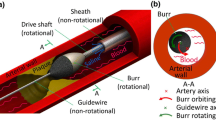
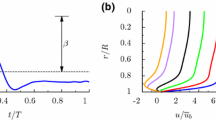
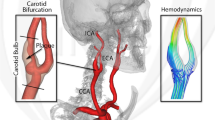
Abstract
Simulations were made of the pressure and velocity fields throughout an artery before and after removal of plaque using orbital atherectomy plus adjunctive balloon angioplasty or stenting. The calculations were carried out with an unsteady computational fluid dynamic solver that allows the fluid to naturally transition to turbulence. The results of the atherectomy procedure leads to an increased flow through the stenotic zone with a coincident decrease in pressure drop across the stenosis. The measured effect of atherectomy and adjunctive treatment showed decrease the systolic pressure drop by a factor of 2.3. Waveforms obtained from a measurements were input into a numerical simulation of blood flow through geometry obtained from medical imaging. From the numerical simulations, a detailed investigation of the sources of pressure loss was obtained. It is found that the major sources of pressure drop are related to the acceleration of blood through heavily occluded cross sections and the imperfect flow recovery downstream. This finding suggests that targeting only the most occluded parts of a stenosis would benefit the hemodynamics. The calculated change in systolic pressure drop through the lesion was a factor of 2.4, in excellent agreement with the measured improvement. The systolic and cardiac-cycle-average pressure results were compared with measurements made in a multi-patient study treated with orbital atherectomy and adjunctive treatment. The agreements between the measured and calculated systolic pressure drop before and after the treatment were within 3%. This excellent agreement adds further confidence to the results. This research demonstrates the use of orbital atherectomy to facilitate balloon expansion to restore blood flow and how pressure measurements can be utilized to optimize revascularization of occluded peripheral vessels.










Similar content being viewed by others
References
Abraham, J. P., E. M. Sparrow, and R. D. Lovik. Unsteady, three dimensional fluid mechanic analysis of blood flow in plaque-narrowed and plaque-free arteries. Int. J. Heat Mass Transf. 51:5633–5641, 2008.
Abraham, J. P., J. C. K. Tong, and E. M. Sparrow. Breakdown of laminar pipe flow into transitional intermittency and subsequent attainment of fully developed intermittent or turbulent flow. Num. Heat Transf. B 54:103–115, 2008.
Abraham, J. P., J. C. K. Tong, and E. M. Sparrow. Prediction of laminar-turbulent transition and friction factors in transitional flows. ASME International Congress and Exposition, Boston, MA, October 31–November 5, 2008.
Abraham, J. P., E. M. Sparrow, and J. C. K. Tong. Heat transfer in all pipe flow regimes—laminar, transitional/intermittent, and turbulent. Int. J. Heat Mass Transf. 52:557–563, 2009.
Abraham, J. P., E. M. Sparrow, J. C. K. Tong, and D. W. Bettenhausen. Internal flows which transit from turbulent through intermittent to laminar. Int. J. Therm. Sci. 49:256–263, 2010.
Abraham, J. P., E. M. Sparrow, and W. J. Minkowycz. Internal-flow nusselt numbers for the low-reynolds-number end of the laminar-to-turbulent transition regime. Int. J. Heat Mass Transf. 54:584–588, 2011.
Abraham, J. P., E. M. Sparrow, J. M. Gorman, J. R. Stark, and R. E. Kohler. A mass transfer model of temporal drug deposition in artery walls. Int. J. Heat Mass Transf. 59:632–638, 2013.
Abraham, J. P., J. R. Stark, J. M. Gorman, E. M. Sparrow, and R. E. Kohler. A model of drug deposition within artery walls. J. Med. Devices 6:020902, 2013.
Abraham J. P., B. D. Plourde, B. Sun, L. J. Vallez, and C. S. Staniloae. The effect of plaque removal on pressure drop and flowrate through a stenotic lesion. Biol. Med. 8, 2015. Article no. 1000261.
Adams, G., P. Khanna, C. Staniloae, J. P. Abraham, and E. M. Sparrow. Optimal techniques with the diamondback 360 system achieve effective results for the treatment of peripheral arterial disease. J. Cardiovasc. Transl. Res. 4:220–229, 2011.
