Abstract
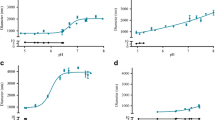
Microemulsions of insulin (50 IU/mL) comprising permeation enhancers were formulated for sublingual delivery. Circular dichroism (CD) spectra indicated conformational stability, while chemical stability was confirmed by high-performance liquid chromatography (HPLC). CD spectra of insulin in combination with permeation enhancers revealed attenuation of molar ellipticity at 274 nm in the order TCTP > TC-AOT > TC > TC-NMT > Sol P > insulin solution. The molar ellipticity ratios at 208/222 nm confirmed dissociation of insulin in the microemulsions with the same rank order. Matrix-assisted laser diffraction ionization mass spectra (MALDI) revealed a significant shift in intensity signals towards monomer and dimers with a substantially high ratio of monomers, especially in the presence of the TCTP and TC-AOT. Permeation through porcine sublingual mucosa correlated with the dissociation data. A high correlation between the ratio of molar ellipticity at 208/222 nm and serum glucose levels (r 2 > 0.958) and serum insulin levels (r 2 > 0.952) strongly suggests the role of dissociation of insulin on enhanced absorption. While all microemulsions revealed a reduction in serum glucose levels and increase in serum insulin levels, significant differences were observed with the TCTP and TC-AOT microemulsions. High pharmacological availability >60 % and bioavailability >55 % compared to subcutaneous insulin at a low dose of 2 IU/kg appears highly promising. The data clearly suggests the additional role of the permeation enhancers on dissociation of insulin on enhanced sublingual absorption from the microemulsions.






Similar content being viewed by others
References
Talegaonkar S et al. Microemulsions: a novel approach to enhanced drug delivery. Recent Patents Drug Deliv Formulation. 2008;2:238–57.
Li L, Nandi I, Kim KH. Development of an ethyl laurate-based microemulsion for rapid-onset intranasal delivery of diazepam. Int J Pharm. 2007;237:77–85.
Yin YM et al. Docetaxel microemulsion for enhanced oral bioavailability: preparation and in vitro and in vivo evaluation. J Control Release. 2009;140:86–94.
Sintov AC, Levy HV, Botner S. Systemic delivery of insulin via the nasal route using a new microemulsion system: in vitro and in vivo studies. J Control Release. 2010;148:168–76.
Kim CK, Ryuu SA, Park KM, Lim SL, Hwang SJ. Preparation and physicochemical characterization of phase inverted water/oil microemulsion containing cyclosporine A. Int J Pharm. 1996;147:131–4.
Graf A, Jack KS, Whittaker AK, Hook SM, Rades T. Protein delivery using nanoparticles based on microemulsions with different structure-types. Eur j Pharm Sci. 2000;33:434–44.
Kinesh VP, Neelam DP, Punit BP, Bhavesh SB, Pragna KS. Novel approaches for oral delivery of insulin and current status of oral insulin products. Int j Pharm Sci Nanotechnol. 2010;3:1057–64.
Sharma G, Wilson K, van der Walle CF, Sattar N, Petrie JR, Ravi Kumar MNV. Microemulsions for oral delivery of insulin: design, development and evaluation in streptozotocin induced diabetic rats. Eur J Pharm Biopharm. 2010;76:159–69.
Ilek AC, Elebi NC, Tırnaksız F, Tay A. A lecithin-based microemulsion of rh-insulin with aprotinin for oral administration: Investigation of hypoglycemic effects in non-diabetic and STZ-induced diabetic rats. Int J Pharm. 2005;298:176–85.
George SM. Pfizer dumps exubera. Nat Biotechnol. 2007;25:1331–2.
Aungst BJ, Rogers NJ, Shefter E. Comparison of nasal, rectal, buccal, sublingual and intramuscular insulin efficacy and the effects of a bile salt absorption promoter. J Pharmacol Exp Ther. 1988;244:23–7.
Jie L et al. Solid lipid nanoparticles for pulmonary delivery of insulin. Int J Pharm. 2008;356:333–44.
Mark MB, Eric MG, Eric JM, Cory B. Pure insulin nanoparticle agglomerates for pulmonary delivery. Langmuir. 2008;24:13614–20.
Cui CY et al. Sublingual delivery of insulin: effects of enhancers on the mucosal lipid fluidity and protein conformation, transport, and in-vivo hypoglycaemic activity. Biol Pharm Bull. 2005;28:2279–88.
Peng H, Gao Q, Chen Q, Zhu Y, Huang KX. Hypoglycemic effects of insulin sublingual drops containing azone on rats and rabbits. Chinese Peptide Symposia. Session G. 2002;221–223.
Cafalu WT. Concept strategies and feasibility of non-invasive insulin delivery. Diabetes Care. 2004;27:230–46.
Li Y, Shao Z, Mitra AK. Dissociation of insulin oligomers by bile salts micelles and its effect on α-chymotrypsin- mediated proteolytic degradation. Pharm Res. 1992;9:864–9.
