, Bahnsen JS, Skyggebjerg RB, Fodera V, Nielsen HM (2015) Penetratin-mediated transepithelial insulin permeation: importance of cationic residues and pH for complexation and permeation. AAPS J 17:1200–1209
Kumar S, Zakrewsky M, Chen M, Menegatti S, Muraski JA, Mitragotri S (2015) Peptides as skin penetration enhancers: mechanisms of action. J Controlled Release 199:168–178
Kwon SS, Kim SY, Kong BJ, Kim KJ, Noh GY, Im NR, Lim JW, Ha JH, Kim J, Park SN (2015) Cell penetrating peptide conjugated liposomes as transdermal delivery system of Polygonumaviculare L. extract. Int J Pharm 483:26–37
Langel U (2015) Cell-penetrating peptides: methods and protocols. Humana Press, New York
Lehto T, Kurrikoff K, Langel U (2012) Cell-penetrating peptides for the delivery of nucleic acids. Expert Opin Drug Deliv 9:823–836
Lim JM, Chang MY, Park SG, Kang NG, Song YS, Lee YH, Yoo YC, Cho WG, Choi SY, Kang SH (2003) Penetration enhancement in mouse skin and lipolysis in adipocytes by TAT-GKH, a new cosmetic ingredient. J Cosmet Sci 54:483–492
Lindgren M, Hallbrink M, Prochiantz A, Langel U (2000) Cell-penetrating peptides. Trends Pharmacol Sci 21:99–103
Liu X, Zhang W, Wei G, Lu W (2012) Poly (arginine) 8 enhanced intestinal absorption of insulin-loaded nanoparticles. Acta Pharm Sin 47:512–516
Liu X, Liu C, Zhang W, Xie C, Wei G, Lu W (2013) Oligoarginine-modified biodegradable nanoparticles improve the intestinal absorption of insulin. Int J Pharm 448:159–167
Lopes LB, Brophy CM, Furnish E, Flynn CR, Sparks O, Komalavilas P, Joshi L, Panitch A, Bentley MVL (2005) Comparative study of the skin penetration of protein transduction domains and a conjugated peptide. Pharm Res 22:750–757
Lopes LB, Furnish E, Komalavilas P, Seal BL, Panitch A, Bentley MVL, Brophy CM (2008) Enhanced skin penetration of P20 phosphopeptide using protein transduction domains. Eur J Pharm Biopharm 68:441–445
Ma DX, Shi NQ, Qi XR (2011) Distinct transduction modes of arginine-rich cell-penetrating peptides for cargo delivery into tumor cells. Int J Pharm 419:200–208
Madani F, Lindberg S, Langel U, Futaki S, Graslund A (2011) Mechanisms of cellular uptake of cell-penetrating peptides. J Biophys 2011:1–10
Mae M, Langel U (2006) Cell-penetrating peptides as vectors for peptide, protein and oligonucleotide delivery. Curr Opin Pharmacol 6:509–514
Magzoub M, Sandgren S, Lundberg P, Oglęcka K, Lilja J, Wittrup A, Eriksson LG, Langel U, Belting M, Graslund A (2006) N-terminal peptides from unprocessed prion proteins enter cells by macropinocytosis. Biochem Biophys Res Commun 348:379–385
Maiolo JR, Ferrer M, Ottinger EA (2005) Effects of cargo molecules on the cellular uptake of arginine-rich cell-penetrating peptides. Biochim Biophys Acta (BBA)-Biomembranes 1712:161–172
Manosroi J, Lohcharoenkal W, Gotz F, Werner RG, Manosroi W, Manosroi A (2013a) Transdermal absorption and stability enhancement of salmon calcitonin by Tat peptide. Drug Dev Ind Pharm 39:520–525
Manosroi J, Tangjai T, Werner R, Gotz F, Manosroi W, Manosroi A (2013b) Potent and prolonged hypoglycemic activity of an oral insulin–Tat mixture in diabetic mice. Drug Res 63:351–356
Milletti F (2012) Cell-penetrating peptides: classes, origin, and current landscape. Drug Discov Today 17:850–860
Mitragotri S, Burke PA, Langer R (2014) Overcoming the challenges in administering biopharmaceuticals: formulation and delivery strategies. Nat Rev Drug Discov 13:655–672
Morales JO, McConville JT (2014) Novel strategies for the buccal delivery of macromolecules. Drug Dev Ind Pharm 40:579–590