Albuquerque, D. M. S., J. M. C. Pereira, and J. C. F. Pereira. Residual least-squares error estimate for unstructured h-adaptive meshes. Num. Heat Transf. B 67:187–210, 2015.
Baird, R. N., D. R. Bird, P. C. Clifford, R. J. Lusby, R. Skidmore, and J. P. Woodcock. Upstream stenosis: Its diagnosis by doppler signals form the femoral artery. Arch. Surg. 115:1316–1322, 1980.
Bluestein, D., Y. Alemu, I. Avrahami, M. Gharib, K. Dumont, et al. Influence of microcalcifications on vulnerable plaque mechanics. J. Biomech. 41:1111–1118, 2008.
Caiazzo, A., R. Guibert, Y. Boudjemline, and I. E. Vignon-Clementel. Blood flow simulations for the design of stented valve reducer in enlarged ventricular outflow tracts. Cardiovasc. Eng. Technol. 6:485–500, 2015.
Chaudhury, R. A., V. B. Atlasman, G. Pathangey, N. Pracht, R. J. Adrian, and D. H. Frakes. A high performance pulsatile pump for aortic flow experiments in 3-Dimensional models. Cardiovasc. Eng. Technol. 7(2):148–158, 2016.
Cho, Y., and K. Kensey. Effects of the non-Newtonian viscosity of blood on flows in a diseased arterial vessel. Part 1: Steady flows. Biorheology 28:241–261, 1991.
Crosetto, P., P. Reymond, S. Deparis, D. Kontaxakis, N. Stergiopulos, and A. Quarteroni. A fluid-structure interaction simulation of aortic blood flow. Comput. Fluids 43:46–57, 2011.
Domenichini, F., and G. Pedrizzetti. Asymptotic model of fluid-tissue interaction for mitral valve dynamics. Cardiovasc. Eng. Technol. 6:95–104, 2015.
Ellahi, R., S. Rahman, U. Nadeem, and K. Vafai. The blood flow of Prandtl fluid through a tapered stenosed arteries in permeable walls with magnetic field. Commun. Theor. Phys. 63:353–358, 2015.
Friedman, M. H., C. B. Bargeron, and F. F. Mark. Variability of geometry, hemodynamics and intimal response of human arteries. Monogr. Atheroscler. Basel, Karger 15:109–116, 1990.
Gebreegziabher, T., E. M. Sparrow, J. P. Abraham, E. Ayorinde, and T. Singh. High-frequency pulsatile pipe flows encompassing all flow regimes. Num. Heat Transf. A 60:811–826, 2011.
George, S. M., L. M. Eckert, D. R. Martin, and D. P. Giddens. Hemodynamics in normal an diseased livers; applications of image-based computational models. Cardiovasc. Eng. Technol. 6:80–91, 2015.
Gijsen, F., F. van de Vosse, and J. Janssen. The influence of the non-Newtonian properties of blood on the flow in large arteries: steady flow in a carotid bifurcation model. J. Biomech. Eng. 32:601–608, 1999.
Grigoni, M., C. Daniele, M. Umberto, C. Del Gaudio, G. D’Avenio, et al. A mathematical description of blood spiral flow in vessels: application to a numerical study of flow in arterial bending. J. Biomech. Eng. 38:1375–1386, 2005.
Helgeson, Z., J. Jenkins, J. P. Abraham, and E. M. Sparrow. Particle trajectories and agglomeration/accumulation in branching arteries subjected to orbital atherectomy. Open Biomed. Eng. J. 5:25–38, 2011.
Janela, J., A. Moura, and A. Sequeira. A 3D non-Newtonian fluid-structure interaction model for blood flow in arteries. J. Comput. Appl. Mech. 234:2783–2791, 2010.
Barth T. J., and D. C. Jesperson. The design and applications of upwind schemes on unstructured meshes. AIAA paper no. 89-03, 1989.
Johnston, B., P. Johnston, S. Corney, and D. Kilpatrick. Non-Newtonian blood flow in human right coronary arteries: Steady state simulations. J. Biomech. Eng. 37:709–720, 2004.
