Abstract
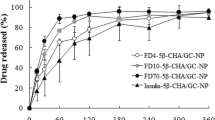
In search of an effective and less toxic absorption enhancer, we synthesized primary amine acetylation of generation 2 polyamidoamine (G2 PAMAM) dendrimer (Ac-G2) by the reaction of G2 PAMAM dendrimer with acetic anhydride, and evaluated the effects of Ac-G2 on the intestinal absorption of poorly absorbable water-soluble drugs using an in situ closed-loop method in rats. The results indicated that Ac50-G2 had a greatest absorption enhancing effect for 5(6)-carboxyfluorescein (CF) in various acetylation levels of G2 PAMAM dendrimers. Ac50-G2 with various concentrations (0.1–1.0%, w/v) could significantly improve the intestinal absorption of alendronate, CF, and fluorescein isothiocyanate-labeled dextrans (FD4), although they did not enhance the absorption of macromolecular drug of FD10, and the absorption enhancement effect of Ac50-G2 was concentration-dependent. Furthermore, we examined the intestinal membrane damage with or without Ac50-G2. The results displayed Ac50-G2 at lower concentrations (0.1–0.5%, w/v) did not cause any observed toxic effect to the intestinal membranes. These findings suggested Ac50-G2 at lower concentrations (below 0.5%, w/v) might be promising as an effective and safe absorption enhancers to promote the intestinal absorption of poorly absorbable drugs.





Similar content being viewed by others
Reference
Yamamoto A, Taniguchi T, Rikyuu K, Tsuji T, Fujita T, Murakami M, Muranishi S. Effects of various protease inhibitors on the intestinal absorption and degradation of insulin in rats. Pharm Res. 1994;11:1496–500.
Dong Z, Hamid KA, Gao Y, Lin Y, Katsumi H, Sakane T, Yamamoto A. Polyamidoamine dendrimers can improve the pulmonary absorption of insulin and calcitonin in rats. J Pharm Sci. 2011;100:1866–78.
Chonkar A, Nayak U, Udupa N. Smart polymers in nasal drug delivery. Indian J Pharm Sci. 2015;77:367–75.
Incecayir T, Sun J, Tsume Y, Xu H, Gose T, Nakanishi T, Tamai I, Hilfinger J, Lipka E, Amidon GL. Carrier-mediated prodrug uptake to improve the oral bioavailability of polar drugs: an application to an oseltamivir analogue. J Pharm Sci. 2016;105:925–34.
Esfand R, Tomalia DA. Poly (amidoamine) (PAMAM) dendrimers: from biomimicry to drug delivery and biomedical applications. Drug Discov Today. 2001;6:427–36.
Svenson S, Tomalia DA. Dendrimers in biomedical applications-reflections on the field. Adv Drug Deliv Rev. 2005;57:2106–29.
Yang H. Targeted nanosystems: Advances in targeted dendrimers for cancer therapy. Nanomedicine. 2016;12:309–16.
Liu Y, Tee JK, Chiu GN. Endrimers in oral drug delivery application: current explorations, toxicity issues and strategies for improvement. Curr Pharm Des. 2015;21:2629–42.
Huang B, Dong WJ, Yang GY, Wang W, Ji CH, Zhou FN. Dendrimer-coupled sonophoresis-mediated transdermal drug-delivery system for diclofenac. Drug Des Devel Ther. 2015;9:3867–76.
Bugno J, Hsu HJ, Hong S. Tweaking dendrimers and dendritic nanoparticles for controlled nano-bio interactions: potential nanocarriers for improved cancer targeting. J Drug Target. 2015;23:642–50.
Serramía MJ, Álvarez S, Fuentes-Paniagua E, Clemente MI, Sánchez-Nieves J, Gómez R, de la Mata J, Muñoz-Fernández MÁ. In vivo delivery of siRNA to the brain by carbosilane dendrimer. J Control Release. 2015;200:60–70.
Bai S, Thomas C, Ahsan F. Dendrimers as a carrier for pulmonary delivery of enoxaparin, a low-molecular weight heparin. J Pharm Sci. 2007;96:2090–106.
