Abstract
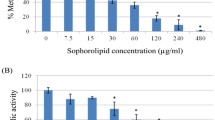
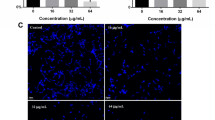
Biosurfactant produced from Pseudomonas aeruginosa DSVP20 was evaluated for its potential to disrupt Candida albicans biofilm formed on polystyrene (PS) surfaces in this investigation. P. aeruginosa DSVP20 exhibited optimum production of biosurfactant (5.8 g L−1) after 96 h of growth with an ability to reduce surface tension of the aqueous solution from 72 to 28 mN m−1. Analysis of purified biosurfactant with FT-IR, 1H and 13C NMR and MALDI-TOF MS revealed it to be di-rhamnolipid (RL-2) in nature. Biofilm disrupting ability of RL-2 (0.16 mg mL−1) on Candida cells when checked using XTT reduction assay revealed that about 50 % of the cells remain adhered to 96-well plate after 2 h of treatment, while up to 90 % reduction in pre-formed C. albicans biofilm on PS surface was observed with RL-2 (5.0 mg mL−1) in a dose-dependent manner. Microscopic analyses (SEM and CLSM) further confirm the influence of RL-2 on disruption of Candida biofilm extracellular matrix on PS surface which can be exploited as a potential alternative to the available conventional therapies.











Similar content being viewed by others
References
Yu, H., & Huang, G. H. (2011). Soil Sediment Contamination, 20, 274–278.
Nitschke, M., & Costa, S. (2007). Trends in Food Science and Technology, 18, 252–259.
Dusane, D. H., Nancharaiah, Y. V., Zinjarde, S. S., & Venugopalan, V. P. (2010). Colloids and Surfaces. B, Biointerfaces, 81, 242–248.
Irie, Y., O’Toole, G. A., & Yuk, M. H. (2005). FEMS Microbiology Letters, 250, 237–243.
Rodrigues, L. R., Banat, I. M., van der Mei, H. C., Teixeira, J. A., & Oliveira, R. (2006). Journal of Applied Microbiology, 100, 470–480.
Stanghellini, M. E., & Miller, R. M. (1997). Plant Disease, 81, 4–12.
Wang, X., Gong, L., Liang, S., Han, X., Zhu, C., & Li, Y. (2005). Harmful Algae, 4, 433–443.
de Araujo, L. V., Abreu, F., Lins, U., de Melo Santa Anna, L. M., Nitschke, M., & Freire, D. M. G. (2011). Food Research International, 44, 481–488.
Cao, Y., Dai, B., Wang, Y., Huang, S., Xu, Y., Cao, Y., et al. (2008). International Journal of Antimicrobial Agents, 32, 73–77.
An, M., Shen, H., Cao, Y. B., Zhang, J. D., Cai, Y., Wang, R., et al. (2009). International Journal of Antimicrobial Agents, 33, 258–263.
Chandra, J., Kuhn, D. M., Mukherjee, P. K., Hoyer, L. L., McCormick, T., & Ghannoum, M. A. (2001). Journal of Bacteriology, 183, 5385–5394.
Agarwal, V., Lal, P., & Pruthi, V. (2008). Mycopathologia, 165, 13–19.
Jain, N., Kohli, R., Cook, E., Gialanella, P., Chang, T., & Fries, B. C. (2007). Applied and Environmental Microbiology, 73, 1697–1703.
Bruzual, I., Riggle, P., Hadley, S., & Kumamoto, C. A. (2007). Journal of Antimicrobial Chemotherapy, 59, 441–450.
Law, D., Moore, C. B., Wardle, H. M., Ganguli, L. A., Keaney, M. G. L., & Denning, D. W. (1994). Journal of Antimicrobial Chemotherapy, 34, 659–668.
Banat, I. M., Makkar, R. S., & Cameotra, S. S. (2000). Applied Microbiology and Biotechnology, 53, 495–508.
Rivardo, F., Turner, R. J., Allegrone, G., Ceri, H., & Martinotti, M. G. (2009). Applied Microbiology and Biotechnology, 83, 541–553.
Pruthi, V., & Cameotra, S. S. (2003). Journal of Surfactants and Detergents, 6, 65–68.
Plaza, G., Zjawiony, I., & Banat, I. (2006). Journal of Petroleum Science and Engineering, 50, 71–77.
Chen, S. Y., Wei, Y. H., & Chang, J. S. (2007). Applied Microbiology and Biotechnology, 76, 67–74.
Caiazza, N. C., Shanks, R. M. Q., & O’Toole, G. A. (2005). Journal of Bacteriology, 187, 7351–7361.
Pemmaraju, S. C., Sharma, D., Singh, N., Panwar, R., Cameotra, S. S., & Pruthi, V. (2012). Applied Biochemistry and Biotechnology. doi:10.1007/s12010-012-9613-z.
Cameotra, S. S., & Singh, P. (2009). Microbial Cell Factories, 8, 16.
Sneath, P. H. A., Mair, N. S., & Sharpe, M. E. (1986). Bergey’s Manual of Systematic Bacteriology Vol. 2 (pp. 999–1436). Baltimore: Williams & Wilkins.
