Abstract
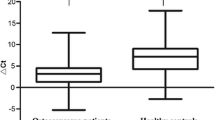
Medulloblastoma is a malignant tumor with high incidence and poor prognosis in adolescents and children. MicroRNA-137 (miR-137) has been found to be abnormally expressed in cancers such as pancreatic cancer. The purpose of this study is to explore the expression of miR-137 in MB and its role in cell physiological activities to determine the significance of miR-137 in the prognosis of MB. First, the expression of miR-137 in MB tissues and cell lines was analyzed by qRT-PCR. Then the Kaplan–Meier survival curve was used to analyze the significance of miR-137 expression in the prognosis, and the Cox regression model was used to explore the correlation between miR-137 expression and clinical characteristics. The effects of miR-137 on MB cell activities were analyzed by MTT assay, Transwell assays, and flow cytometry. It can be concluded from the results that the expression of miR-137 is down-regulated in MB tissues and cells. The down-regulation of miR-137 was significantly related to the poor prognosis of MB, and significantly related to clinical indicators. Up-regulated miR-137 inhibited cell proliferation, migration, invasion, and cell cycle progression, as well as induced cell apoptosis by targeting KDM1A. This study can conclude that miR-137 may be used as a prognostic biomarker of MB.





Similar content being viewed by others

Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Bernardo, B. C., Ooi, J. Y., Lin, R. C., & McMullen, J. R. (2015). miRNA therapeutics: A new class of drugs with potential therapeutic applications in the heart. Future Medicinal Chemistry, 7(13), 1771–1792. https://doi.org/10.4155/fmc.15.107
Bi, W. P., Xia, M., & Wang, X. J. (2018). miR-137 suppresses proliferation, migration and invasion of colon cancer cell lines by targeting TCF4. Oncology Letters, 15(6), 8744–8748. https://doi.org/10.3892/ol.2018.8364
Collange, N. Z., Brito Sde, A., Campos, R. R., Santos, E. A., & Botelho, R. V. (2016). Treatment of medulloblastoma in children and adolescents. Revista Da Associação Médica Brasileira, 62(4), 297. https://doi.org/10.1590/1806-9282.62.04.297
Ding, X., Zhang, J., Feng, Z., Tang, Q., & Zhou, X. (2020). MiR-137–3p Inhibits colorectal cancer cell migration by regulating a KDM1A-dependent epithelial-mesenchymal transition. Digestive Diseases and Sciences. https://doi.org/10.1007/s10620-020-06518-6
Ding, F., Zhang, S., Gao, S., Shang, J., Li, Y., Cui, N., & Zhao, Q. (2018). MiR-137 functions as a tumor suppressor in pancreatic cancer by targeting MRGBP. Journal of Cellular Biochemistry, 119(6), 4799–4807. https://doi.org/10.1002/jcb.26676
Du, Y., Chen, Y., Wang, F., & Gu, L. (2016). miR-137 plays tumor suppressor roles in gastric cancer cell lines by targeting KLF12 and MYO1C. Tumour Biology, 37(10), 13557–13569. https://doi.org/10.1007/s13277-016-5199-3
Duan, J., Lu, G., Li, Y., Zhou, S., Zhou, D., & Tao, H. (2019). miR-137 functions as a tumor suppressor gene in pituitary adenoma by targeting AKT2. International Journal of Clinical and Experimental Pathology, 12(5), 1557–1564.
Feng, Q., Wu, Q., Liu, X., Xiong, Y., & Li, H. (2017). MicroRNA-137 acts as a tumor suppressor in osteosarcoma by targeting enhancer of zeste homolog 2. Experimental and Therapeutic Medicine, 13(6), 3167–3174. https://doi.org/10.3892/etm.2017.4435
Ferrante, M., & Conti, G. O. (2017). Environment and neurodegenerative diseases: An update on miRNA role. Microrna, 6(3), 157–165. https://doi.org/10.2174/2211536606666170811151503
Fu, J. H., Yang, S., Nan, C. J., Zhou, C. C., & Mu, H. Q. (2018). MiR-182 affects renal cancer cell proliferation, apoptosis, and invasion by regulating PI3K/AKT/mTOR signaling pathway. Europian Review for Medical and Pharmacological Sciences, 22(2), 351–357.
