Abstract
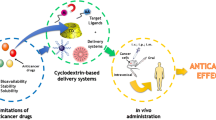
The efficacy and applicability of anticancer drugs are greatly restricted by severe systemic toxicities and drug resistance. Targeting drug delivery strategies have been developed to prevent the shortcomings of chemotherapy. Among various approaches to specifically target drug-loaded carrier systems to the required pathological sites, ligand-attached cyclodextrin-based targeting complexes are a promising drug delivery system, which is achieved mainly through specific molecular interactions between the drugs and cell surface receptors. The principal targeting tactics include conjugation of cyclodextrin with targeting moieties or encapsulation drugs in cyclodextrins. The cyclodextrin-based supramolecules, polymers, or nanoparticles bearing bioactive substances such as folate, estrogens, carbohydrates, peptides, etc. have been reviewed.










Similar content being viewed by others
References
Thun MJ, DeLancey JO, Center MM, Jemal A, Ward EM. The global burden of cancer: priorities for prevention. Carcinogenesis. 2010;31(1):100–10.
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127(12):2893–917.
Gatenby RA. A change of strategy in the war on cancer. Nature. 2009;459(7246):508–9.
Kelloff GJ, Sigman CC. Cancer biomarkers: selecting the right drug for the right patient. Nat Rev Drug Discov. 2012;11(3):201–14.
Undevia SD, Gomez-Abuin G, Ratain MJ. Pharmacokinetic variability of anticancer agents. Nat Rev Cancer. 2005;5(6):447–58.
Wong HL, Wu XY, Bendayan R. Nanotechnological advances for the delivery of CNS therapeutics. Adv Drug Deliv Rev. 2012;64(7):686–700.
Sailor MJ, Park JH. Hybrid nanoparticles for detection and treatment of cancer. Adv Mater. 2012;24(28):3779–802.
Chaudhuri P, Paraskar A, Soni S, Mashelkar RA, Sengupta S. Fullerenol-cytotoxic conjugates for cancer chemotherapy. ACS Nano. 2009;3(9):2505–14.
Zhang X, Wang C. Supramolecular amphiphiles. Chem Soc Rev. 2011;40(1):94–101.
Purkayastha P, Jaffer SS, Ghosh P. Physicochemical perspective of cyclodextrin nano and microaggregates. Phys Chem Chem Phys. 2012;14(16):5339–48.
Dahan A, Miller JM, Hoffman A, Amidon GE, Amidon GL. The solubility–permeability interplay in using cyclodextrins as pharmaceutical solubilizers: mechanistic modeling and application to progesterone. J Pharm Sci-Us. 2010;99(6):2739–49.
Loftsson T, Brewster ME. Cyclodextrins as functional excipients: methods to enhance complexation efficiency. J Pharm Sci-Us. 2012;101(9):3019–32.
Ludwig JA, Weinstein JN. Biomarkers in cancer staging, prognosis and treatment selection. Nat Rev Cancer. 2005;5(11):845–56.
Paliwal SR, Paliwal R, Agrawal GP, Vyas S. Targeted breast cancer nanotherapeutics: options and opportunities with estrogen receptors. Crit Rev Ther Drug Carrier Syst. 2012;29(5):421–46.
Kim HY, Sohn J, Wijewickrama GT, Edirisinghe P, Gherezghiher T, Hemachandra M, et al. Click synthesis of estradiol–cyclodextrin conjugates as cell compartment selective estrogens. Bioorg Med Chem. 2010;18(2):809–21.
Ohrvik VE, Witthoft CM. Human folate bioavailability. Nutrients. 2011;3(4):475–90.
Mattheolabakis G, Rigas B, Constantinides PP. Nanodelivery strategies in cancer chemotherapy: biological rationale and pharmaceutical perspectives. Nanomedicine. 2012;7(10):1577–90.
Muller C, Schibli R. Folic acid conjugates for nuclear imaging of folate receptor-positive cancer. J Nucl Med. 2011;52(1):1–4.
Clementi A, Aversa M, Corsaro C, Spooren J, Stancanelli R, O’Connor C, et al. Synthesis and characterization of a colloidal novel folic acid-β-cyclodextrin conjugate for targeted drug delivery. J Incl Phenom Macrocycl. 2011;69(3):321–5.
Onodera R, Motoyama K, Arima H. Design and evaluation of folate-appended methyl-β-cyclodextrin as a new antitumor agent. J Incl Phenom Macro. 2011;70(3–4):321–6.
Zhao MX, Huang HF, Xia Q, Ji LN, Mao ZW. γ-Cyclodextrin–folate complex-functionalized quantum dots for tumor-targeting and site specific labeling. J Mater Chem. 2011;21(28):10290–7.
