Abstract
Purpose of Review
The majority of women diagnosed with early-stage breast cancer will undergo radiotherapy. Clinical decisions regarding the delivery of radiotherapy are typically based on clinical and pathologic factors. In contrast, systemic therapy clinical decisions incorporate specific breast cancer biology. This review will focus on how breast cancer biology may help tailor radiotherapy for early-stage patients.
Recent Findings
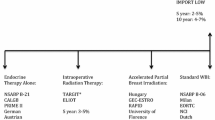
Radiotherapy for early-stage breast cancer is typically delivered for breast conservation. Regional nodal radiotherapy use in early-stage disease is expected to expand in the wake of recent clinical trial publications demonstrating improved disease-free survival in those with one to three axillary nodal metastases. There is a growing body of data demonstrating that intrinsic breast cancer subtypes and luminal multigene assays developed for systemic therapy are also associated with local regional recurrence.
Summary
Including tumor biology in addition to standard clinical and pathological factors may lead to a more tailored use of radiotherapy in early-stage breast cancer.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Siegel R, Miller K, Jemal A. Cancer statistics 2016. CA Cancer J Clin. 2016;66:7–30.
Sareigo J. Regional variation in breast cancer treatment throughout the United States. Am J Surg. 2008;196:572–4.
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A, editors. AJCC cancer staging manual, 7th ed. France: Springer; 2010.
Agarwal S, Pappas L, Neumayer L, et al. Effect of breast conservation therapy vs mastectomy on disease-specific survival for early-stage breast cancer. JAMA Surg. 2014;149(3):267–74.
EBCTCG (Early Breast Cancer Trialists’ Collaborative Group). Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomized trials. Lancet. 2014;383:2127–35.
Poortmans PM, Collette S, Kirkove C, et al. Internal mammary and medial supraclavicular irradiation in breast cancer. N Engl J Med. 2015;374(4):317–27.
Whelan TJ, Olivotto IA, Parulekar WR, et al. Regional nodal irradiation in early-stage breast cancer. N Engl J Med. 2015;373(4):307–16.
Sorlie T, Perou C, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. PNAS. 2001;98(19):10869–74.
Peppercorn J, Perou CM, Carey LA. Molecular subtypes in breast cancer evaluation and management: divide and conquer. Cancer Investig. 2008;26:1–10.
Prat A, Pineda E, Adamo B, et al. Clinical implications of the intrinsic molecular subtypes of breast cancer. Breast. 2015;24:S26–35.
Sineshaw HM, Gaudet M, Ward EM, et al. Association of race/ethnicity, socioeconomic status, and breast cancer subtypes in the National Cancer Data Base (2010–2011). Breast Cancer Res Treat. 2014;145:753–63.
Harris L, Ismaila N, McShane L, et al. Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016;34:1134–50.
Tuttle T, Jarosek S, Habermann E, et al. Omission of radiation therapy after breast-conserving surgery in the United States. Cancer. 2012;118:2004–13.
Clarke M, Collins R, Darby S, et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomized trials. Lancet. 2005;366(9503):2087–106.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10 801 women in 17 randomized trials. Lancet. 2011;378:1707–16.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomized trials. Lancet. 2011;378:771–84.
Pegram MD, Pauletti G, Slamon DJ, et al. HER-2/neu as a predictive marker of response to breast cancer therapy. Breast Cancer Res Treat. 1998;52(1–3):65–77.
Hughes KS, Schnaper LA, Bellon JR, et al. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013;31:2382–7.
Fisher B, Bryant J, Dignam JJ, et al. Tamoxifen, radiation therapy, or both for prevention of ipsilateral breast tumor recurrence after lumpectomy in women with invasive breast cancers of one centimeter or less. J Clin Oncol. 2002;20:4141–9.
Fyles AW, McCready DR, Manchul LA, et al. Tamoxifen with or without breast irradiation in women 50 years of age or older with early breast cancer. N Engl J Med. 2004;351:963–70.
Arvold N, Taghian A, Niemierko A, et al. Age, breast cancer subtype approximation, and local recurrence after breast-conserving therapy. J Clin Oncol. 2010;29:3885–91.
Voduc KD, Cheang MC, Tyldesley S, Gelmon K, Nielsen TO, Kennecke H. Breast cancer subtypes and the risk of local and regional relapse. J Clin Oncol. 2010;28:1684–91.
• Liu F, Shi W, Done S, et al. Identification of a low-risk luminal a breast cancer cohort that may not benefit from breast radiotherapy. J Clin Oncol. 2015;33:2035–40. This manuscript demonstrated the importance of intrinsic subtype reconstructed by IHC and its relation to LRR in clinical trial samples from a population of all hormone-sensitive, node-negative breast cancer that was randomized to receive radiotherapy or observation after lumpectomy. By combining luminal A intrinsic subtype and clinical pathologic factors, they identified a cohort that had very low LRR after lumpectomy without radiotherapy.