Huang, Y. (2019). MiR-137 inhibits cell proliferation in acute lymphoblastic leukemia by targeting JARID1B. Digestive Diseases and Sciences, 103(3), 215–224. https://doi.org/10.1007/s10620-020-06518-6
Khatua, S., Song, A., Citla Sridhar, D., & Mack, S. C. (2018). Childhood medulloblastoma: Current therapies, emerging molecular landscape and newer therapeutic insights. Current Neuropharmacology, 16(7), 1045–1058. https://doi.org/10.2174/1570159x15666171129111324
Koschmann, C., Bloom, K., Upadhyaya, S., Geyer, J. R., & Leary, S. E. (2016). Survival after relapse of medulloblastoma. Journal of Pediatric Hematology/oncology, 38(4), 269–273. https://doi.org/10.1097/mph.0000000000000547
Kumar, V., Kumar, V., Chaudhary, A. K., Coulter, D. W., McGuire, T., & Mahato, R. I. (2018). Impact of miRNA-mRNA profiling and their correlation on medulloblastoma tumorigenesis. Molecular Therapy, 12, 490–503. https://doi.org/10.1016/j.omtn.2018.06.004
Kumar, V., Kumar, V., McGuire, T., Coulter, D. W., Sharp, J. G., & Mahato, R. I. (2017). Challenges and recent advances in medulloblastoma therapy. Trends in Pharmacological Sciences, 38(12), 1061–1084. https://doi.org/10.1016/j.tips.2017.09.002
Lee, S. J., Jeong, J. H., Kang, S. H., Kang, J., Kim, E. A., Lee, J., Jung, H. J., Park, H. Y., & Chae, Y. S. (2019). MicroRNA-137 inhibits cancer progression by targeting Del-1 in triple-negative breast cancer cells. International Journal of Molecular Sciences, 20(24), 6162. https://doi.org/10.3390/ijms20246162
Li, B., Shen, M., Yao, H., Chen, X., & Xiao, Z. (2019). Long noncoding RNA TP73-AS1 modulates medulloblastoma progression in vitro and in vivo by sponging miR-494-3p and targeting EIF5A2. Oncotargets and Therapy, 12, 9873–9885. https://doi.org/10.2147/ott.s228305
Li, K. K., Pang, J. C., Lau, K. M., Zhou, L., Mao, Y., Wang, Y., Poon, W. S., & Ng, H. K. (2013). MiR-383 is downregulated in medulloblastoma and targets peroxiredoxin 3 (PRDX3). Brain Pathology, 23(4), 413–425. https://doi.org/10.1111/bpa.12014
Lodewijk, L., Prins, A. M., Kist, J. W., Valk, G. D., Kranenburg, O., Rinkes, I. H., & Vriens, M. R. (2012). The value of miRNA in diagnosing thyroid cancer: A systematic review. Cancer Biomarkers, 11(6), 229–238. https://doi.org/10.3233/cbm-2012-0273
Lu, T. X., & Rothenberg, M. E. (2018). MicroRNA. Journal of Allergy and Clinical Immunology, 141(4), 1202–1207. https://doi.org/10.1016/j.jaci.2017.08.034
Majello, B., Gorini, F., Saccà, C. D., & Amente, S. (2019). Expanding the role of the histone lysine-specific demethylase LSD1 in cancer. Cancers, 11(3), 324. https://doi.org/10.3390/cancers11030324
Nör, C., & Ramaswamy, V. (2018). Clinical and pre-clinical utility of genomics in medulloblastoma. Expert Review of Neurotherapeutics, 18(8), 633–647. https://doi.org/10.1080/14737175.2018.1503536
Pajtler, K. W., Weingarten, C., Thor, T., Künkele, A., Heukamp, L. C., Büttner, R., Suzuki, T., Miyata, N., Grotzer, M., Rieb, A., Sprüssel, A., Eggert, A., Schramm, A., & Schulte, J. H. (2013). The KDM1A histone demethylase is a promising new target for the epigenetic therapy of medulloblastoma. Acta Neuropathologica Communications, 1, 19. https://doi.org/10.1186/2051-5960-1-19
Pezuk, J. A., Brassesco, M. S., de Oliveira, R. S., Machado, H. R., Neder, L., Scrideli, C. A., & Tone, L. G. (2017). PLK1-associated microRNAs are correlated with pediatric medulloblastoma prognosis. Childs Nervous System, 33(4), 609–615. https://doi.org/10.1007/s00381-017-3366-5
Quinlan, A., & Rizzolo, D. (2017). Understanding medulloblastoma. JAAPA, 30(10), 30–36. https://doi.org/10.1097/01.jaa.0000524717.71084.50
Senfter, D., Samadaei, M., Mader, R. M., Gojo, J., Peyrl, A., Krupitza, G., Kool, M., Sill, M., Haberler, C., Ricken, G., Czech, T., Slavc, I., & Madlener, S. (2019). High impact of miRNA-4521 on FOXM1 expression in medulloblastoma. Cell Death & Disease, 10(10), 696. https://doi.org/10.1038/s41419-019-1926-1
Thompson, E. M., Hielscher, T., Bouffet, E., Remke, M., Luu, B., Gururangan, S., McLendon, R. E., Bigner, D. D., Lipp, E. S., Perreault, S., & Cho, Y. J. (2016). Prognostic value of medulloblastoma extent of resection after accounting for molecular subgroup: a retrospective integrated clinical and molecular analysis. The Lancet Oncology, 17(4), 484–495. https://doi.org/10.1016/s1470-2045(15)00581-1
Wang, D., Lu, G., Shao, Y., & Xu, D. (2018). MiR-182 promotes prostate cancer progression through activating Wnt/β-catenin signal pathway. Biomedicine & Pharmacotherapy, 99, 334.
