Abstract
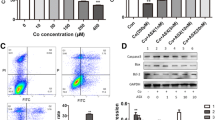
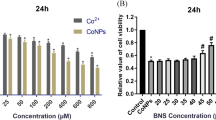
Limited information is available on the potential risk of degradation products of metal-on-metal bearings in joint arthroplasty. The aim of this study was to investigate the cytotoxicity and genotoxicity of orthopedic-related cobalt nanoparticles on human T cells in vitro. T cells were collected using magnetic CD3 microbeads and exposed to different concentrations of cobalt nanoparticles and cobalt chloride. Cytotoxicity was evaluated by methyl thiazolyl tetrazolium and lactate dehydrogenase release assay. Cobalt nanoparticles dissolution in culture medium was determined by inductively coupled plasma-mass spectrometry. To study the probable mechanism of cobalt nanoparticles effects on T cells, superoxide dismutase, catalase, and glutathione peroxidase level was measured. Cobalt nanoparticles and cobalt ions could inhibit cell viability and enhance lactate dehydrogenase release in a concentration- and time-dependent manner (P < 0.05). The levels of cobalt ion released from cobalt nanoparticles in the culture medium were less than 40% and increased with cobalt nanoparticles concentration. Cobalt nanoparticles could induce primary DNA damage in a concentration-dependent manner, and the DNA damage caused by cobalt nanoparticles was heavier than that caused by cobalt ions. Cobalt nanoparticles exposure could significantly decrease superoxide dismutase, catalase, and glutathione peroxidase activities at subtoxic concentrations (6 μM, <CC50). These findings suggested that cobalt nanoparticles could generate potential risks to the T cells of patients suffer from metal-on-metal total hip arthroplasty, and the inhibition of antioxidant capacity may play important role in cobalt nanoparticles effects on T cells.





Similar content being viewed by others
References
MacDonald SJ, McCalden RW, Chess DG, Bourne RB, Rorabeck CH, Cleland D, Leung F (2003) Metal-on-metal versus polyethylene in hip arthroplasty: a randomized clinical trial. Clin Orthop Relat Res 406:282–296
Merritt K, Brown SA (1996) Distribution of cobalt chromium wear and corrosion products and biologic reactions. Clin Orthop Relat Res 329:S233–S243
Doorn PF, Campbell PA, Worrall J, Benya PD, McKellop HA, Amstutz HC (1988) Metal wear particle characterization from metal on metal total hip replacements: transmission electron microscopy study of periprosthetic tissues and isolated particles. J Biomed Mater Res 42:103–111
Firkins PJ, Tipper JL, Saadatzadeh MR, Ingham E, Stone MH, Farrar R, Fisher J (2001) Quantitative analysis of wear and wear debris from metal-on-metal hip prostheses tested in a physiological hip joint simulator. Biomed Mater Eng 11:143–157
Davies AP, Willert HG, Campbell PA, Learmonth ID, Case CP (2005) An unusual lymphocytic perivascular infiltration in tissues around contemporary metal-on-metal joint replacements. J Bone Joint Surg Am 87:18–27
Korovessis P, Petsinis G, Repanti M, Repantis T (2006) Metallosis after contemporary metal-on-metal total hip arthroplasty—five to nine-year follow-up. J Bone Joint Surg Am 88:1183–1191
Milosev I, Trebse R, Kovac S, Cor A, Pisot V (2006) Survivorship and retrieval analysis of Sikomet metal-on-metal total hip replacements at a mean of seven years. J Bone Joint Surg Am 88:1173–1182
Boardman DR, Middleton FR, Kavanagh TG (2006) A benign psoas mass following metal-on-metal resurfacing of the hip. J Bone Joint Surg Br 88:402–404
Campbell P, Shimmin A, Walter L, Solomon M (2008) Metal sensitivity as a cause of groin pain in metal-on-metal hip resurfacing. J Arthroplasty 23:1080–1085
Pandit H, Glyn-Jones S, McLardy-Smith P, Gundle R, Whitwell D, Gibbons CL, Ostlere S, Athanasou N, Gill HS, Murray DW (2008) Pseudotumours associated with metal-on-metal hip resurfacings. J Bone Joint Surg Br 90:847–851
Ladon D, Doherty A, Newson R, Turner J, Bhamra M, Case CP (2004) Changes in metal levels and chromosome aberrations in the peripheral blood of patients after metal-on-metal hip arthroplasty. J Arthroplasty 19:78–83
Milosev I, Remskar M (2008) In vivo production of nanosized metal wear debris formed by tribochemical reaction as confirmed by high-resolution TEM and XPS analyses. J Biomed Mater Res A 91:1100–1110
Sun C, Lee JSH, Zhang M (2008) Magnetic nanoparticles in MR imaging and drug delivery. Adv Drug Deliver Rev 60:1252–1265
Lu AH, Salabas EL, Schüth F (2007) Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew Chem Int Ed 46:1222–1244
Kwon YM, Xia Z, Glyn-Jones S, Beard D, Gill HS, Murray DW (2009) Dose-dependent cytotoxicity of clinically relevant cobalt nanoparticles and ions on macrophages in vitro. Biomed Mater 4:025018
Colognato R, Bonelli A, Ponti J, Farina M, Bergamaschi E, Sabbioni E, Migliore L (2008) Comparative genotoxicity of cobalt nanoparticles and ions on human peripheral leukocytes in vitro. Mutagenesis 23:377–382
