Abstract
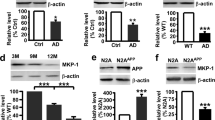
c-Jun N-terminal kinases (JNKs), which belong to a mitogen-activated protein kinase (MAPK) family, are involved in the regulation of several physiological functions in mammals and act as mediators of apoptosis, obesity, and memory storage in the brain, including the processes of neuronal de- and regeneration. JNK subfamily is encoded by three separate but related genes: jnk1, jnk2, and jnk3, giving rise to at least ten distinct splice variants of the JNK proteins. JNK3 is thought to be a major contributor to neurodegeneration in mammalian brain. The role of JNK1 in the pathological processes affecting cognitive function, especially in diseases such as Alzheimer’s disease (AD), is less clear. In order to evaluate the effects of JNK1 deficiency in an experimental model of familial Alzheimer’s disease, double transgenic APPswe/PS1dE9 mice were crossed with the JNK1 heterozygous deficient animals (jnk1+/−). As expected, a ∼50 % reduction in JNK1 protein levels was observed in the hippocampi of 9-month-old APPswe/PS1dE9/jnk1+/− mice, compared with the APPswe/PS1dE9 group. JNK1 deficiency resulted in reduced BACE1 expression, suggesting alterations in amyloidogenic pathway. However, no significant inter-group differences in the total number of β-amyloid plaques were observed in the hippocampal region. In addition, protein levels of PPAR gamma coactivator-1α (PGC-1α), a molecule involved in mitochondrial biogenesis and energy homeostasis, were decreased in 9-month-old APPswe/PS1dE9 mice but not in APPswe/PS1dE9/jnk1+/− animals. Furthermore, JNK1 deficiency did not have an effect on pro-inflammatory marker expression in the hippocampus. Heterozygous deficiency of JNK1 results in the decrease of BACE1 protein levels, which is not accompanied by the reduction in the total number of β-amyloid plaques in the hippocampi of APPswe/PS1dE9 mice. Moreover, PGC-1α expression is restored in APPswe/PS1dE9/jnk1+/− animals, which indicates a possible role of JNK1 in brain mitochondrial regulation. Nevertheless, our results suggest that partial inhibition of JNK1 is not sufficient to prevent the neuropathological processes in this model. It may be necessary to inhibit both the JNK1 and JNK3 simultaneously, especially as previous studies suggest that JNK3 contributes to AD neuropathology.






Similar content being viewed by others
References
Hardy JA, Higgins GA (1992) Alzheimer’s disease: the amyloid cascade hypothesis. Science 256:184–185
Hardy JA, Allsop D (1991) Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol Sci 12:383–388
Karran E, Hardy J (2014) A critique of the drug discovery and phase 3 clinical programs targeting the amyloid hypothesis for Alzheimer disease. Ann Neurol 76:185–205
Drachman DA (2014) The amyloid hypothesis, time to move on: amyloid is the downstream result, not cause, of Alzheimer’s disease. Alzheimers Dement 10:372–380
Swerdlow RH, Khan SM (2004) A “mitochondrial cascade hypothesis” for sporadic Alzheimer’s disease. Med Hypotheses 63:8–20
Swerdlow RH, Burns JM, Khan SM (2010) The Alzheimer’s disease mitochondrial cascade hypothesis. J Alzheimers Dis 20(Suppl 2):S265–S279
Nisbet RM, Polanco JC, Ittner LM, Götz J (2015) Tau aggregation and its interplay with amyloid-β. Acta Neuropathol 129:207–220
Swerdlow RH (2012) Mitochondria and cell bioenergetics: increasingly recognized components and a possible etiologic cause of Alzheimer’s disease. Antioxid Redox Signal 16:1434–1455
Aizenstein HJ, Nebes RD, Saxton JA, Price JC, Mathis CA, Tsopelas ND, Ziolko SK, James JA et al (2008) Frequent amyloid deposition without significant cognitive impairment among the elderly. Arch Neurol 65:1509–1517
Castello MA, Soriano S (2014) On the origin of Alzheimer’s disease. Trials and tribulations of the amyloid hypothesis. Ageing Res Rev 13:10–22
