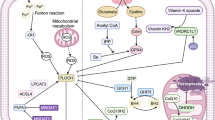
Abstract
Mitochondria are double-membrane organelles involved in the transduction of energy from different metabolic substrates into adenosine triphosphate (ATP) in mammalian cells. The oxidative phosphorylation system is comprised by the activity of the respiratory chain and the complex V (ATP synthase/ATPase). This system is dependent on oxygen gas (O2) in order to maintain a flux of electrons in the respiratory chain, since O2 is the final acceptor of these electrons. Electron leakage from this complex system leads to the continuous generation of reactive species in the cells. The mammalian cells exhibit certain mechanisms to attenuate the consequences originated from the constant exposure to these reactive species. In this context, the transcription factor nuclear factor erythroid 2-related factor 2 (Nrf2) and one of the enzymes whose expression is modulated by Nrf2, heme oxygenase-1 (HO-1), take a central role in inducing cytoprotection in humans. Mitochondrial abnormalities are observed during intoxication and in certain diseases, including neurodegeneration. Mitochondrial protection promoted by natural compounds has attracted the attention of researchers due to the promising effects these agents induce experimentally. In this regard, we examined here whether and how gastrodin (GAS), a phenolic glucoside, would prevent the paraquat (PQ)-induced mitochondrial impairment in the SH-SY5Y cells. The cells were exposed to GAS (25 μM) for 4 h prior to the challenge with PQ at 100 μM for additional 24 h. The silencing of Nrf2 by siRNA or the inhibition of HO-1 by ZnPP IX suppressed the GAS-elicited cytoprotection. Therefore, GAS promoted mitochondrial protection by an Nrf2/HO-1-dependent manner.










Similar content being viewed by others
References
Harris JJ, Jolivet R, Attwell D (2012) Synaptic energy use and supply. Neuron 75:762–777. https://doi.org/10.1016/j.neuron.2012.08.019
Magistretti PJ, Allaman I (2015) A cellular perspective on brain energy metabolism and functional imaging. Neuron 86:883–901. https://doi.org/10.1016/j.neuron.2015.03.035
Brown GC (1992) Control of respiration and ATP synthesis in mammalian mitochondria and cells. Biochem J 284:1–13
Mailloux RJ, Bériault R, Lemire J, Singh R, Chénier DR, Hamel RD, Appanna VD (2007) The tricarboxylic acid cycle, an ancient metabolic network with a novel twist. PLoS One 2:e690
Papa S, Martino PL, Capitanio G, Gaballo A, De Rasmo D, Signorile A, Petruzzella V (2012) The oxidative phosphorylation system in mammalian mitochondria. Adv Exp Med Biol 942:3–37. https://doi.org/10.1007/978-94-007-2869-1_1
Capuano F, Guerrieri F, Papa S (1997) Oxidative phosphorylation enzymes in normal and neoplastic cell growth. J Bioenerg Biomembr 29:379–384
Genova ML, Bianchi C, Lenaz G (2005) Supercomplex organization of the mitochondrial respiratory chain and the role of the Coenzyme Q pool: pathophysiological implications. Biofactors 25:5–20
Solaini G, Sgarbi G, Lenaz G, Baracca A (2007) Evaluating mitochondrial membrane potential in cells. Biosci Rep 27:11–21. https://doi.org/10.1007/s10540-007-9033-4
Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I Kinetics of oxygen utilization. J Biol Chem 217:383–393
Gibson GE, Blass JP, Beal MF, Bunik V (2005) The alpha-ketoglutarate-dehydrogenase complex: a mediator between mitochondria and oxidative stress in neurodegeneration. Mol Neurobiol 31:43–63
Naoi M, Maruyama W, Shamoto-Nagai M, Yi H, Akao Y, Tanaka M (2005) Oxidative stress in mitochondria: decision to survival and death of neurons in neurodegenerative disorders. Mol Neurobiol 31:81–93
