Abstract
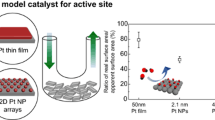
The two-dimensional Pt submonolayers on Au(111) were used as model catalyst system to study kinetics of hydrogen oxidation reaction (HOR). The morphology of different Pt submonolayers was characterized by ex situ scanning tunneling microscopy combined with statistical image analysis. The HOR kinetics data were analyzed using Levich–Koutecky formalism and presented as a function of the mean size of Pt clusters for each Pt submonolayer. The Pt submonolayers with smaller Pt clusters were found less active for HOR. This trend is well correlated with the continuum elasticity analysis of the average active strain in Pt clusters indicating that smaller clusters have less tensile strain. The density functional theory calculations were found in agreement with our results demonstrating that the size-dependent strain in Pt clusters has significant effect on the energy of the d-band center, i.e., the Pt clusters’ activity.






Similar content being viewed by others
References
A.S. Edelstein, R.C. Cammarata (eds.), Nanomaterials: synthesis, properties and applications (IOP Publishing, Bristol, 1996)
G. Cao, Nanostructures and nanomaterials: synthesis, properties and applications (Imperial College Press, London, 2004)
R. Adzic, J. Zhung, K. Sasaki, M.B. Vukmirovic, M. Shao, J.X. Wang, A.U. Nilekar, M. Mavrikakis, J.A. Valerio, F. Uribe, Top Catal 46, 249 (2007)
K. Sasaki, J.X. Wang, H. Naohara, N. Marinkovic, K. More, H. Inada, R.R. Adzic, Electrochim Acta 55, 2645 (2010)
P.A. Dowben, Surf Sci Rep 40, 161 (2000)
J. Greeley, J.K. Nørskov, M. Mavrikakis, Annu Rev Phys Chem 53, 319 (2002)
M. Mavrakakis, B. Hammar, J.K. Nørskov, Phys Rev Lett 81, 2819 (1998)
A. Schlapka, M. Liseihka, A. Gross, U. Kasberger, P. Jakob, Phys Rev Lett 91, 016101 (2003)
E. Kamshoff, E. Hahn, K. Kern, Phys Rev Lett 73, 704 (1994)
M.Ø. Pedersen, S. Helveg, A. Ruban, I. Stensgaard, E. Læsgaard, J.K. Nørskovand, F. Besenbacher, Surf Sci 426, 395 (1999)
M. Mavrikakis, P. Stoltze, J.K. Nørskov, Catal Lett 64, 101 (2000)
P. Strasser, S. Koh, T. Anniyev, J. Greeley, K. More, C. Yu, Z. Liu, S. Kaya, D. Nordlund, H. Ogasawara, M.F. Toney, A. Nilson, Nat Chem 2, 454 (2010)
J. Znahg, M.B. Vukmirovic, Y. Xu, M. Marvikakis, R.R. Adzic, Angew Chem Int Ed 44, 2132 (2005)
L.A. Kibler, A.M. El-Aziz, R. Hoyer, D.M. Kolb, Angew Chem Int Ed 44, 2080 (2005)
J. Greeley, J.K. Norskov, L.A. Kibler, A.M. El-Aziz, D.M. Kolb, Chemphyschem 7, 1032 (2006)
H.-F. Waibel, M. Kleinert, L.A. Kibler, D.M. Kolb, Electrochim Acta 47, 1461 (2002)
S.R. Brankovic, J. McBreen, R.R. Adzic, J Electroanal Chem 503, 99 (2001)
J. Zhang, F.H.B. Lima, M.H. Shao, K. Sasaki, J.X. Wang, J. Hanson, R.R. Adzic, J Phys Chem B 109, 22701 (2005)
Y.D. Jin, Y. Shen, S.J. Dong, J Phys Chem B 108, 8142 (2004)
K. Sasaki, Y. Mo, J.X. Wang, M. Balasubramanian, F. Uribe, J. McBreen, R.R. Adzic, Electrochim Acta 48, 3841 (2003)
R. Kern, P. Muller, Surf Sci 392, 103 (1997)
S.R. Brankovic, N. Dimitrov, K. Sieradzki, Electrochem Solid State Lett 2, 443 (1999)
S.R. Brankovic, J.X. Wang, R.R. Adzic, Surf Sci 474, L173 (2001)
D. Gokcen, S.-E. Bae, S.R. Brankovic, Electrochim Acta 56, 5545 (2011)
D. Gokcen, S.-E. Bae, S.R. Brankovic, J Electrochem Soc 157, D582 (2010)
N. Otsu, IEEE Trans Syst Man Cyber. SMC-9, 62 (1979)
D. Gokcen, S.E. Bae, S.R. Brankovic, Electrochem Solid State Lett (in press). Submitted (results available in supporting material) (2011)
A.J. Bard, L.R. Faulkner, Electrochemical methods (Wiley, New York, 1980)
N.M. Markovic, B.N. Grgur, P.N. Ross, J Phys Chem B 101, 5405 (1997)
R.C. Cammarata, Surf Sci 279, 341 (1992), see also R.C. Cammarata, K. Sieradzki, Annu Rev Mater Sci 24, 215 (1994)
M.C. Payne, D.C. Allan, T. Arias, J.D. Johannopoulus, Rev Mod Phys 64, 1045 (1992)
D. Vanderbilt, Phys Rev B 41, 7892 (1990)
H.J. Monkhorst, J.D. Pack, Phys Rev B 13, 5186 (1976)
B. Hammer, L.B. Hansen, J.K. Nørskov, Phys Rev B 59, 7413 (1999)
K. Uosaki, S. Ye, H. Naohara, Y. Oda, T. Haba, T. Kondo, J Phys Chem B 101, 7566 (1997)
P. Sharma, S. Ganti, N. Bhate, Appl Phys Lett 82, 535 (2003)
J.H. Wassermann, J.S. Vermaak, Surf Sci 22, 164 (1970)
H. Zhang, R.L. Penn, R.J. Hamers, J.F. Banfield, J Phys Chem B 103, 4656 (1999)
J.X. Wang, T.E. Springer, R.R. Adzic, J Electrochem Soc 153, A1732 (2006)
J.X. Wang, T.E. Springer, P. Liu, M. Shao, R.R. Adzic, J Phys Chem C 111, 12425 (2007)
L. Grabow, Y. Xu, M. Mavrikakis, Phys Chem Chem Phys 8, 3369 (2006)
B. Hammer, J.K. Nørskov, Adv Catal 45, 71 (2000)
P. Liu, J.A. Rodriguez, Y. Takahashi, K. Nakamura, J Catal 262, 294 (2009)
Acknowledgment
The authors would like to thank Prof. P. Sharma from University of Houston for the useful discussions regarding strain calculations. DFT calculations are carried out at the Center for Functional Nanomaterials, Brookhaven National Laboratory, which is supported by the U.S. Department of Energy, Office of Basic Energy Sciences, under Contract No. DE-AC02-98CH10886. This material is based upon the work supported by the National Science Foundation under the contract CHE-0955922.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bae, SE., Gokcen, D., Liu, P. et al. Size Effects in Monolayer Catalysis—Model Study: Pt Submonolayers on Au(111). Electrocatalysis 3, 203–210 (2012). https://doi.org/10.1007/s12678-012-0082-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-012-0082-5