Gautam N, Das S, Mahapatra SK, Chakraborty SP, Kundu PK, Roy S (2010) Age associated oxidative damage in lymphocytes. Oxid Med Cell Longev 3:275–282
Ponti J, Sabbioni E, Munaro B, Broggi F, Marmorato P, Franchini F, Colognato R, Rossi F (2009) Genotoxicity and morphological transformation induced by cobalt nanoparticles and cobalt chloride: an in vitro study in Balb/3T3 mouse fibroblasts. Mutagenesis 24:439–445
Colgnato R, Bonelli A, Ponti J, Farina M, Bergamaschi E, Sabbioni E, Migliore L (2008) Comparative genotoxicity of cobalt nanoparticles and ions on human peripheral leukocytes in vitro. Mutagenesis 23:377–382
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Konca K, Lankoff A, Banasik A, Lisowska H, Kuszewski T, Gozdz S, Koza Z, Wojcik A (2003) A cross-platform public domain PC image-analysis program for the comet assay. Mutat Res 534:15–20
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126
Rotruck JT, Pope A, Ganther HE, Swanson AB (1973) Selenium: biochemical roles as component of glutathione peroxidase. Science 179:588–590
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–267
Tice RR, Agurell E, Anderson D, Burlinson B, Hartmann A, Kobayashi H, Miyamae Y, Rojas E, Ryu JC, Sasaki YF (2000) Single cell gel/Comet assay: guidelines for in vitro genetic toxicology testing. Environ Mol Mutagen 35:206–221
Papageorgiou I, Brown C, Schins R, Singh S, Newson R, Davis S, Fish J, Ingham E, Case CP (2007) The effect of nano- and micron-sized particles of cobalt–chromium alloy on human fibroblasts in vitro. Biomaterials 28:2946–2958
Saeedi Saravi SS, Karami S, Karami B, Shokrzadeh M (2009) Toxic effects of cobalt chloride on hematological factors of common carp. Bio Trace Ele Res 132:144–152
Limbach LK, Wick P, Manser P, Grass RN, Bruinink A, Stark WJ (2007) Exposure of engineered nanoparticles to human lung epithelial cells: influence of chemical composition and catalytic activity on oxidative stress. Environ Sci Technol 41:4158–4163
Papis E, Gornati R, Prati M, Ponti J, Sabbioni E, Bernardini G (2007) Gene expression in nanotoxicology research: analysis by differential display in BALB3T3 fibroblasts exposed to cobalt particles and ions. Toxicol Lett 170:185–192
Lison D, De Boeck M, Verougstraete V, Kirsch-Volders M (2001) Update on the genotoxicity and carcinogenicity of cobalt compounds. Occup Environ Med 58:619–625
Petit A, Mwale F, Tkaczyk C, Antoniou J, Zukor DJ, Huk OL (2005) Induction of protein oxidation by cobalt and chromium ions in human U937 macrophages. Biomaterials 26:4416–4422
Hartwig A, Asmuss M, Ehleben I, Herzer U, Kostelac D, Pelzer A, Schwerdtle T, Burkle A (2002) Interference by toxic metal ions with DNA repair processes and cell cycle control: molecular mechanisms. Environ Health Perspect 110:797–799
Papis E, Rossi F, Raspanti M, Dalle-Donne I, Colombo G, Milzani A, Bernardini G, Gornati R (2009) Engineered cobalt oxide nanoparticles readily enter cells. Toxicol Lett 189:253–259
Pulskamp K, Diabate S, Krug HF (2007) Carbon nanotubes show no sign of acute toxicity but induce intracellular reactive oxygen species in dependence on contaminatants. Toxicol Lett 168:58–74
Park EJ, Choi J, Park Y, Park K (2008) Oxidative stress induced by cerium oxide nanoparticles in cultured BEAS-2B cells. Toxicology 1–2:90–100
Simko M, Gazso A, Fiedeler U, Nentwich M (2011) Nanoparticles, free radicals and oxidative stress. NanoTrust Dossiers 12:1–3
Granchi D, Savarino L, Ciapetti G, Cenni E, Rotini R, Mieti M, Baldini N, Giunti A (2003) Immunological changes in patients with primary osteoarthritis of the hip after total joint replacement. J Bone Joint Surg Br 85:758–764
Hart AJ, Hester T, Sinclair K, Powell JJ, Goodship AE, Pele L, Fersht NL, Skinner J (2006) The association between metal ions from hip resurfacing and reduced T-cell counts. J Bone Joint Surg Br 88:449–454
Hart AJ, Skinner JA, Winship P, Faria N, Kulinskaya E, Webster D, Muirhead-Allwood S, Aldam CH, Anwar H, Powell JJ (2008) Circulating levels of cobalt and chromium from metal-on-metal hip replacement are associated with CD8+ T-cell lymphopenia. J Bone Joint Surg Br 91:835–842
IARC (2006) IARC monographs on the evaluation of carcinogenic risks to humans: cobalt in hard metals and cobalt sulfate, gallium arsenide, indium phosphide and vanadium pentoxide. IARC Monogr Eval Carcinog Risks Hum 86:1–294
Acknowledgement
This study was funded by the National Natural Science Foundation of China (No. 81171743).
Author information
Authors and Affiliations
Corresponding author
Additional information
Haitao Jiang and Fan Liu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Jiang, H., Liu, F., Yang, H. et al. Effects of Cobalt Nanoparticles on Human T Cells In Vitro. Biol Trace Elem Res 146, 23–29 (2012). https://doi.org/10.1007/s12011-011-9221-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-011-9221-8