Cadenas E (2004) Mitochondrial free radical production and cell signaling. Mol Asp Med 25:17–26. https://doi.org/10.1016/j.mam.2004.02.005
Sies H (2014) Role of metabolic H2O2 generation: redox signaling and oxidative stress. J Biol Chem 289:8735–8741. https://doi.org/10.1074/jbc.R113.544635
Sies H, Berndt C, Jones DP (2017) Oxidative stress. Annu Rev Biochem 86:715–748. https://doi.org/10.1146/annurev-biochem-061516-045037
Atamna H, Frey WH 2nd (2007) Mechanisms of mitochondrial dysfunction and energy deficiency in Alzheimer’s disease. Mitochondrion 7:297–310. https://doi.org/10.1016/j.mito.2007.06.001
Gu Z, Nakamura T, Lipton SA (2010) Redox reactions induced by nitrosative stress mediate protein misfolding and mitochondrial dysfunction in neurodegenerative diseases. Mol Neurobiol 41:55–72. https://doi.org/10.1007/s12035-010-8113-9
Ahmad W (2013) Overlapped metabolic and therapeutic links between Alzheimer and diabetes. Mol Neurobiol 47:399–424. https://doi.org/10.1007/s12035-012-8352-z
de Oliveira MR (2015) Vitamin A and retinoids as mitochondrial toxicants. Oxidative Med Cell Longev 2015:140267–140213. https://doi.org/10.1155/2015/140267
de Oliveira MR (2016) Fluoxetine and the mitochondria: a review of the toxicological aspects. Toxicol Lett 258:185–191. https://doi.org/10.1016/j.toxlet.2016.07.001
de Oliveira MR, Jardim FR (2016) Cocaine and mitochondria-related signaling in the brain: a mechanistic view and future directions. Neurochem Int 92:58–66. https://doi.org/10.1016/j.neuint.2015.12.006
Cadonic C, Sabbir MG, Albensi BC (2016) Mechanisms of mitochondrial dysfunction in Alzheimer’s disease. Mol Neurobiol 53:6078–6090. https://doi.org/10.1007/s12035-015-9515-5
Blajszczak C, Bonini MG (2017) Mitochondria targeting by environmental stressors: implications for redox cellular signaling. Toxicology 391:84–89. https://doi.org/10.1016/j.tox.2017.07.013
Rodríguez-Arribas M, Yakhine-Diop SMS, Pedro JMB, Gómez-Suaga P, Gómez-Sánchez R, Martínez-Chacón G, Fuentes JM, González-Polo RA et al (2017) Mitochondria-associated membranes (MAMs): overview and its role in Parkinson’s disease. Mol Neurobiol 54:6287–6303. https://doi.org/10.1007/s12035-016-0140-8
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295. https://doi.org/10.1074/jbc.R900010200
Nguyen T, Yang CS, Pickett CB (2004) The pathways and molecular mechanisms regulating Nrf2 activation in response to chemical stress. Free Radic Biol Med 37:433–441
Nguyen T, Sherratt PJ, Pickett CB (2003) Regulatory mechanisms controlling gene expression mediated by the antioxidant response element. Annu Rev Pharmacol Toxicol 43:233–260
Li W, Yu S, Liu T, Kim JH, Blank V, Li H, Kong AN (2008) Heterodimerization with small Maf proteins enhances nuclear retention of Nrf2 via masking the NESzip motif. Biochim Biophys Acta 1783:1847–1856. https://doi.org/10.1016/j.bbamcr.2008.05.024
Dinkova-Kostova AT, Holtzclaw WD, Kensler TW (2005) The role of Keap1 in cellular protective responses. Chem Res Toxicol 18:1779–1791. https://doi.org/10.1021/tx050217c
Toroser D, Yarian CS, Orr WC, Sohal RS (2006) Mechanisms of gamma-glutamylcysteine ligase regulation. Biochim Biophys Acta 1760:233–244. https://doi.org/10.1016/j.bbagen.2005.10.010
Arnér ES, Holmgren A (2000) Physiological functions of thioredoxin and thioredoxin reductase. Eur J Biochem 267:6102–6109
Schaffer S, Asseburg H, Kuntz S, Muller WE, Eckert GP (2012) Effects of polyphenols on brain ageing and Alzheimer’s disease: focus on mitochondria. Mol Neurobiol 46:161–178. https://doi.org/10.1007/s12035-012-8282-9
Cuadrado A, Manda G, Hassan A, Alcaraz MJ, Barbas C, Daiber A, Ghezzi P, León R et al (2018) Transcription factor NRF2 as a therapeutic target for chronic diseases: a systems medicine approach. Pharmacol Rev 70:348–383. https://doi.org/10.1124/pr.117.014753