Wang, Y., Zhang, Q., Guo, B., Feng, J., & Zhao, D. (2020). miR-1231 is downregulated in prostate cancer with prognostic and functional implications. Oncology Research and Treatment, 43(3), 78–86. https://doi.org/10.1159/000504606
Wei, Q., Zhao, L., Jiang, L., Bi, J., Yu, Z., Zhao, L., Song, X., Sun, M., Chen, Y., & Wei, M. (2019). Prognostic relevance of miR-137 and its liver microenvironment regulatory target gene AFM in hepatocellular carcinoma. Journal of Cellular Physiology, 234(7), 11888–11899. https://doi.org/10.1002/jcp.27855
Yang, G. J., Lei, P. M., Wong, S. Y., Ma, D. L., & Leung, C. H. (2018). Pharmacological inhibition of LSD1 for cancer treatment. Molecules, 23(12), 3194. https://doi.org/10.3390/molecules23123194
Yang, B., Dai, J. X., Pan, Y. B., Ma, Y. B., & Chu, S. H. (2019). Examining the biomarkers and molecular mechanisms of medulloblastoma based on bioinformatics analysis. Oncology Letters, 18(1), 433–441. https://doi.org/10.3892/ol.2019.10314
Zang, Y., Zhu, J., Li, Q., Tu, J., Li, X., & Hu, R. (2020a). miR-137–3p modulates the progression of prostate cancer by regulating the JNK3/EZH2 axis. Oncotargets and Therapy, 13, 7921–7932. https://doi.org/10.2147/ott.s256161
Zang, Y., Zhu, J., Li, Q., Tu, J., Li, X., Hu, R., & Yang, D. (2020b). miR-137-3p modulates the progression of prostate cancer by regulating the JNK3/EZH2 axis. Oncotargets and Therapy, 13, 7921–7932. https://doi.org/10.2147/ott.s256161
Zhang, W., Chen, J. H., Shan, T., Aguilera-Barrantes, I., Wang, L. S., Huang, T. H., Rader, J. S., Sheng, X., & Huang, Y. W. (2018). miR-137 is a tumor suppressor in endometrial cancer and is repressed by DNA hypermethylation. Laboratoty Investigation, 98(11), 1397–1407. https://doi.org/10.1038/s41374-018-0092-x
Zhou, M., Venkata, P. P., Viswanadhapalli, S., Palacios, B., Alejo, S., Chen, Y., He, Y., Pratap, U. P., Liu, J., Zou, Y., Lai, Z., Suzuki, T., Brenner, A. J., Tekmal, R. R., Vadlamudi, R. K., & Sareddy, G. R. (2021). KDM1A inhibition is effective in reducing stemness and treating triple negative breast cancer. Breast Cancer Research and Treatment, 185(2), 343–357. https://doi.org/10.1007/s10549-020-05963-1
Acknowledgements
Our sincere thanks to Ping Liang, Lusheng Li, and Xuan Zhe for helping to conceive and design this research. Thanks to Yang Cheng and Xueling Zhao for their assistance in design, analysis, and important contributions to the preparation of the manuscript. Thanks to the staff of the Department of Neurosurgery of Children's Hospital of Chongqing Medical University for their help in this research.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflict of interest to declare that are relevant to the content of this article.
Ethical Approval
This study was approved by the ethics committee of Children’s Hospital of Chongqing Medical University.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
The participant has consented to the submission of the research to the journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ji, W., Zhe, X., Li, L. et al. Prognostic Value of miR-137 in Children with Medulloblastoma and its Regulatory Effect on Tumor Progression. Neuromol Med 24, 215–223 (2022). https://doi.org/10.1007/s12017-021-08684-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08684-w