Cardoso F, van’t Veer LJ, Bogaerts J, et al. 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med. 2016;375:717–29.
Mamounas EP, Tang G, Fisher B, et al. Association between the 21-gene recurrence score assay and risk of loco regional recurrence in node-negative, estrogen receptor-positive breast cancer: results from NSABP B-14 and NSABP B-20. J Clin Oncol. 2010;28:1677–83.
Solin LJ, Gray R, Goldstein LJ, et al. Prognostic value of biologic subtype and the 21-gene recurrence score relative to local recurrence after breast conservation treatment with radiation for early stage breast carcinoma: results from the Eastern Cooperative Oncology Group E2197 study. Breast Cancer Res Treat. 2012;134:683–92.
Fitzal F, Filipits M, Fesl C, et al. Predicting local recurrence using PAM50 in postmenopausal endocrine responsive breast cancer patients. J Clin Oncol. 2014;32:5s. (suppl; abstr 1008)
Fitzal F, Filipits M, Ruduas M, et al. The genomic expression test EndoPredict is a prognostic tool for identifying risk of local recurrence in postmenopausal endocrine receptor-positive, her2neu-negative breast cancer patients randomized within the prospective ABCSG 8 trial. Brit J Ca. 2015;112:1405–10.
Drukker C, Elias S, Nijenhuis M, et al. Gene expression profiling to predict the risk of locoregional recurrence in breast cancer: a pooled analysis. Breast Cancer Res Treat. 2014;148:599–613.
Kyndi M, Sørensen FB, Knudsen H, et al. Estrogen receptor, progesterone receptor, HER-2, and response to postmastectomy radiotherapy in high-risk breast cancer: the Danish Breast Cancer Cooperative Group. J Clin Oncol. 2008;26:1419–26.
Kim M, Dawood S, Allen P, et al. Hormone receptor status influences the locoregional benefit of trastuzumab in patients with non-metastatic breast cancer. Cancer. 2012;118(20):4936–43.
Wang SL, Li YX, SONG YW, et al. Triple-negative or HER2-positive status predicts higher rates of locoregional recurrence in node-positive breast cancer patients after mastectomy. Int J Radiation Oncology Biol Phys. 2011;80(4):1095–101.
Wu SG, He ZY, Li Q, et al. Predictive value of breast cancer molecular subtypes in Chinese patients with four or more positive nodes after postmastectomy radiotherapy. Breast. 2012;21:657–61.
• Tramm T, Mohammed H, Myhre S, et al. Development and validation of a gene profile predicting benefit of postmastectomy radiotherapy in patients with high-risk breast cancer: a study of gene expression in the DBCG 82bc cohort. Clin Cancer Res. 2014;20(20):5272–80. This manuscript reports on a seven-gene expression profile that was developed and validated from tissue samples collected on the DBCG82bc post-mastectomy clinical trials that are prognostic for LRR and predict radiotherapy benefit. Additional validation in other datasets is warranted for this assay that could potentially become an important clinical tool.
Tramm T, Kyndi M, Myhre S, et al. Relationship between the prognostic and predictive value of the intrinsic subtypes and a validated gene profile predictive of loco-regional control and benefit from post-mastectomy radiotherapy in patients with high-risk breast cancer. Acta Oncol. 2014;53:1337–46.
• Mamounas EP, Liu Q, Paik S, et al. 21-gene recurrence score and locoregional recurrence in node-positive/ER-positive breast cancer treated with chemo-endocrine therapy. JNCI J Natl Cancer Inst. 2017;109(4):djw259. This analysis demonstrated in tissue samples collected on the NSABP B-28 clinical trial that a biological factor like the Oncotype RS assay is prognostic for LRR in node-positive breast cancer treated with chemo-endocrine therapy and without regional nodal radiotherapy. Low LRR was reported in the low-risk or ≤18 RS groups, including those with four or more positive lymph nodes.
Woodward WA, Barlow WE, Jagsi R, et al. The 21-gene recurrence score and locoregional recurrence rates in patients with node-positive breast cancer treated on SWOG S8814. Int J Radiat Oncol Biol Phys. 96(2). S1-S268, e1-e717. 2016.
Cortazar P, Zhang L, Untch M, et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014;384:164–72.
Mamounas EP, Anderson SJ, Dignam JJ, et al. Predictors of loco-regional recurrence following neoadjuvant chemotherapy: results from combined analysis of NSABP B-18 and B-27. J Clin Oncol. 2012;30(32):3960–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Julia White declares that she has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Local-Regional Evaluation and Therapy
Rights and permissions
About this article
Cite this article
White, J. New Approaches for Tailoring the Use of Radiotherapy in Early-Stage Breast Cancer. Curr Breast Cancer Rep 9, 129–136 (2017). https://doi.org/10.1007/s12609-017-0240-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12609-017-0240-0