Kao, N. S. C., T. W. H. Sheu, and S. F. Tsai. On a wavenumber optimized streamline upwinding method for solving steady incompressible Navier-Stokes equations. Num. Heat Transf. B 67:75–99, 2015.
Khakpour, M., and K. Vafai. A critical assessment of arterial transport models. Int. J. Heat Mass Transf. 51:807–822, 2008.
Khaled, A. R. A., and K. Vafai. The role of porous media in modeling flow and heat transfer in biological tissues. Int. J. Heat Mass Transf. 46:4989–5003, 2003.
Khanafer, K., and K. Vafai. The role of porous media in biomedical engineering as related to magnetic resonance imaging and drug delivery. Heat Mass Transfer 42:939–953, 2006.
Ku, D. N. Blood flow in arteries. Ann. Rev. Fluid Mech. 29:399–434, 1997.
Kung, E. O., A. S. Les, C. A. Figueroa, F. Medina, K. Arcaute, et al. In vitro validation of finite element analysis of blood flow in deformable models. Ann. Biomed. Eng. 39:1947–1960, 2011.
LaDisa, J., I. Guler, L. Olson, D. Hetterick, J. Kersten, et al. Three-dimensional computational fluid dynamics modeling of alterations in coronary wall shear stress produced by stent implantation. Ann. Biomed. Eng. 31:972–980, 2003.
Li, Z., and C. Kleinstreuer. Blood flow and structure interactions in a stented abdominal aortic aneurysm model. Med. Eng. Phys. 27:369–382, 2005.
Lillie, J. S., A. S. Liberson, D. Mix, K. Q. Schwarz, A. Chandra, D. B. Phillips, S. W. Day, and D. A. Borkholder. Pulse wave velocity prediction and compliance assessment in elastic arterial segments. Cardiovasc. Eng. Technol. 6:49–58, 2015.
Liu Y., F. Wang, and Y. T. Li. Fourier analysis of the SIMPLE serials. Num. Heat Transf. B, 2016 (in press).
Lovik, R. D., J. P. Abraham, W. J. Minkowycz, and E. M. Sparrow. Laminarization and turbulentization in a pulsatile pipe flow. Num. Heat Transf. A 56:861–879, 2009.
Lovik R. D., J. P. Abraham, and E. M. Sparrow. Pulsating fluid flows undergoing transitions between laminar, transitional, and turbulent regimes. ASME 2009 Summer Bioengineering Conference, Lake Tahoe, CA, June 17–21, 2009.
Majumdar, S. Role of underrelaxation in momentum interpolation for calculation of flow with nonstaggered grids. Num. Heat Transf. 13:125–132, 1988.
Malve, M., A. Garcia, J. Ohayon, and M. A. Martinez. Unsteady blood flow and mass transfer of a human left coronary artery bifurcation: FSI vs. CFD. Int. Commun. Heat Mass Transf. 39:745–751, 2012.
Martin, D., and F. Boyle. Sequential structural and fluid dynamics analysis of balloon-expandable coronary stents: a multivariate statistical analysis. Cardiovasc. Eng. Technol. 6:314–328, 2015.
Menter, F. R. Two-equation eddy-viscosity turbulence models for engineering applications. AIAA J32:1598–1605, 1994.
Menter F. R., T. Esch, and S. Kubacki. Transition modeling based on local variables. In: 5th International Symposium on Engineering Turbulence Modeling and Measurements, Mallorca. Spain, 2002.
Menter F. R., R. Langtry, R. Likki, Y. Suzen, P. Huang, and S. Volker. A correlation–based transition model using local variables, Part I—model formulation. In: Proceedings of ASME Turbo Expo Power for Land, Sea, and Air, Vienna, Austria, June 14–17, 2004.
Menter F. R., R. Langtry, R. Likki, Y. Suzen, P. Huang, and S. Volker. A correlation–based transition model using local variables, Part II—test cases and industrial applications. In: Proceedings of ASME Turbo Expo Power for Land, Sea, and Air, Vienna, Austria, June 14–17, 2004.
