Abstract
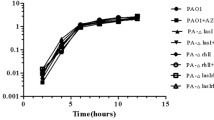
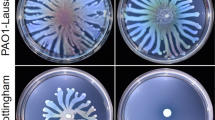
Recent studies have shown that subinhibitory antibiotics play important roles in regulating bacterial genes including virulence factor genes. In this study, the expression of 13 secreted virulence related gene clusters of Pseudomonas aeruginosa, an important opportunistic pathogen, was examined in the presence of subinhibitory concentrations of 4 antibiotics: vancomycin, tetracycline, ampicilin and azithromycin. Activation of gene expression was observed with phzAl, rhlAB, phzA2, lasB, exoY, and exoS. Subinhibitory concentrations of vancomycin resulted in more than 10-fold increase of rhlAB and phzA2 transcription. Both rhamnolipid production and pyocyanin production were significantly elevated, correlating phenotypes with the increased transcription. P. aeruginosa swarming and swimming motility also increased. Similar results were observed with subinhibitory tetracycline, azithromycin and ampicillin. These results indicate that the antibiotics at low concentrations can up-regulate virulence factors and therefore influence bacterial pathogenesis.
Similar content being viewed by others
References
Bagge, N., M. Schuster, M. Hentzer, O. Ciofu, M. Givskov, E.P. Greenberg, and N. Hoiby. 2004. Pseudomonas aeruginosa biofilms exposed to imipenem exhibit changes in global gene expression and beta-lactamase and alginate production. Antimicrob. Agents Chemother. 48, 1175–1187.
Caiazza, N.C., R.M. Shanks, and G.A. O’Toole. 2005. Rhamnolipids modulate swarming motility patterns of Pseudomonas aeruginosa. J. Bacteriol. 187, 7351–7361.
Carfartan, G., P. Gerardin, D. Turck, and M.O. Husson. 2004. Effect of subinhibitory concentrations of azithromycin on adherence of Pseudomonas aeruginosa to bronchial mucins collected from cystic fibrosis patients. J. Antimicrob. Chemother. 53, 686–688.
Chambers, C.E., M.B. Visser, U. Schwab, and P.A. Sokol. 2005. Identification of N-acylhomoserine lactones in mucopurulent respiratory secretions from cystic fibrosis patients. FEMS Microbiol. Lett. 244, 297–304.
Davey, M.E., N.C. Caiazza, and G.A. O’Toole. 2003. Rhamnolipid surfactant production affects biofilm architecture in Pseudomonas aeruginosa PAO1. J. Bacteriol. 185, 1027–1036.
Davies, J. 2006. Are antibiotics naturally antibiotics? J. Ind. Microbiol. Biotechnol. 33, 496–499.
Duan, K., C. Dammel, J. Stein, H. Rabin, and M.G. Surette. 2003. Modulation of Pseudomonas aeruginosa gene expression by host microflora through interspecies communication. Mol. Microbiol. 50, 1477–1491.
Duan, K. and M.G. Surette. 2007. Environmental regulation of Pseudomonas aeruginosa PAO1 Las and Rhl quorum-sensing systems. J. Bacteriol. 189, 4827–4836.
Essar, D.W., L. Eberly, A. Hadero, and I.P. Crawford. 1990. Identification and characterization of genes for a second anthranilate synthase in Pseudomonas aeruginosa: interchangeability of the two anthranilate synthases and evolutionary implications. J. Bacteriol. 172, 884–900.
Fonseca, A.P., C. Extremina, A.F. Fonseca, and J.C. Sousa. 2004. Effect of subinhibitory concentration of piperacillin/tazobactam on Pseudomonas aeruginosa. J. Med. Microbiol. 53, 903–910.
Garske, L.A., S.A. Beatson, A.J. Leech, S.L. Walsh, and S.C. Bell. 2004. Sub-inhibitory concentrations of ceftazidime and tobramycin reduce the quorum sensing signals of Pseudomonas aeruginosa. Pathology 36, 571–575.
Goh, E.B., G. Yim, W. Tsui, J. McClure, M.G. Surette, and J. Davies. 2002. Transcriptional modulation of bacterial gene expression by subinhibitory concentrations of antibiotics. Proc. Natl. Acad. Sci. USA 99, 17025–17030.
Guerra-Santos, L.H., O. Käpeli, and A. Fiechter. 1986. Dependence of Pseudomonas aeruginosa continuous culture biosurfactant production on nutritional and environmental factors. Microbiol. Biotechnol. 24, 443–448.
Hoffman, L.R., D.A. D’Argenio, M.J. MacCoss, Z. Zhang, R.A. Jones, and S.I. Miller. 2005. Aminoglycoside antibiotics induce bacterial biofilm formation. Nature 436, 1171–1175.
Holloway, B.W. 1955. Genetic recombination in Pseudomonas aeruginosa. J. Gen. Microbiol. 13, 572–581.
Ishikawa, J. and T. Horii. 2005. Effects of mupirocin at subinhibitory concentrations on biofilm formation in Pseudomonas aeruginosa. Chemotherapy 51, 361–362.
