Abstract
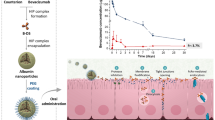
Among the possible delivery routes, the oral administration of a protein is simple and achieves high patient compliance without pain. However, the low bioavailability of a protein drug in the intestine due to the physical barriers of the intestinal epithelia is the most critical problem that needs to be solved. To overcome the low bioavailability of a protein drug in the intestine, we aimed to construct a recombinant Pichia pastoris expressing a human growth hormone (hGH) fusion protein conjugated with a transcytotic peptide (TP) that was screened through peroral phage display to target goblet cells in the intestinal epithelia. The TP-conjugated hGH was successfully produced in P. pastoris in a secreted form at concentrations of up to 0.79 g/l. The function of the TP-conjugated hGH was validated by in vitro and in vivo assays. The transcytotic function of the TP through the intestinal epithelia was verified only in the C terminus conjugated hGH, which demonstrated the induction of IGF-1 in a HepG2 cell culture assay, a higher translocation of recombinant hGH into the ileal villi after oral administration in rats and both IGF-1 induction and higher body weight gain in rats after oral administration. The present study introduces the possibility for the development of an effective oral protein delivery system in the pharmaceutical and animal industries through the introduction of an effective TP into hGH.





Similar content being viewed by others
References
Frokjaer, S., & Otzen, D. E. (2005). Protein drug stability: A formulation challenge. Nature Review Drug Discovery, 4, 298–306.
Marx, V. (2005). Watching peptide drugs grow up. Chemical & Engineering News, 83, 17–24.
Aungst, B. J. (2012). Absorption enhancers: Applications and advances. AAPS Journal, 14, 10–18.
Maculotti, K., Tira, E. M., Sonaggere, M., Perugini, P., Conti, B., Modena, T., & Pavanetto, F. (2009). In vitro evaluation of chondroitin sulphate-chitosan microspheres as carrier for the delivery of proteins. Journal of Microencapsulation, 26, 535–543.
Yun, Y., Cho, Y. W., & Park, K. (2013). Nanoparticles for oral delivery: Targeted nanoparticles with peptidic ligands for oral protein delivery. Advanced Drug Delivery Reviews, 65, 822–832.
Kumar, P., & Mishra, B. (2008). Colon targeted drug delivery systems—an overview. Current Drug Delivery, 5, 186–198.
Maroni, A., Zema, L., Del Curto, M. D., Foppoli, A., & Gazzaniga, A. (2012). Oral colon delivery of insulin with the aid of functional adjuvants. Advanced Drug Delivery Reviews, 64, 540–556.
Zhang, L., & Bulaj, G. (2012). Converting peptides into drug leads by lipidation. Current Medicinal Chemistry, 19, 1602–1618.
Hsieh, P.-W., Hung, C.-F., & Fang, J.-Y. (2009). Current prodrug design for drug discovery. Current Pharmaceutical Design, 15, 2236–2250.
Jana, S., Mandlekar, S., & Marathe, P. (2010). Prodrug design to improve pharmacokinetic and drug delivery properties: Challenges to the discovery scientists. Current Medicinal Chemistry, 17, 3874–3908.
Koren, E., & Torchilin, V. P. (2012). Cell-penetrating peptides: Breaking through to the other side. Trends in Molecular Medicine, 18, 385–393.
Dutt Vadlapudi, A., Krishna Vadlapatla, R., & Mitra, A. K. (2012). Sodium dependent multivitamin transporter (SMVT): A potential target for drug delivery. Current Drug Targets, 13, 994–1003.
Bai, Y., Ann, D. K., & Shen, W.-C. (2005). Recombinant granulocyte colony-stimulating factor-transferrin fusion protein as an oral myelopoietic agent. Proceedings of the National Academy of Sciences United States of America, 102, 7292–7296.