Minkowycz, W. J., J. P. Abraham, and E. M. Sparrow. Numerical simulation of laminar breakdown and subsequent intermittent and turbulent flow in parallel plate channels: Effects of inlet velocity profile and turbulence intensity. Int. J. Heat Mass Transf. 52:4040–4046, 2009.
Muehlhausen, M. P., U. Janoske, and H. Oertel, Jr. Implicit partitioned cardiovascular fluid-structure interaction of the heart cycle using non-Newtonian fluid properties and orthotropic material behavior. Cardiovasc. Eng. Technol. 6:8–18, 2015.
Naughton, N. M., B. D. Plourde, J. R. Stark, S. Hodis, and J. P. Abraham. Impacts of waveforms on the fluid flow, wall shear stress, and flow distribution in cerebral aneurysms and the development of a universal reduced pressure. J. Biomed. Sci. Eng. 7:7–14, 2014.
Neofytou, P., and D. Drikakis. Non-Newtonian flow instability in a channel with a sudden expansion. J. Non-Newtonian Fluid Mech. 111:127–150, 2003.
Nguyen, V. T., S. M. N. Wibowo, Y. A. Leow, H. H. Nguyen, Z. Liang, and H. L. Leo. A patient-specific computational fluid dynamic model for hemodynamic analysis of left ventricle diastolic dysfunctions. Cardiovasc. Eng. Technol. 6:412–429, 2015.
O’Rourke, M. F., J. A. Staessen, D. Vlachopoulos, D. Duprez, and G. E. Plante. Clinical applications of arterial stiffness: definitions and reference values. Am. J. Hypertens. 15:426–444, 2002.
Pannier, B. M., A. P. Avolio, A. Hoeks, G. Mancia, and K. Takazawa. Methods and devices for measuring arterial compliance in humans. Am. J. Hypertens. 15:743–753, 2002.
Ramazani-Rend, R., S. Chelikani, E. M. Sparrow, and J. P. Abraham. Experimental and numerical investigation of orbital atherectomy: absence of cavitation. J. Biomed. Sci. Eng. 3:1108–1116, 2010.
Raw MJ, (1996) Robustness of coupled algebraic multigrid for solving Navier Stokes equations. In: AIAA 34th Aerospace and Sciences Meeting and Exhibit, January 15–18, Reno, NV.
Reymond, P., P. Crosetto, S. Deparis, A. Quarteroni, and N. Stergiopulos. Physiological simulation of blood flow in the aorta: comparison of hemodynamic indices as predicted by 3-D FSI, 3-D rigid wall and 1-D models. Med. Eng. Phys. 35:784–791, 2013.
Rhie C. M., and W. L. Chow. A Numerical Study of the Turbulent Flow Past an Isolated Airfoil with Trailing Edge Separation. AIAA paper no. 82-0998, 1982.
Sabbah, H. N., F. Khaja, J. F. Brymer, E. T. Hawkings, and P. D. Stein. Blood flow in the coronary arteries of man: relation to atherosclerosis. Monogr. Atheroscler. Basel, Karger 15:77–90, 1990.
Sparrow, E. M., J. P. Abraham, and W. J. Minkowycz. Flow separation in a diverging conical duct: effect of Reynolds number and divergence angle. Int. J. Heat Mass Transf. 52:3079–3083, 2009.
Sparrow, E. M., J. C. K. Tong, and J. P. Abraham. Fluid flow in a system with separate laminar and turbulent zones. Num. Heat Transf. A 53:341–353, 2008.
Staniloe, C. S., and R. Korabathina. Orbital atherectomy: device evolution and clinical data. J. Invasive Cardiol. 26:215–219, 2014.
Staniloe, C. S., L. Vales, S. Y. Han, J. Sloves, and A. Fallahi. Physiologic guidance of infrainguinal vascular interventions using the pressure wire. J. Invasive Cardiol. 27:483–488, 2015.
Stark, J. R., J. M. Gorman, E. M. Sparrow, J. P. Abraham, and R. E. Kohler. Controlling the rate of penetration of a therapeutic drug into the wall of an artery by means of a pressurized balloon. J. Biomech. Sci. Eng. 6:527–532, 2013.
