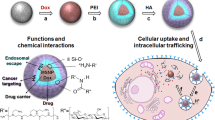
Abstract
The tuning of physicochemical properties like particle sizes, surface modifications, etc., is essential to improve delivery efficiency with insignificant toxicity. A quantitative and mechanistic investigation on cellular uptake and intracellular trafficking of fluorescent-labeled polystyrene particles [50 and 500 nm plain and 200 nm and 1 µm carboxylate-coated polystyrene (PPS and CPS)] against breast cancer cells was performed. Examinations of cell proliferation, viability, and cytotoxicity indicated that those applied polymeric particles at concentrations of 4–40 µg/mL are non-toxic to cells except 200 nm CPS (16 µg/mL) which partially reduced cell viability. Quantitative fluorescent microscopy revealed that 200 nm CPS displayed the highest capability to be internalized by MCF-7 cells, in which the average intracellular particles are about 14 and 35 folds higher than 1 µm CPS and 500 nm PPS, respectively. Analysis of the uptake routes using inhibitors provided clear evidence that 200 CPS was mainly ingested via the clathrin-mediated endocytosis, whereas larger particles (> 500 nm) preferentially utilized the macropinocytosis pathway. Fluorescent live-cell imaging suggested that particles were mainly internalized by cells in an individual form instead of large aggregates, despite late occurrence of partial agglomeration in the cytoplasm. Overall, those outcomes enhanced our understanding of the application of polymeric delivery vehicles for the treatment of breast cancer.










Similar content being viewed by others
References
Alhalili Z, Figueroa D, Johnston MR, Shapter J, Sanderson B (2016) Effect of modification protocols on the effectiveness of gold nanoparticles as drug delivery vehicles for killing of breast cancer cells. Aust J Chem 69(12):1402–1412
Alvarez-Roman R, Naik A, Kalia YN, Guy RH, Fessi H (2004) Skin penetration and distribution of polymeric nanoparticles. J Control Release 99(1):53–62. https://doi.org/10.1016/j.jconrel.2004.06.015
Alvarez RH (2010) Present and future evolution of advanced breast cancer therapy. Breast Cancer Res 12(Suppl 2):S1. https://doi.org/10.1186/bcr2572
Anari E, Akbarzadeh A, Zarghami N (2016) Chrysin-loaded PLGA-PEG nanoparticles designed for enhanced effect on the breast cancer cell line. Artif Cells Nanomed Biotechnol 44(6):1410–1416. https://doi.org/10.3109/21691401.2015.1029633
Borker S, Pokharkar V (2018) Engineering of pectin-capped gold nanoparticles for delivery of doxorubicin to hepatocarcinoma cells: an insight into mechanism of cellular uptake. Artif Cells Nanomed Biotechnol 46(sup2):826–835. https://doi.org/10.1080/21691401.2018.1470525
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
Calero M, Chiappi M, Lazaro-Carrillo A et al (2015) Characterization of interaction of magnetic nanoparticles with breast cancer cells. J Nanobiotechnology 13:16. https://doi.org/10.1186/s12951-015-0073-9
Cartiera MS, Johnson KM, Rajendran V, Caplan MJ, Saltzman WM (2009) The uptake and intracellular fate of PLGA nanoparticles in epithelial cells. Biomaterials 30(14):2790–2798. https://doi.org/10.1016/j.biomaterials.2009.01.057
Ciapetti G, Cenni E, Pratelli L, Pizzoferrato A (1993) In vitro evaluation of cell/biomaterial interaction by MTT assay. Biomaterials 14(5):359–364. https://doi.org/10.1016/0142-9612(93)90055-7
Conner SD, Schmid SL (2003) Regulated portals of entry into the cell. Nature 422(6927):37–44
Cui JF, Zhang ZY, Liu DD et al (2019) Unprecedented piezoresistance coefficient in strained silicon carbide. Nano Lett 19(9):6569–6576. https://doi.org/10.1021/acs.nanolett.9b02821
Danhier F, Ansorena E, Silva JM, Coco R, Le Breton A, Preat V (2012) PLGA-based nanoparticles: an overview of biomedical applications. J Control Release 161(2):505–522. https://doi.org/10.1016/j.jconrel.2012.01.043
Doroudian M, MacLoughlin R, Poynton F, Prina-Mello A, Donnelly SC (2019) Nanotechnology based therapeutics for lung disease. Thorax. https://doi.org/10.1136/thoraxjnl-2019-213037
dos Santos T, Varela J, Lynch I, Salvati A, Dawson KA (2011) Effects of transport inhibitors on the cellular uptake of carboxylated polystyrene nanoparticles in different cell lines. PLoS ONE 6(9):e24438. https://doi.org/10.1371/journal.pone.0024438
