Abstract
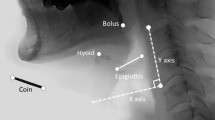
Oropharyngeal dysphagia (OD) is a common post-stroke complication and is associated with respiratory infections. The aim was to assess the biomechanical impairments in swallow function and the afferent and efferent swallowing pathways impairing swallow safety in chronic post-stroke patients. We studied 30 patients with unilateral stroke and chronic OD (> 3 months from stroke onset) with impaired safety of swallow (Penetration–Aspiration Scale [PAS] ≥ 2). We evaluated the efficacy, safety, and kinematics of the swallow response (residue, PAS, laryngeal vestibule closure time [LVCT]) with videofluoroscopy, sensory evoked potentials to pharyngeal electrical stimulation (pSEP), and pharyngeal motor evoked potentials (pMEP) to transcranial magnetic stimulation of both hemispheres. Mean age of patients was 70.1 ± 10.9 years (7 women). Stroke severity at onset was moderate (NIHSS median 10 [IQ range 3–11.5]), and modified Rankin Scale 2.8 ± 1.3. Mean PAS was 5.1 ± 1.9; prevalence of delayed LVCT was 86.7% and 30% presented aspirations. Pharyngeal hypoesthesia was present in 46.7% of patients and 92.3% showed abnormally asymmetrical pSEPs when comparing the ipsilesional with the contralesional hemisphere. Increased duration of swallow was associated with lower pSEP amplitude (P1-N2) in the contralesional hemisphere (p = 0.033). Patients with right hemispheric strokes showed greater reduction of pSEPs amplitude (N1-P1, p = 0.049). In contrast, pharyngeal resting motor threshold and pMEPs were symmetric in 73.3% patients without the physiologic hemispheric dominance. Mild-to-moderate disabled chronic post-stroke patients with OD presented severe impaired biomechanics of swallow response and high prevalence of aspirations. Initial results from the neurophysiological evaluation demonstrated prevalent impairments with disrupted integration of pharyngeal sensory inputs and reduced cortical excitability of efferent pathways. Patients with right hemispheric strokes showed poorer neurophysiological responses.



Similar content being viewed by others
References
Attrill S, White S, Murray J, Hammond S, Doeltgen S. Impact of oropharyngeal dysphagia on healthcare cost and length of stay in hospital: a systematic review. BMC Health Serv Res. 2018;18(1):594. https://doi.org/10.1186/s12913-018-3376-3.
Rofes L, Muriana D, Palomeras E, Vilardell N, Palomera E, Alvarez-Berdugo D, et al. Prevalence, risk factors and complications of oropharyngeal dysphagia in stroke patients: a cohort study. Neurogastroenterol Motil. 2018;30:e13338. https://doi.org/10.1111/nmo.13338.
Vilardell N, Rofes L, Arreola V, Martin A, Muriana D, Palomeras E, et al. Videofluoroscopic assessment of the pathophysiology of chronic poststroke oropharyngeal dysphagia. Neurogastroenterol Motil. 2017;29:1–8. https://doi.org/10.1111/nmo.13111.
Paciaroni M, Mazzotta G, Corea F, Caso V, Venti M, Milia P, et al. Dysphagia following stroke. Eur Neurol. 2004;51:162–7.
Toscano M, Cecconi E, Capiluppi E, Viganò A, Bertora P, Campiglio L, et al. Neuroanatomical, clinical and cognitive correlates of post-stroke dysphagia. Eur Neurol. 2015;74:171–7.
Cabib C, Ortega O, Vilardell N, Mundet L, Clavé P, Rofes L. Chronic post-stroke oropharyngeal dysphagia is associated with impaired cortical activation to pharyngeal sensory inputs. Eur J Neurol. 2017;24:1355–62. https://doi.org/10.1111/ene.13392.
Marian T, Schröder JB, Muhle P, Claus I, Riecker A, Warnecke T, et al. Pharyngolaryngeal sensory deficits in patients with middle cerebral artery infarction: lateralization and relation to overall dysphagia severity. Cerebrovasc Dis Extra. 2017;7:130–9. https://doi.org/10.1159/000479483.
Wilmskoetter J, Martin-Harris B, Pearson WG Jr, Bonilha L, Elm JJ, Horn J, et al. Differences in swallow physiology in patients with left and right hemispheric strokes. Physiol Behav. 2018;194:144–52. https://doi.org/10.1016/j.physbeh.2018.05.010.