Castello MA, Jeppson JD, Soriano S (2014) Moving beyond anti-amyloid therapy for the prevention and treatment of Alzheimer’s disease. BMC Neurol 14:169
Demetrius LA, Driver J (2013) Alzheimer’s as a metabolic disease. Biogerontology 14:641–649
Kaminsky YG, Tikhonova LA, Kosenko EA (2015) Critical analysis of Alzheimer’s amyloid-beta toxicity to mitochondria. Front Biosci (Landmark Ed) 20:173–171
De Felice FG, Ferreira ST (2014) Inflammation, defective insulin signaling, and mitochondrial dysfunction as common molecular denominators connecting type 2 diabetes to Alzheimer disease. Diabetes 63:2262–2272
de Lemos ML, de la Torre AV, Petrov D, Brox S, Folch J, Pallàs M, Lazarowski A, Beas-Zarate C et al (2013) Evaluation of hypoxia inducible factor expression in inflammatory and neurodegenerative brain models. Int J Biochem Cell Biol 45:1377–88
Di Scala C, Chahinian H, Yahi N, Garmy N, Fantini J (2014) Interaction of Alzheimer’s β-amyloid peptides with cholesterol: mechanistic insights into amyloid pore formation. Biochemistry 53:4489–502
Ramos-Rodriguez JJ, Ortiz-Barajas O, Gamero-Carrasco C, de la Rosa PP, Infante-Garcia C, Zopeque-Garcia N et al (2014) Prediabetes-induced vascular alterations exacerbate central pathology in APPswe/PS1dE9 mice. Psychoneuroendocrinology 48:123–135
Hardy J, Bogdanovic N, Winblad B, Portelius E, Andreasen N, Cedazo-Minguez A, Zetterberg H (2014) Pathways to Alzheimer’s disease. J Intern Med 275:296–303
Bain J, McLauchlan H, Elliott M, Cohen P (2003) The specificities of protein kinase inhibitors: an update. Biochem J 371:199–204
Braithwaite SP, Schmid RS, He DN et al (2010) Inhibition of c-Jun kinase provides neuroprotection in a model of Alzheimer’s disease. Neurobiol Dis 39:311–317
Clarke M, Pentz R, Bobyn J, Hayley S (2012) Stressor-like effects of c-Jun N-terminal kinase (JNK) inhibition. PLoS ONE 7, e44073
Gourmaud S, Paquet C, Dumurgier J, Pace C, Bouras C, Gray F, Laplanche JL, Meurs EF et al (2014) Increased levels of cerebrospinal fluid JNK3 associated with amyloid pathology: links to cognitive decline. J Psychiatry Neurosci 39(6):140062
Sclip A, Tozzi A, Abaza A et al (2014) c-Jun N-terminal kinase has a key role in Alzheimer disease synaptic dysfunction in vivo. Cell Death Dis 5, e1019
de Lemos L, Junyent F, Verdaguer E, Folch J, Romero R, Pallàs M, Ferrer I, Auladell C et al (2010) Differences in activation of ERK1/2 and p38 kinase in Jnk3 null mice following KA treatment. J Neurochem 114:1315–1322
Hunot S, Vila M, Teismann P, Davis RJ, Hirsch EC, Przedborski S, Rakic P, Flavell RA (2004) JNK-mediated induction of cyclooxygenase 2 is required for neurodegeneration in a mouse model of Parkinson’s disease. Proc Natl Acad Sci U S A 101:665–670
Ma QL, Yang F, Rosario ER et al (2009) Beta-amyloid oligomers induce phosphorylation of tau and inactivation of insulin receptor substrate via c-Jun N-terminal kinase signaling: suppression by omega-3 fatty acids and curcumin. J Neurosci 29:9078–9089
Morishima Y, Gotoh Y, Zieg J, Barrett T, Takano H, Flavell R, Davis RJ, Shirasaki Y et al (2001) Beta-amyloid induces neuronal apoptosis via a mechanism that involves the c-Jun N-terminal kinase pathway and the induction of Fas ligand. J Neurosci 21:7551–7560
Okuno S, Saito A, Hayashi T et al (2004) The c-Jun N-terminal protein kinase signaling pathway mediates Bax activation and subsequent neuronal apoptosis through interaction with Bim after transient focal cerebral ischemia. J Neurosci 24:7879–7887
Lee JK, Park J, Lee YD, Lee SH, Han PL (1999) Distinct localization of SAPK isoforms in neurons of adult mouse brain implies multiple signaling modes of SAPK pathway. Brain Res Mol Brain Res 70:116–124
Resnick L, Fennell M (2004) Targeting JNK3 for the treatment of neurodegenerative disorders. Drug Discov Today 9:932–939
Kimberly WT, Zheng JB, Town T, Flavell RA, Selkoe DJ (2005) Physiological regulation of the beta-amyloid precursor protein signaling domain by c-Jun N-terminal kinase JNK3 during neuronal differentiation. J Neurosci 25:5533–5543
