Abstract
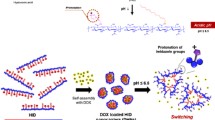
Theranostic systems have been widely investigated for application in the diagnostic therapy of cancers. In this work, hyaluronic acid (HA)-doxorubicin (DOX) conjugate was successfully synthesized by the amide bond formation between amine groups of DOX and carboxylic groups of HA activated with 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide and N-hydroxysulfosuccinimide. The 1H nuclear magnetic resonance (NMR) and gel permeation chromatographic (GPC) analyses confirmed the successful synthesis of HA-DOX conjugates. HA-DOX conjugates self-assembled to micelle-like nanoparticles in aqueous solution. In vitro anti-proliferation tests of cancer cells confirmed the anti-tumor effect of HA-DOX conjugate micelles. For further application, the micelles were coated with a gold (Au) half-shell by the thermal vapor deposition method, which might contribute to prolonging the residence time in the body with a versatile surface modification on the Au surface. TEM analysis confirmed the successful Au coating on HA-DOX conjugate micelles with a thickness of 15 nm. The Au half-shell-coated HADOX conjugate micelles might be successfully exploited for cancer theranostic applications such as chemotherapy, hyperthermia, and bioimaging.

Similar content being viewed by others
References
J. H. Schiller, D. Harrington, C. P. Belani, C. Langer, A. Sandler, J. Krook, J. Zhu, and D. H. Johnson, N. Engl. J. Med., 346, 92 (2002).
L. K. Fung and W. M. Saltzman, Adv. Drug Deliv. Rev., 26, 209 (1997).
B. Sumer and J. Gao, Nanomedicine, 3, 137 (2008).
W. Sadee, Pharm. Res., 25, 2713 (2008).
P. Ghosh, G. Han, M. De, C. K. Kim, and V. M. Rotello, Adv. Drug Deliv. Rev., 60, 1307 (2008).
N. Li, T. Larson, H. H. Nguyen, K. V. Sokolov, and A. D. Ellington, Chem. Commun., 46, 392 (2010).
J. K. Kim and H. Ahn, Macromol. Res., 16, 163 (2008).
M. Y. Lee, S. J. Park, K. T. Park, K. S. Kim, H. Lee, and S. K. Hahn, ACS Nano, 5, 6138 (2011).
J. Chen, M. Yang, Q. Zhang, E. C. Cho, C. M. Cobley, C. Kim, C. Glaus, L. V. Wang, M. J. Welch, and Y. Xia, Adv. Funct. Mater., 20, 3684 (2010).
L. Tong, Q. Wei, A. Wei, and J. -X. Cheng, Photochem. Photobiol., 85, 21 (2009).
L. R. Hirsch, R. J. Stafford, J. A. Bankson, S. R. Sershen, B. Rivera, R. E. Price, J. D. Hazle, N. J. Halas, and J. L. West, Proc. Natl. Acad. Sci. U.S.A., 100, 13549 (2003).
Y. Wang, X. Xie, X. Wang, G. Ku, K. L. Gill, D. P. O’Neal, G. Stoica, and L. V. Wang, Nano Lett., 4, 1689 (2004).
J. Yang, J. Lee, J. Kang, S. J. Oh, H. J. Ko, J. H. Son, K. Lee, J. S. Suh, Y. M. Huh, and S. Haam, Adv. Mater., 21, 4339 (2009).
T. C. Laurent, U. B. G. Laurent, and J. R. E. Fraser, Immunol. Cell. Biol., 74, A1–7 (1996).
J. H. Kong, E. J. Oh, S. Y. Chae, K. C. Lee, and S. K. Hahn, Biomaterials, 31, 4121 (2010).
E. J. Oh, K. Park, K. S. Kim, J. Kim, J. A. Yang, and J. H. Kong, J. Control. Release, 141, 2 (2010).
J. K. Park, J. S. Yeom, S. K. Hahn, E. J. Hwang, J. S. Shim, I. H. Cho, S. H. Bhang, and B. S. Kim, Macromol. Res., 18, 1076 (2010).
H. G. Kang, E. J. Oh, J. K. Park, K. S. Kim, S. H. Ryu, and S. K. Hahn, Macromol. Res., 16, 182 (2008).
T. Ito, T. Iida-Tanaka, K. Niidome, K. Kawano, T. Kubo, Z. Yoshikawa, T. Sato, Z. Yang, and Y. Koyama, J. Control. Release, 112, 382 (2006).
A. Aruffo, I. Stamenkovic, M. Melnick, C. B. Underhill, and B. Seed, Cell, 61, 1303 (1990).
J. Entwistle, C. L. Hall, and E. A. Turley, J. Cell. Biochem., 61, 569 (1996).
B. Zhou, J. A. Weigel, L. Fauss, and P. H. Weigel, J. Biol. Chem., 275, 37733 (2000).
Y. Li and G. S. Kwon, Pharm. Res., 17, 607 (2000).
Y. Luo, N. J. Bernshaw, Z. R. Lu, J. Kopecek, and G. D. Prestwich, Pharm. Res., 19, 396 (2002).
J. C. Love, B. D. Gates, D. B. Wolfe, K. E. Paul, and G. M. Whitesides, Nano Lett., 2, 891 (2002).
Y. Lu, G. L. Liu, J. Kim, Y. X. Mejia, and L. P. Lee, Nano Lett., 5, 119 (2005).
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors equally contributed to this work.
Rights and permissions
About this article
Cite this article
Kim, K.S., Park, SJ., Lee, MY. et al. Gold half-shell coated hyaluronic acid-doxorubicin conjugate micelles for theranostic applications. Macromol. Res. 20, 277–282 (2012). https://doi.org/10.1007/s13233-012-0062-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-012-0062-x