Hamdy S, Aziz Q, Rothwell JC, Singh KD, Barlow J, Hughes DG, et al. The cortical topography of human swallowing musculature in health and disease. Nat Med. 1996;2:1217–24.
Hamdy S, Aziz Q, Rothwell JC, Power M, Singh KD, Nicholson DA, et al. Recovery of swallowing after dysphagic stroke relates to functional reorganization in the intact motor cortex. Gastroenterology. 1998;115:1104–12.
Aviv JE, Martin JH, Sacco RL, Zagar D, Diamond B, Keen MS, et al. Supraglottic and pharyngeal sensory abnormalities in stroke patients with dysphagia. Ann Otol Rhinol Laryngol. 1996;105:92–7.
Hamdy S, Aziz Q, Rothwell JC, Crone R, Hughes D, Tallis RC, et al. Explaining oropharyngeal dysphagia after unilateral hemispheric stroke. Lancet. 1997;350:686–92.
Mihai PG, Otto M, Domin M, Platz T, Hamdy S, Lotze M. Brain imaging correlates of recovered swallowing after dysphagic stroke: a fMRI and DWI study. Neuroimage Clin. 2016;12:1013–21.
Teismann IK, Steinstraeter O, Warnecke T, Ringelstein EB, Pantev C, Dziewas R. Measurement of pharyngeal sensory cortical processing: technique and physiologic implications. BMC Neurosci. 2009;10:76.
Onofri SM, Cola PC, Berti LC, da Silva RG, Dantas RO. Correlation between laryngeal sensitivity and penetration/aspiration after stroke. Dysphagia. 2014;29:256–61. https://doi.org/10.1007/s00455-013-9504-7.
Setzen M, Cohen MA, Mattucci KF, Perlman PW, Ditkoff MK. Laryngopharyngeal sensory deficits as a predictor of aspiration. Otolaryngol Head Neck Surg. 2001;124:622–4.
Wilson JT, Hareendran A, Grant M, Baird T, Schulz UG, Muir KW, et al. Improving the assessment of outcomes in stroke: use of a structured interview to assign grades on the modified Rankin Scale. Stroke. 2002;33:2243–6.
Bamford J, Sandercock P, Dennis M, Burn J, Warlow C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet. 1991;337:1521–6.
Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA. MR signal abnormalities at 1.5T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol. 1987;149:351–6.
Clavé P, Arreola V, Romea M, Medina L, Palomera E, Serra-Prat M. Accuracy of the volume-viscosity swallow test for clinical screening of oropharyngeal dysphagia and aspiration. Clin Nutr. 2008;27:806–15. https://doi.org/10.1016/j.clnu.2008.06.011.
Rosenbek JC, Robbins JA, Roecker EB, Coyle JL, Wood JL. A penetration aspiration scale. Dysphagia. 1996;11:93–8.
Rofes L, Arreola V, Romea M, Palomera E, Almirall J, Cabré M, et al. Pathophysiology of oropharyngeal dysphagia in the frail elderly. Neurogastroenterol Motil. 2010;22:851–8. https://doi.org/10.1111/j.1365-2982.2010.01521.x.
Robbins J, Hamilton JW, Lof GL, Kempster GB. Oropharyngeal swallowing in normal adults of different ages. Gastroenterology. 1992;103:823–9.
Kahrilas PJ, Lin S, Rademaker AW, Logemann JA. Impaired deglutitive airway protection: a videofluoroscopic analysis of severity and mechanism. Gastroenterology. 1997;113:1457–64.
Kobayashi M, Pascual-Leone A. Transcranial magnetic stimulation in neurology. Lancet Neurol. 2003;2:145–56.
Michou E, Mistry S, Jefferson S, Tyrrell P, Hamdy S. Characterizing the mechanisms of central and peripheral forms of neurostimulation in chronic dysphagic stroke patients. Brain Stimul. 2014;7:66–73. https://doi.org/10.1016/j.brs.2013.09.005.
Rofes L, Ortega O, Vilardell N, Mundet L, Clavé P. Spatiotemporal characteristics of the pharyngeal event-related potential in healthy subjects and older patients with oropharyngeal dysfunction. Neurogastroenterol Motil. 2017;29. https://doi.org/10.1111/nmo.12916.
Rofes L, Arreola V, López I, Martin A, Sebastián M, Ciurana A, et al. Effect of surface sensory and motor electrical stimulation on chronic poststroke oropharyngeal dysfunction. Neurogastroenterol Motil. 2013;25:888–701. https://doi.org/10.1111/nmo.12211.