Lim, C.-J., & Shen, W.-C. (2005). Comparison of monomeric and oligomeric transferrin as potential carrier in oral delivery of protein drugs. Journal of Controlled Release, 106, 273–286.
Russell-Jones, G. (1996). The potential use of receptor-mediated endocytosis for oral drug delivery. Advanced Drug Delivery Reviews, 20, 83–97.
Swaan, P. W. (1998). Recent advances in intestinal macromolecular drug delivery via receptor-mediated transport pathways. Pharmaceutical Research, 15, 826–834.
Higgins, D. R., & Cregg, J. M. (1998). Methods in molecular biology: Pichia protocols. Introduction to Pichia pastoris (pp. 1–15). New York: Humana Press.
Cregg, J. M., Cereghino, J. L., Shi, J., & Higgins, D. R. (2000). Recombinant protein expression in Pichia pastoris. Molecular Biotechnology, 16, 23–52.
Kang, S. K., Woo, J. H., Kim, M. K., Woo, S. S., Choi, J. H., Lee, H. G., et al. (2008). Identification of a peptide sequence that improves transport of macromolecules across the intestinal mucosal barrier targeting goblet cells. Journal of Biotechnology, 135, 210–216.
Thaker, V., Haagensen, A. L., Carter, B., Fedorowicz, Z., & Houston, B. W. (2013). Recombinant growth hormone therapy for cystic fibrosis in children and young adults. Cochrane Database Systematic Reviews, 6, CD008901.
Stephany, R. W. (2010). Hormonal growth promoting agents in food producing animals, Doping in sports: Biochemical principles, effects and analysis (pp. 355–367). New York: Springer.
Sousa, C. M. F., & Passarinha, L. (2013). REVIEW: Pichia pastoris: A recombinant microfactory for antibodies and human membrane proteins. Journal of Microbiology and Biotechnology, 23, 587–601.
Lee, C. H., Woo, J. H., Cho, K. K., Kang, S. H., Kang, S. K., & Choi, Y. J. (2007). Expression and characterization of human growth hormone–Fc fusion proteins for transcytosis induction. Biotechnology and Applied Biochemistry, 46, 211–217.
Andus, T., Bauer, J., & Gerok, W. (1991). Effects of cytokines on the liver. Hepatology, 13, 364–375.
Chen, W. Y., Chen, N., Yun, J., Wagner, T. E., & Kopchick, J. J. (1994). In vitro and in vivo studies of antagonistic effects of human growth hormone analogs. Journal of Biological Chemistry, 269, 15892–15897.
Chaiken, I. M., & Williams, W. V. (1996). Identifying structure-function relationships in four-helix bundle cytokines: Towards de novo mimetics design. Trends in Biotechnology, 14, 369–375.
Kirkitadze, M. D., & Barlow, P. N. (2001). Structure and flexibility of the multiple domain proteins that regulate complement activation. Immunological Reviews, 180, 146–161.
Wright, P. E., & Dyson, H. J. (1999). Intrinsically unstructured proteins: Re-assessing the protein structure-function paradigm. Journal of Molecular Biology, 293, 321–331.
Jin, Y., Song, Y., Zhu, X., Zhou, D., Chen, C., Zhang, Z., & Huang, Y. (2012). Goblet cell-targeting nanoparticles for oral insulin delivery and the influence of mucus on insulin transport. Biomaterials, 33, 1573–1582.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MEST) (No. 2011-0029416). We also acknowledge the National Instrumental Centre for Environmental Management (NICEM) and National Center for Inter-University Research Facilities (NCIRF). Jun-Yeong Lee was supported by BK21 program.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jun-Yeong Lee and Sang-Kee Kang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lee, JY., Kang, SK., Li, HS. et al. Production of Recombinant Human Growth Hormone Conjugated with a Transcytotic Peptide in Pichia pastoris for Effective Oral Protein Delivery. Mol Biotechnol 57, 430–438 (2015). https://doi.org/10.1007/s12033-014-9835-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-014-9835-0