Arima H, Arizono M, Higashi T, Yoshimatsu A, Ikeda H, Motoyama K, et al. Potential use of folate-polyethylene glycol (PEG)-appended dendrimer (G3) conjugate with α-cyclodextrin as DNA carriers to tumor cells. Cancer Gene Ther. 2012;19(5):358–66.
Yao H, Ng SS, Tucker WO, Tsang YKT, Man K, Wang XM, et al. The gene transfection efficiency of a folate-PEI600-cyclodextrin nanopolymer. Biomaterials. 2009;30(29):5793–803.
Yang Y, Zhang YM, Chen Y, Zhao D, Chen JT, Liu Y. Construction of a graphene oxide based noncovalent multiple nanosupramolecular assembly as a scaffold for drug delivery. Chem Eur J. 2012;18(14):4208–15.
Zhou Q, Guo X, Chen T, Zhang Z, Shao SJ, Luo C, et al. Target-specific cellular uptake of folate-decorated biodegradable polymer micelles. J Phys Chem B. 2011;115(43):12662–70.
Harada A. Cyclodextrin-based molecular machines. Accounts Chem Res. 2001;34(6):456–64.
Belitsky JM, Stoddart JF. Targeting galectin-1 with self-assembled multivalent pseudopolyrotaxanes. Frontiers in modern carbohydrate chemistry. ACS symposium series. Am Chem Soc. 2007;960:356–74.
Kauscher U, Ravoo BJ. Mannose-decorated cyclodextrin vesicles: the interplay of multivalency and surface density in lectin–carbohydrate recognition. Beilstein J Org Chem. 2012;8:1543–51.
Diaz-Moscoso A, Guilloteau N, Bienvenu C, Mendez-Ardoy A, Blanco JLJ, Benito JM, et al. Mannosyl-coated nanocomplexes from amphiphilic cyclodextrins and pDNA for site-specific gene delivery. Biomaterials. 2011;32(29):7263–73.
Mazzaglia A, Valerio A, Villari V, Rencurosi A, Lay L, Spadaro S, et al. Probing specific protein recognition by size-controlled glycosylated cyclodextrin nanoassemblies. New J Chem. 2006;30(11):1662–8.
Oda Y, Yanagisawa H, Maruyama M, Hattori K, Yamanoi T. Design, synthesis and evaluation of d-galactose-β-cyclodextrin conjugates as drug-carrying molecules. Bioorg Med Chem. 2008;16(19):8830–40.
Salameh A, Lazar AN, Coleman AW, Parrot-Lopez H. Synthesis and interfacial properties of amphiphilic beta-cyclodextrins and their substitution at the O-6 position with a mono bio-recognisable galactosyl antenna. Tetrahedron. 2005;61(36):8740–5.
Grunstein D, Maglinao M, Kikkeri R, Collot M, Barylyuk K, Lepenies B, et al. Hexameric supramolecular scaffold orients carbohydrates to sense bacteria. J Am Chem Soc. 2011;133(35):13957–66.
Anno T, Higashi T, Motoyama K, Hirayama F, Uekama K, Arima H. Potential use of glucuronylglucosyl-β-cyclodextrin/dendrimer conjugate (G2) as a DNA carrier in vitro and in vivo. J Drug Target. 2012;20(3):272–80.
Mujika JI, Escribano B, Akhmatskaya E, Ugalde JM, Lopez X. Molecular dynamics simulations of iron- and aluminum-loaded serum transferrin: protonation of Tyr188 is necessary to prompt metal release. Biochemistry. 2012;51(35):7017–27.
Mahon E, Salvati A, Bombelli FB, Lynch I, Dawson KA. Designing the nanoparticle-biomolecule interface for “targeting and therapeutic delivery”. J Control Release. 2012;161(2):164–74.
Zhang XQ, Xu X, Bertrand N, Pridgen E, Swami A, Farokhzad OC. Interactions of nanomaterials and biological systems: implications to personalized nanomedicine. Adv Drug Deliv Rev. 2012;64(13):1363–84.
Pun SH, Tack F, Bellocq NC, Cheng JJ, Grubbs BH, Jensen GS, et al. Targeted delivery of RNA-cleaving DNA enzyme (DNAzyme) to tumor tissue by transferrin-modified, cyclodextrin-based particles. Cancer Biol Ther. 2004;3(7):641–50.
Jean C, Gravelle P, Fournie JJ, Laurent G. Influence of stress on extracellular matrix and integrin biology. Oncogene. 2011;30(24):2697–706.
Cao F, Li ZB, Lee A, Liu ZF, Chen K, Wang H, et al. Noninvasive de novo imaging of human embryonic stem cell-derived teratoma formation. Cancer Res. 2009;69(7):2709–13.
Hood JD, Bednarski M, Frausto R, Guccione S, Reisfeld RA, Xiang R, et al. Tumor regression by targeted gene delivery to the neovasculature. Science. 2002;296(5577):2404–7.
