Abstract
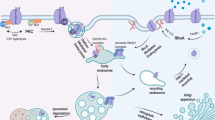
We studied dopamine levels in three compartments of the dopaminergic synapse, including the presynaptic neuron cytosol, dopamine storage vesicles, and the synaptic gap. By considering three transport pathways (dopamine transporter (DAT), vesicular transporter (VT), and exocytosis), four simulated scenarios were investigated: homeostasis, application of cocaine, methamphetamine, and reserpine. Recent experiments show that upon cocaine administration, the Drosophila melanogaster DAT permeation rate constant is decreased by 55% and we adopted this value for the human DAT. Amphetamine and methamphetamine block DAT and VT, while reserpine blocks VT; however, their decreased permeation rate constants are not available. A system of three differential equations of dopamine levels as a function of time was developed respectively for the synaptic compartments and was solved numerically. Per computational inference, the cytosol dopamine concentration was noted to increase in the case of methamphetamine and reserpine, but was practically unchanged in the case of the cocaine administration. Accordingly, our study suggests that amphetamines and other substances that block VT, but not cocaine or substances that only block DAT, may be etiologically important in the cytosolic dopamine mediation of neurodegeneration in Parkinson disease/Parkinsonism.



Similar content being viewed by others
References
Bernstein AI, Stout KA, Miller GW (2014) The vesicular monoamine transporter 2: an underexplored pharmacological target. Neurochem Int 73:89–97. https://doi.org/10.1016/j.neuint.2013.12.003
Best JA, Nijhout HF, Reed MC (2009) Homeostatic mechanisms in dopamine synthesis and release: a mathematical model. Theor Biol Med Model 6:21. https://doi.org/10.1186/1742-4682-6-21
Björklund A, Dunnett SB (2007) Dopamine neuron systems in the brain: an update. Trends Neurosci 30:194–202. https://doi.org/10.1016/j.tins.2007.03.006
Borštnar R, Repič M, Kržan M, Mavri J, Vianello R (2011) Irreversible inhibition of monoamine oxidase B by the antiparkinsonian medicines rasagiline and selegiline: a computational study. Eur J Org Chem 2011:6419–6433. https://doi.org/10.1002/ejoc.201100873
Braak H, Ghebremedhin E, Rüb U, Bratzke H, Del Tredici K (2004) Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res 318:121–134
Brodnik ZD, Ferris MJ, Jones SR, España RA (2017) Reinforcing doses of intravenous cocaine produce only modest dopamine uptake inhibition. ACS Chem Neurosci 8:281–289. https://doi.org/10.1021/acschemneuro.6b00304
Chen L, Ding Y, Cagniard B, Van Laar AD, Mortimer A, Chi W, Hastings TG, Kang UJ, Zhuang X (2008) Unregulated cytosolic dopamine causes neurodegeneration associated with oxidative stress in mice. J Neurosci 28:425–433. https://doi.org/10.1523/JNEUROSCI.3602-07.2008
Chiti F, Dobson CM (2017) Protein misfolding, amyloid formation, and human disease: a summary of progress over the last decade. Annu Rev Biochem 86:27–68. https://doi.org/10.1146/annurev-biochem-061516-045115
Chow MJ, Ambre JJ, Ih Ruo T, Atkinson AJ, Bowsher DJ, Fischman MW (1985) Kinetics of cocaine distribution, elimination, and chronotropic effects. Clin Pharmacol Ther 38:318–324. https://doi.org/10.1038/clpt.1985.179
Christine CW, Garwood ER, Schrock LE, Austin DE, McCulloch CE (2010) Parkinsonism in patients with a history of amphetamine exposure. Mov Disord 25:228–231. https://doi.org/10.1002/mds.22915