Calabrese V, Cornelius C, Cuzzocrea S, Iavicoli I, Rizzarelli E, Calabrese EJ (2011) Hormesis, cellular stress response and vitagenes as critical determinants in aging and longevity. Mol Asp Med 32:279–304. https://doi.org/10.1016/j.mam.2011.10.007
Baur JA, Sinclair DA (2008) What is xenohormesis? Am J Pharmacol Toxicol 3:152–159. https://doi.org/10.3844/ajptsp.2008.152.159
Calabrese V, Cornelius C, Dinkova-Kostova AT, Calabrese EJ (2009) Vitagenes, cellular stress response, and acetylcarnitine: relevance to hormesis. Biofactors 35:146–160. https://doi.org/10.1002/biof.22
Calabrese V, Cornelius C, Trovato A, Cavallaro M, Mancuso C, Di Rienzo L, Condorelli D, De Lorenzo A et al (2010) The hormetic role of dietary antioxidants in free radical-related diseases. Curr Pharm Des 16:877–883
de Oliveira MR (2016) Phloretin-induced cytoprotective effects on mammalian cells: a mechanistic view and future directions. Biofactors 42:13–40. https://doi.org/10.1002/biof.1256
de Oliveira MR (2016) Evidence for genistein as a mitochondriotropic molecule. Mitochondrion 29:35–44. https://doi.org/10.1016/j.mito.2016.05.005
de Oliveira MR, Nabavi SF, Manayi A, Daglia M, Hajheydari Z, Nabavi SM (2016) Resveratrol and the mitochondria: from triggering the intrinsic apoptotic pathway to inducing mitochondrial biogenesis, a mechanistic view. Biochim Biophys Acta 1860:727–745. https://doi.org/10.1016/j.bbagen.2016.01.017
de Oliveira MR, Nabavi SM, Braidy N, Setzer WN, Ahmed T, Nabavi SF (2016) Quercetin and the mitochondria: a mechanistic view. Biotechnol Adv 34:532–549. https://doi.org/10.1016/j.biotechadv.2015.12.014
de Oliveira MR, Nabavi SF, Habtemariam S, Erdogan Orhan I, Daglia M, Nabavi SM (2015) The effects of baicalein and baicalin on mitochondrial function and dynamics: a review. Pharmacol Res 100:296–308. https://doi.org/10.1016/j.phrs.2015.08.021
Oliveira MR, Nabavi SF, Daglia M, Rastrelli L, Nabavi SM (2016) Epigallocatechin gallate and mitochondria—a story of life and death. Pharmacol Res 104:70–85. https://doi.org/10.1016/j.phrs.2015.12.027
Jardim FR, de Rossi FT, Nascimento MX, da Silva Barros RG, Borges PA, Prescilio IC, de Oliveira MR (2018) Resveratrol and brain mitochondria: a review. Mol Neurobiol 55:2085–2101. https://doi.org/10.1007/s12035-017-0448-z
de Oliveira MR (2018) Carnosic acid as a promising agent in protecting mitochondria of brain cells. Mol Neurobiol (IN PRESS) https://doi.org/10.1007/s12035-017-0842-6
de Oliveira MR, da Costa Ferreira G, Brasil FB, Peres A (2018) Pinocembrin suppresses H2O2-induced mitochondrial dysfunction by a mechanism dependent on the Nrf2/HO-1 axis in SH-SY5Y cells. Mol Neurobiol 55:989–1003. https://doi.org/10.1007/s12035-016-0380-7
Ahmed T, Javed S, Javed S, Tariq A, Šamec D, Tejada S, Nabavi SF, Braidy N et al (2017) Resveratrol and Alzheimer’s disease: mechanistic insights. Mol Neurobiol 54:2622–2635. https://doi.org/10.1007/s12035-016-9839-9
Chung HT, Ryter SW, Kim HP (2013) Heme oxygenase-1 as a novel metabolic player. Oxidative Med Cell Longev 2013:814058–814052. https://doi.org/10.1155/2013/814058
Vanella L, Barbagallo I, Tibullo D, Forte S, Zappalà A, Li Volti G (2016) The non-canonical functions of the heme oxygenases. Oncotarget 7:69075–69086. https://doi.org/10.18632/oncotarget.11923
Ollinger R, Wang H, Yamashita K, Wegiel B, Thomas M, Margreiter R, Bach FH (2007) Therapeutic applications of bilirubin and biliverdin in transplantation. Antioxid Redox Signal 9:2175–2185
