Abstract
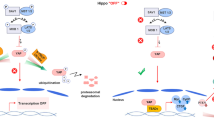
Recognizing proteins that lead to a decreased efficiency of treatment in cancer cells constitutes a main goal for biomedical and biotechnological research and applications. Establishing recombinant cells that overexpress a gene of interest stably is important for treatment studies and drug/compound screening. Survivin is an anti-apoptotic protein which can be a potential candidate for regulating cell death and survival. To investigate the association between survivin increment and apoptosis rate, survivin-reconstituted HEK (HEK-S) cell was developed as in vitro model. RT-PCR and Western blot demonstrated that survivin was constitutively overexpressed in HEK-S cells. Both morphological observation and survival assay showed that HEK-S cells were significantly resistant to apoptotic stimuli. Survivin overexpression led to a decrease in caspase 3/7 activity, whereas YM155 led to a corresponding enhance of caspase activity. ROS level was decreased but ATP content increased in HEK-S cells. Also, HEK-S showed less red- fluorescence and reduced cell proliferation compared to HEK after stimulation. Resistance to laser irradiation was clearly visible as compared with control. Moreover, scratching analysis demonstrated the ability of survivin to cause neighboring cells to increase resistance to drug, whereas YM155 enhanced apoptotic rate and declined invasion in HEK-S cells.






Similar content being viewed by others
References
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A., & Jemal, A. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 68, 394–424.
GBD. (2016). Causes of Death Collaborators (2017) Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet, 390(10100), 1151–1210. https://doi.org/10.1016/S0140-6736(18)32203-7
Deveraux, Q. L., & Reed, J. C. (1999). IAP family proteins-suppressors of apoptosis. Genes & Development, 13, 239–252.
Elmore, S. (2007). Apoptosis: a review of programmed cell death. Toxicologic Pathology, 35, 495–516.
Green, D. R., & Llambi, F. (2015). Cell death signaling. Cold Spring Harbor Perspectives in Biology, 7, a006080.
Chang, H. Y., & Yang, X. (2000). Proteases for cell suicide: functions and regulation of caspases. Microbiology and Molecular Biology Reviews, 64, 821–846.
Altieri, D. C. (2003). Validating survivin as a cancer therapeutic target. Nature Reviews Cancer, 3, 46–54.
Singh, R., Letai, A., & Sarosiek, K. (2019). Regulation of apoptosis in health and disease: the balancing act of BCL-2 family proteins. Nature Reviews Molecular Cell Biology, 20, 175–193.
Tewari, K., & Dhaneshwar, S. (2012). Inhibitors of apoptosis proteins (IAPs): clinical significance in cancer treatment research. J. Cancer Res. Updates, 1, 212–220.
Pfeffer, C. M., & Singh, A. T. K. (2018). Apoptosis: a target for anticancer therapy. International Journal of Molecular Sciences, 19, 448. https://doi.org/10.3390/ijms19020448
Altieri, D. C. (2003). Survivin and apoptosis control. Advances in Cancer Research, 88, 31–52.
Altieri, D. C. (2003). Survivin, versatile modulation of cell division and apoptosis in cancer. Oncogene, 22, 8581–8589.
Altieri, D. C. (2015). Survivin-the inconvenient IAP. Seminars in Cell & Developmental Biology, 39, 91–96.
Johnson, M. E., & Howerth, E. W. (2004). Survivin: a bifunctional inhibitor of apoptosis protein. Veterinary Pathology, 41, 599–607.
Tanaka, K., Iwamoto, S., Gon, G., Nohara, T., Iwamoto, M., & Tanigawa, N. (2000). Expression of survivin and its relationship to loss of apoptosis in breast carcinomas. Clinical Cancer Research, 6, 127–134.
Yamamoto, T., & Tanigawa, N. (2001). The role of survivin as a new target of diagnosis and treatment in human cancer. Medical Electron Microscopy, 34, 207–212.
Dohi, T., Beltrami, E., Wall, N. R., Plescia, J., & Altieri, D. C. (2004). Mitochondrial survivin inhibits apoptosis and promotes tumorigenesis. The Journal of Clinical Investigation, 114, 1117–1127.
Garg, H., Suri, P., Gupta, J. C., Talwar, G. P., & Dubey, S. (2016). Survivin: a unique target for tumor therapy. Cancer Cell International, 16, 49.
Amroudie, M. N., & Ataei, F. (2019). Experimental and theoretical study of IBC domain from human IP3R2; molecular cloning, bacterial expression and protein purification. International Journal of Biological Macromolecules, 124, 1321–1327.