Lagalwar S, Guillozet-Bongaarts AL, Berry RW et al (2006) Formation of phospho-SAPK/JNK granules in the hippocampus is an early event in Alzheimer disease. J Neuropathol Exp Neurol 65:455–464
Reineckea K, Herdegena T, Eminela S, Aldenhoff JB, Schiffelholz T (2013) Knockout of c-Jun N-terminal kinases 1, 2 or 3 isoforms induces behavioural changes. Behav Brain Res 245:88–95
Yang DD, Kuan CY, Whitmarsh AJ, Rincon M, Zheng TS, Davis RJ et al (1997) Absence of excitotoxicity-induced apoptosis in the hippocampus of mice lacking the Jnk3 gene. Nature 389:865–70
Qi SH, Liu Y, Hao LY, Guan QH, Gu YH, Zhang J, Yan H, Wang M et al (2010) Neuroprotection of ethanol against ischemia/reperfusion-induced brain injury through decreasing c-Jun N-terminal kinase 3 (JNK3) activation by enhancing GABA release. Neuroscience 167:1125–1137
Wang Q, Walsh DM, Rowan MJ et al (2004) Block of long-term potentiation by naturally secreted and synthetic amyloid beta-peptide in hippocampal slices is mediated via activation of the kinases c-Jun N-terminal kinase, cyclin-dependent kinase 5, and p38 mitogen-activated protein kinase as well as metabotropic glutamate receptor type 5. J Neurosci 24:3370–3378
Sclip A, Antoniou X, Colombo A et al (2011) c-Jun N-terminal kinase regulates soluble Aβ oligomers and cognitive impairment in AD mouse model. J Biol Chem 286:43871–43880
Thakur A, Wang X, Siedlak SL et al (2007) c-Jun phosphorylation in Alzheimer disease. J Neurosci Res 85:1668–1673
Zhou Q, Lam PY, Han D, Cadenas E et al (2008) c-Jun N-terminal kinase regulates mitochondrial bioenergetics by modulating pyruvate dehydrogenase activity in primary cortical neurons. J Neurochem 104:325–335
Hirosumi J, Tuncman G, Chang L, Gorgun CZ, Uysal KT, Maeda K, Karin M, Hotamisligil GS (2002) A central role for JNK in obesity and insulin resistance. Nature 420:333–336
Zhang X, Xu A, Chung SK, Cresser JH, Sweeney G, Wong RL, Lin A, Lam KS (2011) Selective inactivation of c-Jun NH2-terminal kinase in adipose tissue protects against diet-induced obesity and improves insulin sensitivity in both liver and skeletal muscle in mice. Diabetes 60:486–495
Belgardt BF, Mauer J, Brüning JC (2010) Novel roles for JNK1 in metabolism. Aging (Albany NY) 2:621–626
Sabio G, Davis RJ (2010) c-Jun NH2-terminal kinase 1 (JNK1): roles in metabolic regulation of insulin resistance. Trends Biochem Sci 35:490–496
Lee YH, Giraud J, Davis RJ, White MF (2003) c-Jun N-terminal kinase (JNK) mediates feedback inhibition of the insulin signaling cascade. J Biol Chem 278:2896–2902
Werner ED, Lee J, Hansen L, Yuan M, Shoelson SE (2004) Insulin resistance due to phosphorylation of insulin receptor substrate-1 at serine 302. J Biol Chem 279:35298–35305
Waetzig V, Czeloth K, Hidding U et al (2005) c-Jun N-terminal kinases (JNKs) mediate pro-inflammatory actions of microglia. Glia 50:235–246
Wang Y, Zhang Y, Wei Z, Li H, Zhou H, Zhang Z, Zhang Z (2009) JNK inhibitor protects dopaminergic neurons by reducing COX-2 expression in the MPTP mouse model of subacute Parkinson’s disease. J Neurol Sci 285:172–177
Savage MJ, Lin YG, Ciallella JR et al (2002) Activation of c-Jun N-terminal kinase and p38 in an Alzheimer’s disease model is associated with amyloid deposition. J Neurosci 22:3376–3385
Gong B, Pan Y, Vempati P, Zhao W, Knable L, Ho L, Wang J, Sastre M et al (2013) Nicotinamide riboside restores cognition through an upregulation of proliferator-activated receptor-γ coactivator 1α regulated β-secretase 1 degradation and mitochondrial gene expression in Alzheimer’s mouse models. Neurobiol Aging 34:1581–1588
Katsouri L, Parr C, Bogdanovic N, Willem M, Sastre M (2011) PPARγ co-activator-1α (PGC-1α) reduces amyloid-β generation through a PPARγ-dependent mechanism. J Alzheimers Dis 25:151–162
Qin W, Haroutunian KP, Cardozo CP, Ho L, Buxbaum JD, Pasinetti GM (2009) PGC-1alpha expression decreases in the Alzheimer disease brain as a function of dementia. Arch Neurol 66:352–361