Morishita M, Kamei N, Ehara J, Isowa K, Takayama K (2007) A novel approach using functional peptides for efficient intestinal absorption of insulin. J Controlled Release 118:177–184
Moulton HM, Nelson MH, Hatlevig SA, Reddy MT, Iversen PL (2004) Cellular uptake of antisense morpholino oligomers conjugated to arginine-rich peptides. Bioconjugate Chem 15:290–299
Munyendo WL, Lv H, Benza-Ingoula H, Baraza LD, Zhou J (2012) Cell penetrating peptides in the delivery of biopharmaceuticals. Biomolecules 2:187–202
Nakase I, Hirose H, Tanaka G, Tadokoro A, Kobayashi S, Takeuchi T, Futaki S (2009) Cell-surface accumulation of flock house virus-derived peptide leads to efficient internalization via macropinocytosis. Mol Ther 17:1868–1876
Nan YH, Park IS, Hahm KS, Shin SY (2011) Antimicrobial activity, bactericidal mechanism and LPS-neutralizing activity of the cell-penetrating peptide pVEC and its analogs. J Pept Sci 17:812–817
Nielsen EJB, Yoshida S, Kamei N, Iwamae R, Khafagy ES, Olsen J, Rahbek UL, Pedersen BL, Takayama K, Takeda-Morishita M (2014) In vivo proof of concept of oral insulin delivery based on a co-administration strategy with the cell-penetrating peptide penetratin. J Controlled Release 189:19–24
Oehlke J, Scheller A, Wiesner B, Krause E, Beyermann M, Klauschenz E, Melzig M, Bienert M (1998) Cellular uptake of an α-helical amphipathic model peptide with the potential to deliver polar compounds into the cell interior non-endocytically. Biochim Biophys Acta (BBA)-Biomembranes 1414:127–139
Padari K, Koppel K, Lorents A, Hallbrink M, Mano M, Pedroso de Lima MC, Pooga M (2010) S413-PV cell-penetrating peptide forms nanoparticle-like structures to gain entry into cells. Bioconjugate Chem 21:774–783
Park J, Ryu J, Jin LH, Bahn JH, Kim JA, Yoon CS, Kim DW, Han KH, Eum WS, Kwon HY (2002) 9-polylysine protein transduction domain: enhanced penetration efficiency of superoxide dismutase into mammalian cells and skin. Mol Cells 13:202–208
Patel LN, Wang J, Kim KJ, Borok Z, Crandall ED, Shen WC (2009) Conjugation with cationic cell-penetrating peptide increases pulmonary absorption of insulin. Mol Pharm 6:492–503
Patel VF, Liu F, Brown MB (2011) Advances in oral transmucosal drug delivery. J Controlled Release 153:106–116
Pooga M, Hallbrink M, Zorko M (1998) Cell penetration by transportan. FASEB J 12:67–77
Pouny Y, Rapaport D, Mor A, Nicolas P, Shai Y (1992) Interaction of antimicrobial dermaseptin and its fluorescently labeled analogs with phospholipid membranes. Biochemistry 31:12416–12423
Reissmann S (2014) Cell penetration: scope and limitations by the application of cell-penetrating peptides. J Pept Sci 20:760–784
Rennert R, Neundorf I, Jahnke HG, Suchowerskyj P, Dournaud P, Robitzki A, Beck-Sickinger AG (2008) Generation of carrier peptides for the delivery of nucleic acid drugs in primary cells. ChemMedChem 3:241–253
Rothbard JB, Garlington S, Lin Q, Kirschberg T, Kreider E, McGrane PL, Wender PA, Khavari PA (2000) Conjugation of arginine oligomers to cyclosporin a facilitates topical delivery and inhibition of inflammation. Nat Med 6:1253–1257
Ruczynski J, Wierzbicki PM, Kogut-Wierzbicka M, Mucha P, Siedlecka-Kroplewska K, Rekowski P (2014) Cell-penetrating peptides as a promising tool for delivery of various molecules into the cells. Folia Histochem Cytobiol 52:257–269
Sakuma S, Suita M, Masaoka Y, Kataoka M, Nakajima N, Shinkai N, Yamauchi H, Hiwatari K, Tachikawa H, Kimura R (2010) Oligoarginine-linked polymers as a new class of penetration enhancers. J Controlled Release 148:187–196
Sattar M, Sayed OM, Lane ME (2014) Oral transmucosal drug delivery–current status and future prospects. Int J Pharm 471:498–506