Fakhoury I, Saad W, Bouhadir K, Nygren P, Schneider-Stock R, Gali-Muhtasib H (2016) Uptake, delivery, and anticancer activity of thymoquinone nanoparticles in breast cancer cells. J Nanopart Res 18(7):210. https://doi.org/10.1007/s11051-016-3517-8
Firdessa R, Oelschlaeger TA, Moll H (2014) Identification of multiple cellular uptake pathways of polystyrene nanoparticles and factors affecting the uptake: relevance for drug delivery systems. Eur J Cell Biol 93(8–9):323–337. https://doi.org/10.1016/j.ejcb.2014.08.001
Foroozandeh P, Aziz AA (2018) Insight into cellular uptake and intracellular trafficking of nanoparticles. Nanoscale Res Lett 13(1):339. https://doi.org/10.1186/s11671-018-2728-6
Fotakis G, Timbrell JA (2006) In vitro cytotoxicity assays: comparison of LDH, neutral red, MTT and protein assay in hepatoma cell lines following exposure to cadmium chloride. Toxicol Lett 160(2):171–177. https://doi.org/10.1016/j.toxlet.2005.07.001
Gal N, Massalha S, Samuelly-Nafta O, Weihs D (2015) Effects of particle uptake, encapsulation, and localization in cancer cells on intracellular applications. Med Eng Phys 37(5):478–483. https://doi.org/10.1016/j.medengphy.2015.03.003
Gratton SE, Ropp PA, Pohlhaus PD et al (2008) The effect of particle design on cellular internalization pathways. Proc Natl Acad Sci USA 105(33):11613–11618. https://doi.org/10.1073/pnas.0801763105
Hu YB, Dammer EB, Ren RJ, Wang G (2015) The endosomal-lysosomal system: from acidification and cargo sorting to neurodegeneration. Transl Neurodegener 4:18. https://doi.org/10.1186/s40035-015-0041-1
Jiang L, Li X, Liu L, Zhang Q (2013) Cellular uptake mechanism and intracellular fate of hydrophobically modified pullulan nanoparticles. Int J Nanomed 8:1825–1834. https://doi.org/10.2147/IJN.S44342
Jiang X, Musyanovych A, Rocker C, Landfester K, Mailander V, Nienhaus GU (2011) Specific effects of surface carboxyl groups on anionic polystyrene particles in their interactions with mesenchymal stem cells. Nanoscale 3(5):2028–2035. https://doi.org/10.1039/c0nr00944j
Kosheleva OK, Lai TC, Chen NG, Hsiao M, Chen CH (2016) Selective killing of cancer cells by nanoparticle-assisted ultrasound. J Nanobiotechnol 14(1):46. https://doi.org/10.1186/s12951-016-0194-9
Kumar P, Nagarajan A, Uchil PD (2018) Analysis of cell viability by the lactate dehydrogenase assay. Cold Spring Harb Protoc. https://doi.org/10.1101/pdb.prot095497
Kumari A, Yadav SK, Yadav SC (2010) Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf B Biointerfaces 75(1):1–18. https://doi.org/10.1016/j.colsurfb.2009.09.001
Mahmoudi M, Sant S, Wang B, Laurent S, Sen T (2011) Superparamagnetic iron oxide nanoparticles (SPIONs): development, surface modification and applications in chemotherapy. Adv Drug Deliver Rev 63(1–2):24–46
Nam HY, Kwon SM, Chung H et al (2009) Cellular uptake mechanism and intracellular fate of hydrophobically modified glycol chitosan nanoparticles. J Control Release 135(3):259–267
Nambiar S, Osei E, Fleck A, Darko J, Mutsaers AJ, Wettig S (2018) Synthesis of curcumin-functionalized gold nanoparticles and cytotoxicity studies in human prostate cancer cell line. Appl Nanosci 8(3):347–357
Nelemans LC, Gurevich L (2020) Drug delivery with polymeric nanocarriers-cellular uptake mechanisms. Materials (Basel). https://doi.org/10.3390/ma13020366
Ngamwongsatit P, Banada PP, Panbangred W, Bhunia AK (2008) WST-1-based cell cytotoxicity assay as a substitute for MTT-based assay for rapid detection of toxigenic Bacillus species using CHO cell line. J Microbiol Methods 73(3):211–215. https://doi.org/10.1016/j.mimet.2008.03.002
Nounou MI, ElAmrawy F, Ahmed N, Abdelraouf K, Goda S, Syed-Sha-Qhattal H (2015) Breast cancer: conventional diagnosis and treatment modalities and recent patents and technologies. Breast Cancer (Auckl) 9(Suppl 2):17–34. https://doi.org/10.4137/BCBCR.S29420
Panyam J, Zhou WZ, Prabha S, Sahoo SK, Labhasetwar V (2002) Rapid endo-lysosomal escape of poly(DL-lactide-co-glycolide) nanoparticles: implications for drug and gene delivery. FASEB J 16(10):1217–1226. https://doi.org/10.1096/fj.02-0088com
Rosman C, Pierrat S, Henkel A et al (2012) A new approach to assess gold nanoparticle uptake by mammalian cells: combining optical dark-field and transmission electron microscopy. Small 8(23):3683–3690. https://doi.org/10.1002/smll.201200853
Shi J, Kantoff PW, Wooster R, Farokhzad OC (2017) Cancer nanomedicine: progress, challenges and opportunities. Nat Rev Cancer 17(1):20–37. https://doi.org/10.1038/nrc.2016.108