Pae HO, Kim EC, Chung HT (2008) Integrative survival response evoked by heme oxygenase-1 and heme metabolites. J Clin Biochem Nutr 42:197–203. https://doi.org/10.3164/jcbn.2008029
Rochette L, Zeller M, Cottin Y, Vergely C (2018) Redox functions of heme oxygenase-1 and biliverdin reductase in diabetes. Trends Endocrinol Metab 29:74–85. https://doi.org/10.1016/j.tem.2017.11.005
Bach FH (2006) Carbon monoxide: from the origin of life to molecular medicine. Trends Mol Med 12:348–350
Lawrence T (2009) The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol 1:a001651. https://doi.org/10.1101/cshperspect.a001651
Hoesel B, Schmid JA (2013) The complexity of NF-κB signaling in inflammation and cancer. Mol Cancer 12:86. https://doi.org/10.1186/1476-4598-12-86
de Oliveira MR, Peres A, Ferreira GC, Schuck PF, Gama CS, Bosco SMD (2017) Carnosic acid protects mitochondria of human neuroblastoma SH-SY5Y cells exposed to Paraquat through activation of the Nrf2/HO-1Axis. Mol Neurobiol 54:5961–5972. https://doi.org/10.1007/s12035-016-0100-3
Yang XD, Zhu J, Yang R, Liu JP, Li L, Zhang HB (2007) Phenolic constituents from the rhizomes of Gastrodia elata. Nat Prod Res 21:180–186. https://doi.org/10.1080/14786410601081997
Liu Y, Gao J, Peng M, Meng H, Ma H, Cai P, Xu Y, Zhao Q et al (2018) A review on central nervous system effects of gastrodin. Front Pharmacol 9:24. https://doi.org/10.3389/fphar.2018.00024
de Oliveira MR, Brasil FB, Fürstenau CR (2018) Evaluation of the mitochondria-related redox and bioenergetics effects of gastrodin in SH-SY5Y cells exposed to hydrogen peroxide. J Mol Neurosci 64:242–251. https://doi.org/10.1007/s12031-018-1027-0
Blanco-Ayala T, Andérica-Romero AC, Pedraza-Chaverri J (2014) New insights into antioxidant strategies against paraquat toxicity. Free Radic Res 48:623–640. https://doi.org/10.3109/10715762.2014.899694
Suntres ZE (2002) Role of antioxidants in paraquat toxicity. Toxicology 180:65–77
Morán JM, Ortiz-Ortiz MA, Ruiz-Mesa LM, Fuentes JM (2010) Nitric oxide in paraquat-mediated toxicity: a review. J Biochem Mol Toxicol 24:402–409. https://doi.org/10.1002/jbt.20348
Robb EL, Gawel JM, Aksentijević D, Cochemé HM, Stewart TS, Shchepinova MM, Qiang H, Prime TA et al (2015) Selective superoxide generation within mitochondria by the targeted redox cycler MitoParaquat. Free Radic Biol Med 89:883–894. https://doi.org/10.1016/j.freeradbiomed.2015.08.021
de Oliveira MR, Ferreira GC, Schuck PF (2016) Protective effect of carnosic acid against paraquat-induced redox impairment and mitochondrial dysfunction in SH-SY5Y cells: role for PI3K/Akt/Nrf2 pathway. Toxicol in Vitro 32:41–54. https://doi.org/10.1016/j.tiv.2015.12.005
de Oliveira MR, Schuck PF, Bosco SMD (2017) Tanshinone I induces mitochondrial protection through an Nrf2-dependent mechanism in Paraquat-treated human neuroblastoma SH-SY5Y cells. Mol Neurobiol 54:4597–4608. https://doi.org/10.1007/s12035-016-0009-x
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
de Oliveira MR, Peres A, Ferreira GC (2017) Pinocembrin attenuates mitochondrial dysfunction in human neuroblastoma SH-SY5Y cells exposed to methylglyoxal: role for the Erk1/2-Nrf2 signaling pathway. Neurochem Res 42:1057–1072. https://doi.org/10.1007/s11064-016-2140-5
de Oliveira MR, Andrade CMB, Fürstenau CR (2018) Naringenin exerts anti-inflammatory effects in Paraquat-treated SH-SY5Y cells through a mechanism associated with the Nrf2/HO-1 axis. Neurochem Res 43:894–903. https://doi.org/10.1007/s11064-018-2495-x
de Oliveira MR, de Souza ICC, Fürstenau CR (2018) Carnosic acid induces anti-inflammatory effects in Paraquat-treated SH-SY5Y cells through a mechanism involving a crosstalk between the Nrf2/HO-1 axis and NF-κB. Mol Neurobiol 55:890–897. https://doi.org/10.1007/s12035-017-0389-6