Matamala JM, Núñez C, Lera L, Verdugo RJ, Sánchez H, Albala C, et al. Motor evoked potentials by transcranial magnetic stimulation in healthy elderly people. Somatosens Mot Res. 2013;30:201–5. https://doi.org/10.3109/08990220.2013.796922.
Martino R, Foley N, Bhogal S, Diamant N, Speechley M, Teasell R. Dysphagia after stroke-incidence, diagnosis, and pulmonary complications. Stroke. 2005;36:2756–63.
Mann G, Hankey GJ, Cameron D. Swallowing function after stroke: prognosis and prognostic factors at 6 months. Stroke. 1999;30:744–8.
Smithard DG, O'Neill PA, England RE, Park CL, Wyatt R, Martin DF, et al. The natural history of dysphagia following a stroke. Dysphagia. 1997;12:188–93.
Flowers HL, Skoretz SA, Silver FL, Rochon E, Fang J, Flamand-Roze C, et al. Poststroke aphasia frequency, recovery, and outcomes: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2016;97:2188–201.e8. https://doi.org/10.1016/j.apmr.2016.03.006.
Sellars C, Bowie L, Bagg J, Sweeney MP, Miller H, Tilston J, et al. Risk factors for chest infection in acute stroke: a prospective cohort study. Stroke. 2007;38:2284–91.
Rofes L, Vilardell N, Clavé P. Post-stroke dysphagia: progress at last. Neurogastroenterol Motil. 2013;25:278–82. https://doi.org/10.1111/nmo.12112.
Clavé P, de Kraa M, Arreola V, Girvent M, Farré R, Palomera E, et al. The effect of bolus viscosity on swallowing function in neurogenic dysphagia. Aliment Pharmacol Ther. 2006;24:1385–94.
Michou E, Mistry S, Jefferson S, Singh S, Rothwell J, Hamdy S. Targeting unlesioned pharyngeal motor cortex improves swallowing in healthy individuals and after dysphagic stroke. Gastroenterology. 2012;142:29–38. https://doi.org/10.1053/j.gastro.2011.09.040.
Teismann IK, Steinstraeter O, Stoeckigt K, Suntrup S, Wollbrink A, Pantev C, et al. Functional oropharyngeal sensory disruption interferes with the cortical control of swallowing. BMC Neurosci. 2007;8.
Ertekin C. Voluntary versus spontaneous swallowing in man. Dysphagia. 2011;26:183–92.
Acknowledgments
We would like to thank Jane Lewis for English editing assistance.
Funding
Financial support received by grant from the Fondo de Investigaciones Sanitarias, Instituto de Salud Carlos III (14/00453); CIBERehd, Instituto de Salud Carlos III; and Programa de Estabilización de Investigadores y de Intensificación de la Actividad Investigadora en el Sistema Nacional de Salud (INT 15/00026).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in the study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 32 kb)
Supplementary Fig. 1
Prevalence of stroke patients with safety and efficacy impairment of swallow using from 5 to 20 ml volumes of the three viscosities (nectar [274.42 ± 13.14 mPa s], liquid [20.40 ± 0.23 mPa s], and pudding [3931.23 ± 166.15 mPa s]) (PNG 89 kb)
Supplementary Fig. 2
Topography of pharyngeal motor hotspots in patients with left (panel a, n = 20) and right (panel b, n = 10) stroke. The vertex is situated in the bottom centre of each panel (0/0) and the hemispheres are represented in the sides (L = left, R = right, the stroke side highlighted in pale gray). Brain mapping of hotspots obtained through transcranial magnetic stimulation (TMS) are represented in a 2 × 2 cms. Graph of x/y coordinates with back circles (contralesional) and white triangles (ipsilesional). The average hotspot for all patients (obtained from calculation of the arithmetic mean of values in both axes) of each hemisphere (contralesional and ipsilesional) is shown in the centre of the “cloud” of individual hotspots with a white circle and black triangle (with vertical and horizontal brackets for standard deviation), respectively (PNG 231 kb)
Rights and permissions
About this article
Cite this article
Cabib, C., Nascimento, W., Rofes, L. et al. Neurophysiological and Biomechanical Evaluation of the Mechanisms Which Impair Safety of Swallow in Chronic Post-stroke Patients. Transl. Stroke Res. 11, 16–28 (2020). https://doi.org/10.1007/s12975-019-00701-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-019-00701-2