Shukla R, Bansal V, Chaudhary M, Basu A, Bhonde RR, Sastry M (2005) Biocompatibility of gold nanoparticles and their endocytotic fate inside the cellular compartment: a microscopic overview. Langmuir 21(23):10644–10654
Sutherland RL, Hall RE, Taylor IW (1983) Cell proliferation kinetics of MCF-7 human mammary carcinoma cells in culture and effects of tamoxifen on exponentially growing and plateau-phase cells. Cancer Res 43(9):3998–4006
Thilagam R, Gnanamani A (2020) Preparation, characterization and stability assessment of keratin and albumin functionalized gold nanoparticles for biomedical applications. Appl Nanosci. https://doi.org/10.1007/s13204-020-01250-z
Thompson EA, Graham E, MacNeill CM et al (2014) Differential response of MCF7, MDA-MB-231, and MCF 10A cells to hyperthermia, silver nanoparticles and silver nanoparticle-induced photothermal therapy. Int J Hyperthermia 30(5):312–323. https://doi.org/10.3109/02656736.2014.936051
Tominaga H, Ishiyama M, Ohseto F et al (1999) A water-soluble tetrazolium salt useful for colorimetric cell viability assay. Anal Commun 36(2):47–50
van Meerloo J, Kaspers GJ, Cloos J (2011) Cell sensitivity assays: the MTT assay. Methods Mol Biol 731:237–245. https://doi.org/10.1007/978-1-61779-080-5_20
Vishwakarma SK, Bardia A, Lakkireddy C, Paspala SAB, Habeeb MA, Khan AA (2018) Real-time cellular and molecular dynamics of bi-metallic self-therapeutic nanoparticle in cancer cells. Appl Nanosci 8(1–2):115–124
Xie Y, Liu D, Cai C et al (2016) Size-dependent cytotoxicity of Fe3O4 nanoparticles induced by biphasic regulation of oxidative stress in different human hepatoma cells. Int J Nanomed 11:3557–3570. https://doi.org/10.2147/IJN.S105575
Yuan JD, ZhuGe DL, Tong MQ et al (2018) pH-sensitive polymeric nanoparticles of mPEG-PLGA-PGlu with hybrid core for simultaneous encapsulation of curcumin and doxorubicin to kill the heterogeneous tumour cells in breast cancer. Artif Cells Nanomed Biotechnol 46(sup1):302–313. https://doi.org/10.1080/21691401.2017.1423495
Zeighamian V, Darabi M, Akbarzadeh A et al (2016) PNIPAAm-MAA nanoparticles as delivery vehicles for curcumin against MCF-7 breast cancer cells. Artif Cell Nanomed B 44(2):735–742
Zhang Z, Du Y, Huang S et al (2020a) Macroscale superlubricity enabled by graphene-coated surfaces. Adv Sci (Weinh) 7(4):1903239. https://doi.org/10.1002/advs.201903239
Zhang ZY, Cui JF, Zhang JB, Liu DD, Yu ZJ, Guo DM (2019) Environment friendly chemical mechanical polishing of copper. Appl Surf Sci 467:5–11. https://doi.org/10.1016/j.apsusc.2018.10.133
Zhang ZY, Guo DM, Wang B, Kang RK, Zhang B (2015a) A novel approach of high speed scratching on silicon wafers at nanoscale depths of cut. Sci Rep-Uk. https://doi.org/10.1038/srep16395(ARTN 16395)
Zhang ZY, Huo YX, Guo DM (2013) A model for nanogrinding based on direct evidence of ground chips of silicon wafers. Sci China Technol Sci 56(9):2099–2108. https://doi.org/10.1007/s11431-013-5286-2
Zhang ZY, Liao LX, Wang XZ, Xie WX, Guo DM (2020b) Development of a novel chemical mechanical polishing slurry and its polishing mechanisms on a nickel alloy. Appl Surf Sci. https://doi.org/10.1016/j.apsusc.2019.144670(ARTN 144670)
Zhang ZY, Wang B, Kang RK, Zhang B, Guo DM (2015b) Changes in surface layer of silicon wafers from diamond scratching. Cirp Ann-Manuf Technol 64(1):349–352. https://doi.org/10.1016/j.cirp.2015.04.005
Acknowledgements
This work was supported to Dr. Jie Chen by the National Natural Science Foundation of China (81600063) and Natural Science Foundation of Hunan Province (2017JJ3484).
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Video 1, 2, and 3: Cellular uptake and dynamic and intracellular redistribution of 500 nm plain polystyrene particles and 200 nm and 1 μm carboxylate-coated polystyrene particles incubated individually with MCF-7 cells obtained by fluorescence and bright field live-cell imaging. (AVI 1734 kb)
Supplementary file2 (AVI 1443 kb)
Supplementary file3 (AVI 1450 kb)
Rights and permissions
About this article
Cite this article
Zhou, Mr., Xie, Pf., Gong, J. et al. Mechanistic investigation of cellular internalization routes of polymeric particles on breast cancer cells: relevance for drug delivery applications. Appl Nanosci 10, 2239–2254 (2020). https://doi.org/10.1007/s13204-020-01376-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-020-01376-0