Covey DP, Juliano SA, Garris PA (2013) Amphetamine elicits opposing actions on readily releasable and reserve pools for dopamine. PLoS One 8:e60763. https://doi.org/10.1371/journal.pone.0060763
Curtin K, Fleckenstein AE, Robison RJ, Crookston MJ, Smith KR, Hanson GR (2015) Methamphetamine/amphetamine abuse and risk of Parkinson’s disease in Utah: a population-based assessment. Drug Alcohol Depend 146:30–38. https://doi.org/10.1016/j.drugalcdep.2014.10.027
de la Torre R, Farré M, Navarro M, Pacifici R, Zuccaro P, Pichini S (2004) Clinical pharmacokinetics of amfetamine and related substances: monitoring in conventional and non-conventional matrices. Clin Pharmacokinet 43:157–185. https://doi.org/10.2165/00003088-200443030-00002
Dezsi L, Vecsei L (2017) Monoamine oxidase B inhibitors in Parkinson’s disease. CNS Neurol Disord Drug Targets 16:425–439. https://doi.org/10.2174/1871527316666170124165222
Djikic T, Martí Y, Spyrakis F, Lau T, Benedetti P, Davey G, Schloss P, Yelekci K (2018) Human dopamine transporter: the first implementation of a combined in silico/in vitro approach revealing the substrate and inhibitor specificities. J Biomol Struct Dyn:1–16. https://doi.org/10.1080/07391102.2018.1426044
Fuentes P, Paris I, Nassif M, Caviedes P, Segura-Aguilar J (2007) Inhibition of VMAT-2 and DT-diaphorase induce cell death in a substantia nigra-derived cell line-an experimental cell model for dopamine toxicity studies. Chem Res Toxicol 20:776–783. https://doi.org/10.1021/tx600325u
Fumagalli F, Gainetdinov RR, Valenzano KJ, Caron MG (1998) Role of dopamine transporter in methamphetamine-induced neurotoxicity: evidence from mice lacking the transporter. J Neurosci 18:4861–4869. https://doi.org/10.1523/jneurosci.18-13-04861.1998
Fumagalli F, Gainetdinov RR, Wang YM, Valenzano KJ, Miller GW, Caron MG (1999) Increased methamphetamine neurotoxicity in heterozygous vesicular monoamine transporter 2 knock-out mice. J Neurosci 19:2424–2431. https://doi.org/10.1523/jneurosci.19-07-02424.1999
Herrera A, Muñoz P, Steinbusch HWM, Segura-Aguilar J (2017) Are dopamine oxidation metabolites involved in the loss of dopaminergic neurons in the nigrostriatal system in Parkinson’s disease? ACS Chem Neurosci 8:702–711. https://doi.org/10.1021/acschemneuro.7b00034
Jia J, Chen X, Zhu W, Luo Y, Hua Z, Xu Y (2008) CART protects brain from damage through ERK activation in ischemic stroke. Neuropeptides 42:653–661. https://doi.org/10.1016/j.npep.2008.05.006
Kamada T, Chow T, Hiroi T, Imaoka S, Morimoto K, Ohde H, Funae Y (2002) Metabolism of selegiline hydrochloride, a selective monoamine b-type inhibitor, in human liver microsomes. Drug Metab Pharmacokinet 17:199–206
Kenakin T (2009) Pharmacokinetics. In: Kenakin T, A pharmacology primer, 3rd edn. Academic Press, London, pp 179-214
Kuehn BM (2011) Meth use linked to risk of Parkinson disease. JAMA 306:814. https://doi.org/10.1001/jama.2011.1205
Letchworth SR, Sexton T, Childers SR, Vrana KE, Vaughan RA, Davies HM, Porrino LJ (1999) Regulation of rat dopamine transporter mRNA and protein by chronic cocaine administration. J Neurochem 73:1982–1989
Liu Y, Peter D, Roghani A, Schuldiner S, Prive GG, Eisenberg D, Brecha N, Edwards RH (1992) A cDNA that suppresses MPP+ toxicity encodes a vesicular amine transporter. Cell 70:539–551. https://doi.org/10.1016/0092-8674(92)90425-C