Kohler, T., L.K. Curty, F. Barja, C. Van Delden, and J.C. Pechere. 2000. Swarming of Pseudomonas aeruginosa is dependent on cell-to-cell signaling and requires flagella and pili. J. Bacteriol. 182, 5990–5996.
Kurachi, M. 1958. Studies on the biosynthesis of pyocyanine. Isolation and determination of pyocyanine. Bull. Inst. Chem. Res. Kyoto Univ. 36, 163–173.
Lau, G.W., D.J. Hassett, H. Ran, and F. Kong. 2004. The role of pyocyanin in Pseudomonas aeruginosa infection. Trends Mol. Med. 10, 599–606.
Linares, J.F., I. Gustafsson, F. Baquero, and J.L. Martinez. 2006. Antibiotics as intermicrobial signaling agents instead of weapons. Proc. Natl. Acad. Sci. USA 103, 19484–19489.
Mavrodi, D.V., R.F. Bonsall, S.M. Delaney, M.J. Soule, G. Phillips, and L.S. Thomashow. 2001. Functional analysis of genes for biosynthesis of pyocyanin and phenazine-1-carboxamide from Pseudomonas aeruginosa PAO1. J. Bacteriol. 183, 6454–6465.
Mukhopadhyay, S., G.E. Staddon, C. Eastman, M. Palmer, E.R. Davies, and F. Carswell. 1994. The quantitative distribution of nebulized antibiotic in the lung in cystic fibrosis. Respir. Med. 88, 203–211.
Nalca, Y., L. Jansch, F. Bredenbruch, R. Geffers, J. Buer, and S. Haussler. 2006. Quorum-sensing antagonistic activities of azithromycin in Pseudomonas aeruginosa PAO1: a global approach. Antimicrob. Agents Chemother. 50, 1680–1688.
Ochsner, U.A., A. Fiechter, and J. Reiser. 1994. Isolation, characterization, and expression in Escherichia coli of the Pseudomonas aeruginosa rhlAB genes encoding a rhamnosyltransferase involved in rhamnolipid biosurfactant synthesis. J. Biol. Chem. 269, 19787–19795.
Ohman, D.E., S.J. Cryz, and B.H. Iglewski. 1980. Isolation and characterization of a Pseudomonas aeruginosa PAO mutant that produces altered elastase. J. Bacteriol. 142, 836–842.
Pearson, J.P., E.C. Pesci, and B.H. Iglewski. 1997. Roles of Pseudomonas aeruginosa las and rhl quorum-sensing systems in control of elastase and rhamnolipid biosynthesis genes. J. Bacteriol. 179, 5756–5767.
Stover, C.K., X.Q. Pham, A.L. Erwin, S.D. Mizoguchi, P. Warrener, M.J. Hickey, F.S. Brinkman, W.O. Hufnagle, D.J. Kowalik, M. Lagrou, R.L. Garber, L. Goltry, E. Tolentino, S. Westbrock-Wadman, Y. Yuan, L.L. Brody, S.N. Coulter, K.R. Folger, A. Kas, K. Larbig, R. Lim, K. Smith, D. Spencer, G.K. Wong, Z. Wu, I.T. Paulsen, J. Reizer, M.H. Saier, R.E. Hancock, S. Lory, and M.V. Olson. 2000. Complete genome sequence of Pseudomonas aeruginosa PA01, an opportunistic pathogen. Nature 406, 959–964.
Surette, M.G. and J. Davies. 2008. A new look at secondary metabolites, bacterial cell-cell Signaling. In S.C. Winans and B.L. Bassler (eds.). American Society of Microbiology, Washington, D.C., USA.
Tateda, K., R. Comte, J.C. Pechere, T. Kohler, K. Yamaguchi, and C. Van Delden. 2001. Azithromycin inhibits quorum sensing in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 45, 1930–1933.
Tateda, K., T.J. Standiford, J.C. Pechere, and K. Yamaguchi. 2004. Regulatory effects of macrolides on bacterial virulence: potential role as quorum-sensing inhibitors. Curr. Pharm. Des. 10, 3055–3065.
Wozniak, D.J. and R. Keyser. 2004. Effects of subinhibitory concentrations of macrolide antibiotics on Pseudomonas aeruginosa. Chest 125, 62–69.
Yim, G., H.H. Wang, and J. Davies. 2007. Antibiotics as signalling molecules. Philos. Trans. R. Soc. Lond. B Biol. Sci. 362, 1195–1200.
Yun, S.H., Y.H. Kim, E.J. Joo, J.S. Choi, J.H. Sohn, and S.I. Kim. 2006. Proteome analysis of cellular response of Pseudomonas putida KT2440 to tetracycline stress. Curr. Microbiol. 53, 95–101.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shen, L., Shi, Y., Zhang, D. et al. Modulation of secreted virulence factor genes by subinhibitory concentrations of antibiotics in Pseudomonas aeruginosa . J Microbiol. 46, 441–447 (2008). https://doi.org/10.1007/s12275-008-0054-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-008-0054-x