Zhou Q, Wang M, Du Y, Zhang W, Bai M, Zhang Z, Li Z, Miao J (2015) Inhibition of c-Jun N-terminal kinase activation reverses Alzheimer disease phenotypes in APPswe/PS1dE9 mice. Ann Neurol 77:637–654
Ríos JA, Cisternas P, Arrese M, Barja S, Inestrosa NC (2014) Is Alzheimer’s disease related to metabolic syndrome? A Wnt signaling conundrum. Prog Neurobiol 121:125–146
Calvo-Ochoa E, Arias C (2015) Cellular and metabolic alterations in the hippocampus caused by insulin signalling dysfunction and its association with cognitive impairment during aging and Alzheimer’s disease: studies in animal models. Diabetes Metab Res Rev 31:1–13
Pedrós I, Petrov D, Allgaier M, Sureda F, Barroso E, Beas-Zarate C, Auladell C, Pallàs M et al (1842) Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer’s disease. Biochim Biophys Acta 2014:1556–1666
Sheng B, Wang X, Su B, Lee HG, Casadesus G, Perry G, Zhu X (2012) Impaired mitochondrial biogenesis contributes to mitochondrial dysfunction in Alzheimer’s disease. J Neurochem 120:419–429
Godoy JA, Rios JA, Zolezzi JM, Braidy N, Inestrosa NC (2014) Signaling pathway cross talk in Alzheimer’s disease. Cell Commun Signal 12:23
Chaturvedi RK, Flint BM (2013) Mitochondrial diseases of the brain. Free Radic Biol Med 63:1–29
Swerdlow RH (2014) Bioenergetic medicine. Br J Pharmacol 171:1854–1869
Vassar R, Kuhn PH, Haass C, Kennedy ME, Rajendran L, Wong PC, Lichtenthaler SF (2014) Function, therapeutic potential and cell biology of BACE proteases: current status and future prospects. J Neurochem 130:4–28
Colombo A, Bastone A, Ploia C et al (2009) JNK regulates APP cleavage and degradation in a model of Alzheimer’s disease. Neurobiol Dis 33:518–525
Yoon SO, Park DJ, Ryu JC, Ozer HG, Tep C, Shin YJ, Lim TH, Pastorino L et al (2012) JNK3 perpetuates metabolic stress induced by Aβ peptides. Neuron 75:824–837
Tamagno E, Guglielmotto M, Aragno M, Borghi R, Autelli R, Giliberto L, Muraca G, Danni O et al (2008) Oxidative stress activates a positive feedback between the gamma- and beta-secretase cleavages of the beta-amyloid precursor protein. J Neurochem 104:683–95
Zhang W, Bai M, Xi Y et al (2012) Multiple inflammatory pathways are involved in the development and progression of cognitive deficits in APPswe/PS1dE9 mice. Neurobiol Aging 33:2661–2677
Colombo A, Repici M, Pesaresi M et al (2007) The TAT-JNK inhibitor peptide interferes with beta amyloid protein stability. Cell Death Differ 14:1845–1848
Guglielmotto M, Monteleone D, Giliberto L, Fornarod M, Borghid R, Tamagnoa E, Tabaton M (2011) Amyloid-β42 activates the expression of BACE1 through the JNK pathway. J Alzheimers Dis 27:871–883
Coffey ET (2014) Nuclear and cytosolic JNK signalling in neurons. Nat Rev Neurosci 15:285–299
Ghasemi R, Zarifkar A, Rastegar K et al (2014) Repeated intra-hippocampal injection of beta-amyloid 25–35 induces a reproducible impairment of learning and memory: considering caspase-3 and MAPKs activity. Eur J Pharmacol 726C:33–40
Pesini A, Iglesias E, Garrido N, Bayona-Bafaluy MP, Montoya J, Ruiz-Pesini E (2014) OXPHOS, pyrimidine nucleotides, and Alzheimer’s disease: a pharmacogenomics approach. J Alzheimers Dis 42:87–96
Acknowledgments
This study was funded by grant 2009/SGR00853 Generalitat de Catalunya (autonomous government of Catalonia) and by grants SAF2011-23631 and SAF2012-39852-C02-01 from the Spanish Ministerio de Ciencia e Innovación.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Jaume Folch and Antoni Camins are senior co-authors.
Rights and permissions
About this article
Cite this article
Petrov, D., Luque, M., Pedrós, I. et al. Evaluation of the Role of JNK1 in the Hippocampus in an Experimental Model of Familial Alzheimer’s Disease. Mol Neurobiol 53, 6183–6193 (2016). https://doi.org/10.1007/s12035-015-9522-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9522-6