Wang K, Zhu L, Zhu X, Zhang K, Huang B, Zhang J, Zhang Y, Zhu L et al (2014) Protective effect of paeoniflorin on Aβ25-35-induced SH-SY5Y cell injury by preventing mitochondrial dysfunction. Cell Mol Neurobiol 34:227–234. https://doi.org/10.1007/s10571-013-0006-9
Poderoso JJ, Carreras MC, Lisdero C, Riobó N, Schöpfer F, Boveris A (1996) Nitric oxide inhibits electron transfer and increases superoxide radical production in rat heart mitochondria and submitochondrial particles. Arch Biochem Biophys 328:85–92
LeBel CP, Ischiropoulos H, Bondy SC (1992) Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem Res Toxicol 5:227–231
Quesada A, Ogi J, Schultz J, Handforth A (2011) C-terminal mechano-growth factor induces heme oxygenase-1-mediated neuroprotection of SH-SY5Y cells via the protein kinase Cϵ/Nrf2 pathway. J Neurosci Res 89:394–405. https://doi.org/10.1002/jnr.22543
Jin X, Liu Q, Jia L, Li M, Wang X (2015) Pinocembrin attenuates 6-OHDA-induced neuronal cell death through Nrf2/ARE pathway in SH-SY5Y cells. Cell Mol Neurobiol 35:323–333. https://doi.org/10.1007/s10571-014-0128-8
Jiang G, Hu Y, Liu L, Cai J, Peng C, Li Q (2014) Gastrodin protects against MPP(+)-induced oxidative stress by up regulates heme oxygenase-1 expression through p38 MAPK/Nrf2 pathway in human dopaminergic cells. Neurochem Int 75:79–88. https://doi.org/10.1016/j.neuint.2014.06.003
Wang XL, Xing GH, Hong B, Li XM, Zou Y, Zhang XJ, Dong MX (2014) Gastrodin prevents motor deficits and oxidative stress in the MPTP mouse model of Parkinson's disease: involvement of ERK1/2-Nrf2 signaling pathway. Life Sci 114:77–85. https://doi.org/10.1016/j.lfs.2014.08.004
Peng Z, Wang S, Chen G, Cai M, Liu R, Deng J, Liu J, Zhang T et al (2015) Gastrodin alleviates cerebral ischemic damage in mice by improving anti-oxidant and anti-inflammation activities and inhibiting apoptosis pathway. Neurochem Res 40:661–673. https://doi.org/10.1007/s11064-015-1513-5
Qu LL, Yu B, Li Z, Jiang WX, Jiang JD, Kong WJ (2016) Gastrodin ameliorates oxidative stress and proinflammatory response in nonalcoholic fatty liver disease through the AMPK/Nrf2 pathway. Phytother Res 30:402–411. https://doi.org/10.1002/ptr.5541
Zhang Z, Zhou J, Song D, Sun Y, Liao C, Jiang X (2017) Gastrodin protects against LPS-induced acute lung injury by activating Nrf2 signaling pathway. Oncotarget 8:32147–32156. https://doi.org/10.18632/oncotarget.16740
Picard M, Wallace DC, Burelle Y (2016) The rise of mitochondria in medicine. Mitochondrion 30:105–116. https://doi.org/10.1016/j.mito.2016.07.003
Iqbal K, Alonso AC, Gong CX, Khatoon S, Singh TJ, Grundke-Iqbal I (1994) Mechanism of neurofibrillary degeneration in Alzheimer’s disease. Mol Neurobiol 9:119–123
Elbaz A, Tranchant C (2007) Epidemiologic studies of environmental exposures in Parkinson’s disease. J Neurol Sci 262:37–44
Munoz-Sanjuan I, Bates GP (2011) The importance of integrating basic and clinical research toward the development of new therapies for Huntington disease. J Clin Invest 121:476–483. https://doi.org/10.1172/JCI45364
Schulz JB, Gerlach M, Gille G, Kuhn W, Müngersdorf M, Riederer P, Südmeyer M, Ludolph A (2011) Basic science in Parkinson’s disease: its impact on clinical practice. J Neurol 258:S299–S306. https://doi.org/10.1007/s00415-011-6040-y
Shoshan-Barmatz V, Nahon-Crystal E, Shteinfer-Kuzmine A, Gupta R (2018) VDAC1, mitochondrial dysfunction, and Alzheimer’s disease. Pharmacol Res 131:87–101. https://doi.org/10.1016/j.phrs.2018.03.010
Kourti M, Jiang WG, Cai J (2017) Aspects of carbon monoxide in form of CO-releasing molecules used in cancer treatment: more light on the way. Oxidative Med Cell Longev 2017:9326454. https://doi.org/10.1155/2017/9326454