Quinn R, Andrade JD. Minimizing the aggregation of neutral insulin solution. J Pharm Sci. 1983;72:1472–83.
Lovatt M, Cooper A, Camilleri P. Energetics of cyclodextrin-induced dissociation of insulin. Eur Biophys J. 1996;24:354–7.
Shao Z, Krishnamoorthy R, Mitra AK. Differential effects of anionic, cationic, non-ionic, and physiological surfactants on the dissociation, α-chymotryptic degradation and enteral absorption of insulin hexamers. Pharm Res. 1992;9:1157–63.
Pocker Y, Biswas SB. Self-association of insulin and the role of hydrophobic model of insulin dimerization. Biochemistry. 1981;20:4354–61.
Yong Z et al. A spectroscopic investigation into the interaction between bile salts and insulin in alkaline aqueous solution. J Colloid Interface Sci. 2009;337:322–31.
Kelly SM, Price NC. The use of circular dichroism in the investigation of protein structure and function. Curr Protein Pept Sci. 2000;1:349–84.
Smith GD, Swenson DC, Dodson E, Dodson GG, Reynolds CD. Structural stability in the 4-zinc human insulin hexamers. Proc Natl Acad Sci. 1984;81:7093–7.
Baker EN, Vijayan NM. The structure of 2Zn pig insulin crystals at 1.5 A resolution. Phil Trans R Soc Lond Biol. 1988;319:369–456.
Derewenda U, Swenson D. Phenol stabilizes more helix in a new symmetrical zinc insulin hexamers. Nature. 1989;318:594–6.
Ciszak E, Smith GD. Crystallographic evidence for dual coordination around zinc in the T3R3 human insulin hexamers. Biochemistry. 1994;33:1512–7.
Ciszak E, Beals JM, Frank BH, Baker JC, Carter ND, Smith GD. Role of C-terminal B-chain residues in insulin assembly: the structure of hexameric LysB28ProB29-human insulin. Structure. 1995;3:615–22.
Nettleton EJ et al. Characterization of the oligomeric states of insulin in self-assembly and amyloid fibril formation by mass spectrometry. Biophys J. 2000;79:1053–65.
Livadaris V, Blais JC, Tabet JC. Formation of non-specific protein cluster ion in matrix-assisted laser desorption/ionization: abundances and dynamical aspects. Eur J Mass Spectrum. 2000;6:409–13.
Sheu MT, Chen SY, Chen LC, Ho HO. Influence of micelle solubilization by tocopheryl polyethylene glycol succinate (TPGS) on solubility enhancement and percutaneous penetration of estradiol. J Control Release. 2003;88:355–68.
Gupta RR, Jain SK, Varshney M. AOT water-in-oil microemulsions as a penetration enhancer in transdermal drug delivery of 5-fluorouracil. Colloids Surf B: Biointerfaces. 2005;41:25–32.
Collins P, Laffoon J, Squier CA. Comparative study of porcine oral epithelium. J Dent Res. 1981;60:543.
Sonaje K et al. Biodistribution, pharmacodynamics and pharmacokinetics of insulin analogues in a rat model: oral delivery using pH-Responsive nanoparticles vs. subcutaneous injection. Biomaterials. 2010;31:6849–58.
Wang X, Zheng C, Wu Z, Teng D, Zhang X, Wang Z, et al. Chitosan-NAC nanoparticles as a vehicle for nasal absorption enhancement of insulin. J Biomed Mater Res B Appl Biomater. 2009;88:150–61.
Wang J, Tabata Y, Morimoto K. Aminated gelatin microspheres as a nasal delivery system for peptide drugs: evaluation of in vitro release and in vivo insulin absorption in rats. J Control Release. 2006;113:31–7.
Jose S et al. Cross-linked chitosan microspheres for oral delivery of insulin: Taguchi design and in vivo testing. Colloids Surf B: Biointerfaces. 2012;92:175–9.
Zhang X, Sun M, Zheng A, Cao D, Bi Y, Sun J. Preparation and characterization of insulin-loaded bioadhesive PLGA nanoparticles for oral administration. Eur J Pharm Sci. 2012;45:632–8.
Makhlof A, Tozuka Y, Takeuchi H. Design and evaluation of novel pH-sensitive chitosan nanoparticles for oral insulin delivery. Eur J Pharm Sci. 2011;42:445–51.
Bellary S, Barnett AH. Inhaled insulin: new technology, new possibilities. Int J Clin Pract. 2006;60:728–34.
Acknowledgments
The authors would like to acknowledge University Grants Commission, Government of India for Grant and Fellowship to Nilam Patil. and USV Ltd. India for the gift sample of rh-insulin. We are also thankful to TIFR, India, for the help in the CD spectra and MALDI-MS study.
Conflict of interest
We do not have a financial relationship with the organization that sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patil, N.H., Devarajan, P.V. Enhanced insulin absorption from sublingual microemulsions: effect of permeation enhancers. Drug Deliv. and Transl. Res. 4, 429–438 (2014). https://doi.org/10.1007/s13346-014-0205-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-014-0205-z