De Clerck, E., Rodrıguez-Dıaz, M., Vanhoutte, T., Heyrman, J., Logan, N. A., & De Vos, P. (2004). International Journal of Systematic and Evolutionary Microbiology, 54, 941–946.
Pitcher, D. G., Saunders, N. A., & Owen, R. J. (1989). Letters in Applied Microbiology, 8, 151–156.
Christova, N., Tuleva, B., Lalchev, Z., Jordanova, A., & Jordanov, B. (2004). Zeitschrift für Naturforschung, 59c, 70–74.
Chandrasekaran, E. V., & Bemiller, J. N. (1980). In R. L. Whistler & M. L. Wolfrom (Eds.), Methods in carbohydrate chemistry, constituent analysis of glycosaminoglycans (pp. 89–96). New York: Academic Press.
Worakitsiri, P., Pornsunthorntaweea, O., Thanpitcha, T., Chavadej, S., Weder, C., & Rujiravanit, R. (2011). Synthetic Metals, 161, 298–306.
Thanomsub, B., Pumeechockchai, W., Limtrakul, A., Arunrattiyakorn, P., Petchleelaha, W., Nitoda, T., et al. (2006). Bioresource Technology, 97, 2457–2461.
Silva, W. J., Seneviratne, J., Parahitiyawa, N., Rosa, E. A., Samaranayake, L. P., & Cury, A. A. (2008). Brazilian Dental Journal, 19, 364–369.
Lal, P., Sharma, D., Pruthi, P., & Pruthi, V. (2010). Journal of Applied Microbiology, 109, 128–136.
Felsenstein, J. (1989). PHYLIP-Phylogeny Inference Package (Version 3.2). Cladistics, 5, 164–166.
Parreira, A. G., Tótola, M. R., Jham, G. N., Da Silva, S. L., & Borges, A. C. (2011). British Biotechnology Journal, 1, 18–28.
Mishra, S., Jyot, J., Kuhad, R. C., & Lal, B. (2001). Applied and Environmental Microbiology, 67, 1675–1681.
Hazra, C., Kundu, D., Ghosh, P., Joshi, S., Dandia, N., & Chaudharia, A. (2011). Journal of Chemical Technology and Biotechnology, 86, 185–198.
Wei, Y., Cheng, C., Chien, C., & Wan, H. (2008). Process Biochemistry, 43, 69–77.
Abdel-Mawgoud, A. M., Aboulwafa, M., & Hassouna, N. (2009). Applied Biochemistry and Biotechnology, 157, 329–345.
Bharali, P., & Konwar, B. K. (2011). Applied Biochemistry and Biotechnology, 164, 1444–1460.
Pornsunthorntawee, O., Maksung, S., Huayyai, O., Rujiravanit, R., & Chavadej, S. (2009). Bioresource Technology, 100, 812–818.
Deziel, E., Lépine, F., Milot, S., & Villemur, R. (2000). Biochimica et Biophysica Acta, 1485, 145–152.
Ganesh, A., & Lin, J. (2009). African Journal of Biotechnology, 8, 5847–5854.
Deziel, E., Lépine, F., Dennie, D., Boismenu, D., Mamer, O. A., & Villemur, R. (1999). Biochimica et Biophysica Acta, 1440, 244–252.
Nitschke, M., Costa, S. G., Haddad, R., Goncalves, L. A., Eberlin, M. N., & Contiero, J. (2005). Biotechnology Progress, 21, 1562–1566.
Soberon-Chavez, G., Lepine, F., & Deziel, E. (2005). Applied Microbiology and Biotechnology, 68, 718–725.
Razaa, A. Z., Khana, M. S., Khalid, Z. M., & Rehmanb, A. (2006). Zeitschrift für Naturforschung, 61c, 87–94.
Nett, J. E., Guite, K. M., Ringeisen, A., Holoyda, K. A., & Andes, D. R. (2008). Antimicrobial Agents and Chemotherapy, 52, 3411–3413.
Sotirova, A. V., Spasova, D. I., Galabova, D. N., Karpenko, E., & Shulga, A. (2010). Current Microbiology, 56, 639–644.
Fracchia, L., Cavallo, M., Allegrone, G., & Martinotti, M. G. (2010). In A. Mendez-Vilas (Ed.), Current research technology and education topics in applied microbiology and microbial technology, vol. 2: A Lactobacillus-derived biosurfactant inhibits biofilm formation of human pathogenic Candida albicans biofilm producers. Spain: Formatex.
Davey, M. E., Caiazza, N. C., & OToole, G. A. (2003). Journal of Bacteriology, 185, 1027–1036.
Carrillo, C., Teruel, J., Aranda, F., & Ortiz, A. (2003). Biochimica et Biophysica Acta, 1611, 91–97.
Borecka-Melkusova, S., & Bujadakova, H. (2008). Canadian Journal of Microbiology, 54, 718–724.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, N., Pemmaraju, S.C., Pruthi, P.A. et al. Candida Biofilm Disrupting Ability of Di-rhamnolipid (RL-2) Produced from Pseudomonas aeruginosa DSVP20. Appl Biochem Biotechnol 169, 2374–2391 (2013). https://doi.org/10.1007/s12010-013-0149-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0149-7