Liu C, Kershberg L, Wang J, Schneeberger S, Kaeser PS (2018) Dopamine secretion is mediated by sparse active zone-like release sites. Cell 172:706–718.e15. https://doi.org/10.1016/j.cell.2018.01.008
Lloyd SA, Faherty CJ, Smeyne RJ (2006) Adult and in utero exposure to cocaine alters sensitivity to the Parkinsonian toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Neuroscience 137:905–913. https://doi.org/10.1016/j.neuroscience.2005.09.035
Lohr KM, Stout KA, Dunn AR, Wang M, Salahpour A, Guillot TS, Miller GW (2015) Increased vesicular monoamine transporter 2 (VMAT2; Slc18a2) protects against methamphetamine toxicity. ACS Chem Neurosci 6:790–799. https://doi.org/10.1021/acschemneuro.5b00010
Martorana A, Koch G (2014) Is dopamine involved in Alzheimer’s disease? Front Aging Neurosci 6:252. https://doi.org/10.3389/fnagi.2014.00252
Mavri J, Matute RA, Chu ZT, Vianello R (2016) Path integral simulation of the H/D kinetic isotope effect in monoamine oxidase B catalyzed decomposition of dopamine. J Phys Chem B 120:3488–3492. https://doi.org/10.1021/acs.jpcb.6b00894
Mendelson J, Uemura N, Harris D, Nath RP, Fernandez E, Jacob P, Everhart ET, Jones RT (2006) Human pharmacology of the methamphetamine stereoisomers. Clin Pharmacol Ther 80:403–420. https://doi.org/10.1016/j.clpt.2006.06.013
Muñoz P, Paris I, Sanders LH, Greenamyre JT, Segura-Aguilar J (2012) Overexpression of VMAT-2 and DT-diaphorase protects substantia nigra-derived cells against aminochrome neurotoxicity. Biochim Biophys Acta 1822:1125–1136. https://doi.org/10.1016/j.bbadis.2012.03.010
Paris I, Perez-Pastene C, Cardenas S, Iturriaga-Vasquez P, Iturra P, Muñoz P, Couve E, Caviedes P, Segura-Aguilar J (2010) Aminochrome induces disruption of actin, alpha-, and beta-tubulin cytoskeleton networks in substantia-nigra-derived cell line. Neurotox Res 18:82–92. https://doi.org/10.1007/s12640-009-9148-4
Pavlin M, Mavri J, Repič M, Vianello R (2013) Quantum-chemical approach to determining the high potency of clorgyline as an irreversible acetylenic monoamine oxidase inhibitor. J Neural Transm 120:875–882. https://doi.org/10.1007/s00702-013-1016-y
Pavlin M, Repič M, Vianello R, Mavri J (2016) The chemistry of neurodegeneration: kinetic data and their implications. Mol Neurobiol 53:3400–3415. https://doi.org/10.1007/s12035-015-9284-1
Poberžnik M, Purg M, Repič M, Mavri J, Vianello R (2016) Empirical valence bond simulations of the hydride-transfer step in the monoamine oxidase a catalyzed metabolism of noradrenaline. J Phys Chem B 120:11419–11427. https://doi.org/10.1021/acs.jpcb.6b09011
Pomara C, Cassano T, D’Errico S, Bello S, Romano AD, Riezzo I, Serviddio G (2012) Data available on the extent of cocaine use and dependence: biochemistry, pharmacologic effects and global burden of disease of cocaine abusers. Curr Med Chem 19:5647–5657. https://doi.org/10.2174/092986712803988811
Qi Z, Miller GW, Voit EO (2008) Computational systems analysis of dopamine metabolism. PLoS One 3:e2444. https://doi.org/10.1371/journal.pone.0002444
Repič M, Purg M, Vianello R, Mavri J (2014) Examining electrostatic preorganization in monoamine oxidases A and B by structural comparison and pKa calculations. J Phys Chem B 118:4326–4332. https://doi.org/10.1021/jp500795p