Ataei, F., Torkzadeh-Mahani, M., & Hosseinkhani, S. (2013). A novel luminescent biosensor for rapid monitoring of IP3 by split-luciferase complementary assay. Biosen. Bioelectron., 41, 642–648.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
El-Moatassim, C., Dornand, J., & Mani, J. C. (1992). Extracellular ATP and cell signalling. Biochimica et Biophysica Acta Molecular Cell Research, 1134, 31–45. https://doi.org/10.1016/0167-4889(92)90025-7
Zamaraeva, M. V., Sabirov, R. Z., Maeno, E., Ando-Akatsuka, Y., Bessonova, S. V., & Okada, Y. (2005). Cells die with increased cytosolic ATP during apoptosis: a bioluminescence study with intracellular luciferase. Cell Death and Differentiation, 12, 1390–1397.
Karimzadeh, S., Hosseinkhani, S., Fathi, A., Ataei, F., & Baharvand, H. (2018). Insufficient Apaf-1 expression in early stages of neural differentiation of human embryonic stem cells might protect them from apoptosis. European Journal of Cell Biology, 97, 126–135.
Patel, R. Rinker, L. Peng, J. & Chilian, W.M. (2018) Reactive Oxygen Species: The Good and the Bad In: React. Oxyg. Species Living Cells. InTech. DOI: https://doi.org/10.5772/intechopen.71547.
Bown, S. G. (1983). Phototherapy of tumors. World Journal of Surgery, 7, 700–709.
Abazari, R., Mahjoub, A. R., Ataei, F., Morsali, A., Carpenter-Warren, C. L., Mehdizadeh, K., & Slawin, A. M. Z. (2018). Chitosan immobilization on Bio-MOF nanostructures: a biocompatible pH-responsive nanocarrier for doxorubicin release on MCF-7 cell lines of human breast cancer. Inorganic Chemistry, 57, 13364–13379.
Liang, C. C., Park, A. Y., & Guan, J. L. (2007). In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nature Protocols, 2, 329–333.
Iwasa, T., Okamoto, I., Suzuki, M., Nakahara, T., Yamanaka, K., Hatashita, E., Yamada, Y., Fukuoka, M., Ono, K., & Nakagawa, K. (2008). Radiosensitizing effect of YM155, a novel small-molecule survivin suppressant, in non-small cell lung cancer cell lines. Clinical Cancer Research, 14, 6496–6504.
Cheng, S. M., Chang, Y. C., Liu, C. Y., Lee, J. Y. C., Chan, H. H., Kuo, C. W., Lin, K. Y., Tsai, S. L., Chen, S. H., Li, C. F., Leung, E., Kanwar, J. R., Huang, C. C., Chang, J. Y., & Cheung, C. H. A. (2015). YM155 down-regulates survivin and XIAP, modulates autophagy and induces autophagy-dependent DNA damage in breast cancer cells. British Journal of Pharmacology, 172, 214–234.
Mali, S. (2013). Delivery systems for gene therapy. Indian J. Hum. Genet., 19, 3–8.
Mehdizadeh, K., Ataei, F., & Hosseinkhani, S. (2020). Effects of doxorubicin and docetaxel on susceptibility to apoptosis in high expression level of survivin in HEK and HEK-S cell lines as in vitro models. Biochemical and Biophysical Research Communications, 532, 139–144.
Peng, Q., Juzeniene, A., Chen, J., Svaasand, L. O., Warloe, T., Giercksky, K. E., & Moan, J. (2008). Lasers in medicine. Reports on Progress in Physics, 71(5), 056701.
Singh, N., Krishnakumar, S., Kanwar, R. K., Cheung, C. H. A., & Kanwar, J. R. (2015). Clinical aspects for survivin: a crucial molecule for targeting drug-resistant cancers. Drug Discovery Today, 20, 578–587.
Rauch, A., Hennig, D., Schäfer, C., Wirth, M., Marx, C., Heinzel, T., Schneider, G., & Krämer, O. H. (2014). Survivin and YM155 How faithful is the liaison ? Biochimica et Biophysica Acta(BBA) Reviews on Cancer, 1845, 202–220. https://doi.org/10.1016/j.bbcan.2014.01.003
Acknowledgements
This work was supported by the research council of Tarbiat Modares University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Houshdarpour, R., Ataei, F. & Hosseinkhani, S. Efficient Stable Cell Line Generation of Survivin as an In Vitro Model for Specific Functional Analysis in Apoptosis and Drug Screening. Mol Biotechnol 63, 515–524 (2021). https://doi.org/10.1007/s12033-021-00313-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-021-00313-y