Kim EM, Jeong MH, Kim DW, Jeong HJ, Lim ST, Sohn MH. Iodine 125-labeled mesenchymal–epithelial transition factor binding peptide-click-cRGDyk heterodimer for glioma imaging. Cancer Sci. 2011;102(8):1516–21.
Miao QH, Li SP, Han SY, Wang Z, Wu Y, Nie GJ. Construction of hydroxypropyl–β-cyclodextrin copolymer nanoparticles and targeting delivery of paclitaxel. J Nanopart Res. 2012;14:1043.
Tran NQ, Joung YK, Lih E, Park KM, Park KD. RGD-conjugated in situ forming hydrogels as cell-adhesive injectable scaffolds. Macromol Res. 2011;19(3):300–6.
O' Neill MJ, Guo JF, Byrne C, Darcy R, O' Driscoll CM. Mechanistic studies on the uptake and intracellular trafficking of novel cyclodextrin transfection complexes by intestinal epithelial cells. Int J Pharm. 2011;413(1–2):174–83.
Diaz-Moscoso A, Vercauteren D, Rejman J, Benito JM, Mellet CO, De Smedt SC, et al. Insights in cellular uptake mechanisms of pDNA-polycationic amphiphilic cyclodextrin nanoparticles (CDplexes). J Control Release. 2010;143(3):318–25.
Mendez-Ardoy A, Guilloteau N, Di Giorgio C, Vierling P, Santoyo-Gonzalez F, Mellet CO, et al. β-Cyclodextrin-based polycationic amphiphilic “click” clusters: effect of structural modifications in their DNA complexing and delivery properties. J Org Chem. 2011;76(15):5882–94.
Guo JF, Ogier JR, Desgranges S, Darcy R, O'Driscoll C. Anisamide-targeted cyclodextrin nanoparticles for siRNA delivery to prostate tumours in mice. Biomaterials. 2012;33(31):7775–84.
Ng KK, Lovell JF, Zheng G. Lipoprotein-inspired nanoparticles for cancer theranostics. Accounts Chem Res. 2011;44(10):1105–13.
Jubeli E, Moine L, Vergnaud-Gauduchon J, Barratt G. E-selectin as a target for drug delivery and molecular imaging. J Control Release. 2012;158(2):194–206.
Kang DI, Lee S, Lee JT, Sung BJ, Yoon JY, Kim JK, et al. Preparation and in vitro evaluation of anti-VCAM-1-Fab′-conjugated liposomes for the targeted delivery of the poorly water-soluble drug celecoxib. J Microencapsul. 2011;28(3):220–7.
Irizarry LR, Hambardzumyan D, Nakano I, Gladson CL, Ahluwalia MS. Therapeutic targeting of VEGF in the treatment of glioblastoma. Expert Opin Ther Targets. 2012;16(10):973–84.
Prakash J, Beljaars L, Harapanahalli AK, Zeinstra-Smith M, de Jager-Krikken A, Hessing M, et al. Tumor-targeted intracellular delivery of anticancer drugs through the mannose-6-phosphate/insulin-like growth factor II receptor. Int J Cancer. 2010;126(8):1966–81.
Jiang J, Deng LX, He LC, Liu HT, Wang CH. Expression, purification, refolding, and characterization of octreotide–interleukin-2: a chimeric tumor-targeting protein. Int J Mol Med. 2011;28(4):549–56.
Lau SKM, Shields DJ, Murphy EA, Desgrosellier JS, Anand S, Huang M, et al. EGFR-mediated carcinoma cell metastasis mediated by integrin αvβ5 depends on activation of c-Src and cleavage of MUC1. Plos One. 2012;7(5):e36753.
Lopez-Calderero I, Chavez ES, Garcia-Carbonero R. The insulin-like growth factor pathway as a target for cancer therapy. Clin Transl Oncol. 2010;12(5):326–38.
Liu SV, Liu SS, Pinski J. Luteinizing hormone-releasing hormone receptor targeted agents for prostate cancer. Expert Opin Inv Drug. 2011;20(6):769–78.
Gilyazova DG, Rosenkranz AA, Gulak PV, Lunin VG, Sergienko OV, Khramtsov YV, et al. Targeting cancer cells by novel engineered modular transporters. Cancer Res. 2006;66(21):10534–40.
Hong H, Yang K, Zhang Y, Engle JW, Feng LZ, Yang YA, et al. In vivo targeting and imaging of tumor vasculature with radiolabeled, antibody-conjugated nanographene. Acs Nano. 2012;6(3):2361–70.
David A. Carbohydrate-based biomedical copolymers for targeted delivery of anticancer drugs. Isr J Chem. 2010;50(2):204–19.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, JJ., Zhou, ZW. & Zhou, SF. Cyclodextrin-based targeting strategies for tumor treatment. Drug Deliv. and Transl. Res. 3, 364–374 (2013). https://doi.org/10.1007/s13346-013-0140-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-013-0140-4