Salomäki MO, Marttila L, Kivelä H, Ouvinen T, Lukkari JO (2018) Effect of pH and oxidant on the first steps of polydopamine formation: a thermodynamic approach. J Phys Chem B acs.jpcb.8b02304. https://doi.org/10.1021/acs.jpcb.8b02304
Schultz W (2007) Multiple dopamine functions at different time courses. Annu Rev Neurosci 30:259–288. https://doi.org/10.1146/annurev.neuro.28.061604.135722
Segura-Aguilar J, Huenchuguala S (2018) Aminochrome induces irreversible mitochondrial dysfunction by inducing autophagy dysfunction in Parkinson’s disease. Front Neurosci 12:106. https://doi.org/10.3389/fnins.2018.00106
Segura-Aguilar J, Paris I, Muñoz P, Ferrari E, Zecca L, Zucca FA (2014) Protective and toxic roles of dopamine in Parkinson’s disease. J Neurochem 129:898–915. https://doi.org/10.1111/jnc.12686
Seiden LS, Sabol KE, Ricaurte GA (1993) Amphetamine: effects on catecholamine systems and behavior. Annu Rev Pharmacol Toxicol 33:639–676. https://doi.org/10.1146/annurev.pa.33.040193.003231
Siciliano CA, Calipari ES, Ferris MJ, Jones SR (2014) Biphasic mechanisms of amphetamine action at the dopamine terminal. J Neurosci 34:5575–5582. https://doi.org/10.1523/JNEUROSCI.4050-13.2014
Umek N, Gersak B, Vintar N, Sostaric M, Mavri J (2018) Dopamine autoxidation is controlled by acidic pH. Front Mol Neurosci 11:467. https://doi.org/10.3389/FNMOL.2018.00467
Vianello R, Repič M, Mavri J (2012) How are biogenic amines metabolized by monoamine oxidases? Eur J Org Chem 2012:7057–7065. https://doi.org/10.1002/ejoc.201201122
Vickrey TL, Xiao N, Venton BJ (2013) Kinetics of the dopamine transporter in Drosophila larva. ACS Chem Neurosci 4:832–837. https://doi.org/10.1021/cn400019q
Watanabe Y, Himeda T, Araki T (2005) Mechanisms of MPTP toxicity and their implications for therapy of Parkinson’s disease. Med Sci Monit 11:RA17–RA23
Yablonsky-Alter E, Agovic MS, Gashi E, Lidsky TI, Friedman E, Banerjee SP (2009) Cocaine challenge enhances release of neuroprotective amino acid taurine in the striatum of chronic cocaine treated rats: a microdialysis study. Brain Res Bull 79:215–218. https://doi.org/10.1016/j.brainresbull.2008.12.014
Yuan J, Liu XD, Han M, Lv RB, Wang YK, Zhang GM, Li Y (2017) Comparison of striatal dopamine transporter levels in chronic heroin-dependent and methamphetamine-dependent subjects. Addict Biol 22:229–234. https://doi.org/10.1111/adb.12271
Zhang S, Wang R, Wang G (2018) Impact of dopamine oxidation on dopaminergic neurodegeneration. ACS Chem Neurosci acschemneuro.8b00454. https://doi.org/10.1021/acschemneuro.8b00454
Acknowledgments
We would like to thank Dr. Chiedozie K. Ugwoke for critical review of the manuscript.
Funding
This work was funded by the Slovenian Research Agency (grant numbers P1-0012, P3-0043).
Author information
Authors and Affiliations
Contributions
DP and DTP—calculations and manuscript writing; RV—study design, literature search, and manuscript writing; NU: study design, calculations, literature search, and manuscript writing; JM—study design, calculations, literature search, and manuscript writing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pregeljc, D., Teodorescu-Perijoc, D., Vianello, R. et al. How Important Is the Use of Cocaine and Amphetamines in the Development of Parkinson Disease? A Computational Study. Neurotox Res 37, 724–731 (2020). https://doi.org/10.1007/s12640-019-00149-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-019-00149-0