Shan W, Zhu X, Liu M, Li L, Zhong J, Sun W, Zhang Z, Huang Y (2015) Overcoming the diffusion barrier of mucus and absorption barrier of epithelium by self-assembled nanoparticles for oral delivery of insulin. ACS Nano 9:2345–2356
Shi NQ, Qi XR, Xiang B, Zhang Y (2014) A survey on “Trojan Horse” peptides: opportunities, issues and controlled entry to “Troy”. J Controlled Release 194:53–70
Song J, Kai M, Zhang W, Zhang J, Liu L, Zhang B, Liu X, Wang R (2011) Cellular uptake of transportan 10 and its analogs in live cells: selectivity and structure–activity relationship studies. Peptides 32:1934–1941
Steinbach JM, Seo YE, Saltzman WM (2016) Cell penetrating peptide-modified poly (lactic-co-glycolic acid) nanoparticles with enhanced cell internalization. Acta Biomater 30:49–61
Su Y, Mani R, Hong M (2008) Asymmetric insertion of membrane proteins in lipid bilayers by solid-state NMR paramagnetic relaxation enhancement: a cell-penetrating peptide example. J Am Chem Soc 130:8856–8864
Vives E, Brodin P, Lebleu B (1997) A truncated HIV-1 Tat protein basic domain rapidly translocates through the plasma membrane and accumulates in the cell nucleus. J Biol Chem 272:16010–16017
Xia H, Gao X, Gu G, Liu Z, Zeng N, Hu Q, Song Q, Yao L, PangZ Jiang X (2011) Low molecular weight protamine-functionalized nanoparticles for drug delivery to the brain after intranasal administration. Biomaterials 32:9888–9898
Xia H, Gao X, Gu G, Liu Z, Hu Q, Tu Y, Song Q, Yao L, Pang Z, Jiang X (2012) Penetratin-functionalized PEG–PLA nanoparticles for brain drug delivery. Int J Pharm 436:840–850
Yan L, Wang H, Jiang Y, Liu J, Wang Z, Yang Y, Huang S, Huang Y (2013) Cell-penetrating peptide-modified PLGA nanoparticles for enhanced nose-to-brain macromolecular delivery. Macromol Res 21:435–441
Zatsepin T, Turner J, Oretskaya T, Gait M (2005) Conjugates of oligonucleotides and analogues with cell penetrating peptides as gene silencing agents. Curr Pharm Des 11:3639–3654
Zhang L, Torgerson TR, Liu XY, Timmons S, Colosia AD, Hawiger J, Tam JP (1998) Preparation of functionally active cell-permeable peptides by single-step ligation of two peptide modules. Proc Natl Acad Sci 95:9184–9189
Zhang Z, Lv H, Zhou J (2009) Novel solid lipid nanoparticles as carriers for oral administration of insulin. Die Pharmazie-An Int J Pharm Sci 64:574–578
Zhang L, Song L, Zhang C, Ren Y (2012a) Improving intestinal insulin absorption efficiency through coadministration of cell-penetrating peptide and hydroxypropyl-β-cyclodextrin. Carbohydr Polym 87:1822–1827
Zhang Z-H, Zhang Y-L, Zhou J-P, Lv H-X (2012b) Solid lipid nanoparticles modified with stearic acid–octaarginine for oral administration of insulin. Int J Nanomed 7:3333
Zhu S, Chen S, Gao Y, Guo F, Li F, Xie B, Zhou J, Zhong H (2015) Enhanced oral bioavailability of insulin using PLGA nanoparticles co-modified with cell-penetrating peptides and Engrailed secretion peptide (Sec). Drug Deliv 16:1–12
Zorko M, Langel U (2005) Cell-penetrating peptides: mechanism and kinetics of cargo delivery. Adv Drug Deliv Rev 57:529–545
Acknowledgments
All authors declare (S. Bashyal, G. Noh, T. Keum, Y.W. Choi and S. Lee) that they have no conflict of interest. This article does not contain any studies with human and animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bashyal, S., Noh, G., Keum, T. et al. Cell penetrating peptides as an innovative approach for drug delivery; then, present and the future. Journal of Pharmaceutical Investigation 46, 205–220 (2016). https://doi.org/10.1007/s40005-016-0253-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-016-0253-0