Kaminskas LM, McLeod VM, Ascher DB, Ryan GM, Jones S, Chan LJ, Sloan EK, Velkov T, Williams ED, Porter CJ. Methotrexate-conjugated PEGylated dendrimers show differential patterns of deposition and activity intumor-burdened lymph nodes after intravenous and subcutaneous administration in rats. Mol Pharm. 2015;12:432–43.
Hubbard D, Enda M, Bond T, Moghaddam SP, Conarton J, Scaife C, Volckmann E, Ghandehari H. Transepithelial transport of PAMAM dendrimers across isolated human intestinal tissue. Mol Pharm. 2015;12:4099–107.
Chaplot SP, Rupenthal ID. Dendrimers for gene delivery–a potential approach for ocular therapy? J Pharm Pharmacol. 2014;66:542–56.
Hong S, Bielinska AU, Mecke A, Keszler B, Beals JL, Shi X, Balogh L, Orr BG, Baker JRJ, Banaszak Holl MM. Interaction of poly(amidoamine) dendrimers with supported lipid bilayers and cells: hole formation and the relation to transport. Bioconjug Chem. 2004;15:774–82.
Hong S, Rattan R, Majoros IJ, Mullen DG, Peters JL, Shi X, Bielinska AU, Blanco L, Orr BG, Baker JRJ, Banaszak Holl MM. The role of ganglioside GM (1) in cellular internalization mechanisms of poly(amidoamine) dendrimers. Bioconjug Chem. 2009;20:1503–13.
Fetih G, Habib F, Okada N, Fujita T, Attia M, Yamamoto A. Nitric oxide donors can enhance the intestinal transport and absorption of insulin and 1,7 [Asu ]-eel calcitonin in rats. J Control Release. 2005;106:287–97.
Yun MH, Kwon KI. High-performance liquid chromatography method for determining alendronate sodium in human plasma by detecting fluorescence: application to a pharmacokinetic study in humans. J Pharm Biomed Anal. 2006;40:168–72.
Gao Y, He L, Katsumi H, Sakane T, Fujita T, Yamamoto A. Improvement of intestinal absorption of insulin and water-soluble macromolecular compounds by chitosan oligomers in rats. Int J Pharm. 2008a;359:70–8.
Gao Y, He L, Katsumi H, Sakane T, Fujita T, Yamamoto A. Improvement of intestinal absorption of water-soluble macromolecules by various polyamines: intestinal mucosal toxicity and absorption-enhancing mechanism of spermine. Int J Pharm. 2008b;354:126–34.
Dong Z, Katsumi H, Sakane T, Yamamoto A. Effects of polyamidoamine (PAMAM) dendrimers on the nasalabsorption of poorly absorbable drugs in rats. Int J Pharm. 2010;393:244–52.
Karande P, Jain A, Ergun K, Kispersky V, Mitragotri S. Design principles of chemical penetration enhancers for transdermal drug delivery. Proc Natl Acad Sci U S A. 2005;102:4688–93.
Kolhatkar RB, Kitchens KM, Swaan PW, Ghandehari H. Surface acetylation of polyamidoamine (PAMAM) dendrimers decreases cytotoxicity while maintaining membrane permeability. Bioconjug Chem. 2007;18:2054–60.
Venuganti V, Sahdev P, Hildreth M, Guan X, Perumal O. Structure-skin permeability relationship of dendrimers. Pharm Res. 2011;28:2246–60.
Yang Y, Sunoqrot S, Stowell C, Ji J, Lee C, Kim JW, Khan SA, Hong S. Effect of size, surface charge and hydrophobicity of poly(amidoamine) dendrimers on their skin penetration. Biomacromolecules. 2012;13:2154–62.
Jevprasesphant R, Penny J, Attwood D, McKeown NB, D’Emanuele A. Engineering of dendrimer surfaces to enhance transepithelial transport and reduce cytotoxicity. Pharm Res. 2003;20:1543–50.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81260484) and the Program of Guangxi Provincial Natural Science Foundation of China (2013GXNSFAA019226).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Yan, C., Gu, J., Lv, Y. et al. Improved intestinal absorption of water-soluble drugs by acetylation of G2 PAMAM dendrimer nanocomplexes in rat. Drug Deliv. and Transl. Res. 7, 408–415 (2017). https://doi.org/10.1007/s13346-017-0373-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-017-0373-8