Stehbens, W. E., P. F. Davis, and B. J. Martin. Blood flow in large arteries: applications to atherogenesis and clinical medicine. Monogr. Atheroscler. Basel, Karger 15:1–15, 1990.
Sun, B., L. J. Vallez, B. D. Plourde, J. R. Stark, and J. P. Abraham. Influence of supporting tissue on the deformation and compliance of healthy and diseased arteries. J. Biomed. Sci. Eng. 8:490–499, 2015.
Tang, D., C. Yang, Y. Huang, and D. N. Ku. A 3-D thin-wall model with fluid-structure interactions for blood flow in carotid arteries with symmetric and asymmetric stenosis. Comput. Struct. 72:357–377, 1999.
Tang, D., C. Yang, Y. Huang, and D. N. Ku. Wall stress and strain analysis using a three-dimensional thick-wall model with fluid-structure interactions for blood flow in carotid arteries with stenosis. Comput. Struct. 72:341–356, 1999.
Tang, D., C. Yang, H. Walker, S. Kobayashi, and D. N. Ku. Simulating cyclic artery compression using a 3D unsteady model with fluid-structure interactions. Comput. Struct. 80:1651–1665, 2002.
Tang, D., C. Yang, S. Kobayashi, and D. N. Ku. Effect of a lipid pool on stress/strain distributions in stenotic arteries: 3-D fluid-structure interactions (FSI) models. J. Biomech. Eng. 126:363–370, 2004.
Tang, D., C. Yang, S. Kobayashi, J. Zheng, and R. P. Vito. Effect of stenosis asymmetry on blood flow and artery compression: a three-dimensional fluid-structure interaction model. Ann. Biomed. Eng. 31:1182–1193, 2003.
Torii, R., M. Oshima, T. Kobayashi, K. Takagi, and T. E. Tezduyar. Fluid-structure interaction modeling of aneurysmal conditions with high and normal blood pressures. Comput. Mech. 38:482–490, 2006.
Torii, R., M. Oshima, T. Kobayashi, K. Takagi, and T. E. Tezduyar. Influence of wall elasticity in patient specific hemodynamic simulations. Comput. Fluids 36:160–168, 2007.
Valencia, A., and M. Villanueva. Unsteady flow and mass transfer in models of stenotic arteries considering fluid-structure interaction. Int. Commun. Heat Mass Transf. 33:966–975, 2006.
Vallez, L. J., B. Sun, B. D. Plourde, and J. P. Abraham. Numerical analysis of arterial plaque thickness and its impact on artery wall compliance. J. Cardiovasc. Med. Cardiol. 2:26–34, 2015.
Walburn, F., and D. Schneck. A constitutive equation for whole human blood. Biorheology 13:201–210, 1976.
Wang, S., and K. Vafai. Analysis of low density lipoprotein (LDL) transport within a curved artery. Ann. Biomed. Eng. 43:1571–1584, 2014.
Welch, T. R., R. C. Eberhart, S. Banerjee, and C. J. Chuong. Mechanical interaction of an expanding coiled stent with a plaque-containing arterial wall: a finite-element analysis. Cardiovasc. Eng. Technol. 7:58–63, 2016.
Zhai S., and X. Feng. A block-centered finite-difference method for the time-fractional diffusion equation on nonuniform grids. Num. Heat Transf. B, 2016 (in press).
Zhao, S. Z., X. Y. Xu, A. D. Hughes, S. A. Thom, A. V. Stanton, et al. Blood flow and vessel mechanics in a physiologically realistic model of a human carotid arterial bifurcation. J. Biomech. 33:975–984, 2000.
Acknowledgments
The authors gratefully acknowledge the support of Cardiovascular Systems Inc.
Conflict of Interest
The authors declare no conflicts of interest.
Statement of Human Studies
Patient data were collected in accordance with Institutional Review Board approval.
Animal Studies
No animals were used in this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Ajit P. Yoganathan oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Plourde, B.D., Vallez, L.J., Sun, B. et al. Alterations of Blood Flow Through Arteries Following Atherectomy and the Impact on Pressure Variation and Velocity. Cardiovasc Eng Tech 7, 280–289 (2016). https://doi.org/10.1007/s13239-016-0269-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-016-0269-7