Magierowska K, Brzozowski T, Magierowski M (2018) Emerging role of carbon monoxide in regulation of cellular pathways and in the maintenance of gastric mucosal integrity. Pharmacol Res 129:56–64. https://doi.org/10.1016/j.phrs.2018.01.008
Battino M, Giampieri F, Pistollato F, Sureda A, de Oliveira MR, Pittalà V, Fallarino F, Nabavi SF et al (2018) Nrf2 as regulator of innate immunity: a molecular Swiss army knife! Biotechnol Adv 36:358–370. https://doi.org/10.1016/j.biotechadv.2017.12.012
de Oliveira MR, Brasil FB, Fürstenau CR (2018) Sulforaphane attenuated the pro-inflammatory state induced by hydrogen peroxide in SH-SY5Y cells through the Nrf2/HO-1 signaling pathway. Neurotox Res (IN PRESS) https://doi.org/10.1007/s12640-018-9881-7
Scalbert A, Morand C, Manach C, Rémésy C (2002) Absorption and metabolism of polyphenols in the gut and impact on health. Biomed Pharmacother 56:276–282
Angelino D, Cossu M, Marti A, Zanoletti M, Chiavaroli L, Brighenti F, Del Rio D, Martini D (2017) Bioaccessibility and bioavailability of phenolic compounds in bread: a review. Food Funct 8:2368–2393. https://doi.org/10.1039/c7fo00574a
Goszcz K, Duthie GG, Stewart D, Leslie SJ, Megson IL (2017) Bioactive polyphenols and cardiovascular disease: chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response? Br J Pharmacol 174:1209–1225. https://doi.org/10.1111/bph.13708
Holmström KM, Baird L, Zhang Y, Hargreaves I, Chalasani A, Land JM, Stanyer L, Yamamoto M et al (2013) Nrf2 impacts cellular bioenergetics by controlling substrate availability for mitochondrial respiration. Biol Open 2:761–770. https://doi.org/10.1242/bio.20134853
Ludtmann MH, Angelova PR, Zhang Y, Abramov AY, Dinkova-Kostova AT (2014) Nrf2 affects the efficiency of mitochondrial fatty acid oxidation. Biochem J 457:415–424. https://doi.org/10.1042/BJ20130863
Hayes JD, Dinkova-Kostova AT (2014) The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem Sci 39:199–218. https://doi.org/10.1016/j.tibs.2014.02.002
Holmström KM, Kostov RV, Dinkova-Kostova AT (2016) The multifaceted role of Nrf2 in mitochondrial function. Curr Opin Toxicol 1:80–91. https://doi.org/10.1016/j.cotox.2016.10.002
Acknowledgements
This work was supported by CNPq (Universal 2016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Figure S1
The effects of gastrodin on the levels of nuclear Nrf2 (A) and total heme oxygenase-1 (HO-1) (B) in SH-SY5Y cells. The cells were exposed to gastrodin at 25 μM for 0–24 h, depending on the parameter analyzed. Data are shown as the mean ± SEM of three or five independent experiments each done in triplicate. One-way ANOVA followed by the post hoc Tukey’s test, * p < 0.05 different from the control group. (PDF 83 kb)
Figure S2
The effect of Nrf2 silencing by siRNA (48 h) on the levels of heme oxygenase-1 (HO-1) in gastrodin-treated SH-SY5Y cells. The cells were exposed to gastrodin at 25 μM for 24 h. Data are shown as the mean ± SEM of three or five independent experiments each done in triplicate. One-way ANOVA followed by the post hoc Tukey’s test, *p < 0.05 different from the control group, ** p < 0.05 different from the gastrodin-treated cells transfected with negative control (NC) siRNA.. (PDF 5 kb)
Rights and permissions
About this article
Cite this article
de Oliveira, M.R., de Bittencourt Brasil, F. & Fürstenau, C.R. Inhibition of the Nrf2/HO-1 Axis Suppresses the Mitochondria-Related Protection Promoted by Gastrodin in Human Neuroblastoma Cells Exposed to Paraquat. Mol Neurobiol 56, 2174–2184 (2019). https://doi.org/10.1007/s12035-018-1222-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1222-6