Opinion statement
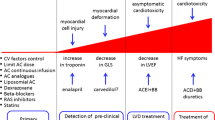
With recent novel therapies, cancer survival has improved and chemotherapy-induced cardiac toxicity including left ventricular dysfunction and heart failure has a greater potential impact on long-term outcomes. Hence, the prevention of cardiotoxicity should be an important objective when planning the therapy of cancer patients. Different pharmacologic and nonpharmacologic approaches have been shown to be effective in small trials, most of them in patients treated with anthracyclines. Treatment of cardiovascular risk factors, the implementation of a healthy lifestyle, and changes in the administration of anthracyclines have been shown to be effective to prevent cardiotoxicity, as is the administration of antioxidants such as dexrazoxane, and some cardiovascular drugs such as ACE-inhibitors and beta-blockers. Ongoing studies are analyzing the effects of these drugs on anthracycline- and trastuzumab-induced cardiotoxicity. Nevertheless, the future of the prevention of cardiac toxicity should include the study of the exact mechanism of toxicity of each cardiotoxic drug and the genetic individual susceptibility of each patient. With this information, correct risk stratification could be performed when planning the initial therapy and new drugs with less cardiotoxicity could be developed. Also, the diagnostic and prognostic value of different cardiac biomarkers and of imaging techniques for each cardiotoxic drug should be established. Finally, further large randomized studies are needed to confirm the results of prior pilot studies on the effects of cardioprotective drugs.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300.
Suter TM, Ewer MS. Cancer drugs and the heart: importance and management. Eur Heart J. 2013;34:1102–11.
Eschenhagen T, Force T, Ewer MS, Keuenaer GW, Suter TM, Anker SD. CV side effects of cancer therapies: a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2011;13:1–10.
Lipshultz S, Lipsitz S, Sallen S, et al. Chronic progressive cardiac dysfunction years after doxorubicin therapy for childhood acute lymphoblastic leukemia. J Clin Oncol. 2005;23:2629–36.
Wouters K, Kremer L, Herman E, Lipshultz S. Protecting against anthracyclines-induce myocardial damage: a review of the most promising strategies. Br J Haematol. 2005;131:561–78.
Zhang S, Liu X, Bawa-Khalfe T, et al. Identification of the molecular basis of doxorubicin-induced cardiotoxicity. Nat Med. 2012;18:1639–42. Study describing the molecular mechanism of anthracycline-induced cardiotoxicity. The results open the possibility to identify patients at high-risk of cardiotoxicity, and of developing less cardiotoxic drugs.
Khouri M, Douglas P, Mackey J, et al. Cancer therapy-induced cardiac toxicity in early breast cancer. Circulation. 2012;126:2749–63.
Zver S, Zadnik V, Bunc M, Rogel P, Cernelc P, Kozelj M. Cardiac toxicity of high-dose cyclophosphamide in patients with multiple myeloma undergoing autologous hematopoietic stem cell transplantation. Int J Hematol. 2007;85:408–14.
García-Alvarez A, García-Albéniz X, Esteve J, Rovira M, Bosch X. Cardiotoxicity of tyrosine-kinase-targeting drugs. Cardiovasc Hematol Agents Med Chem. 2010;8:11–21.
Curigliano G, Cardinale D, Suter T, et al. CV toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidlines. Ann Oncol. 2012;23 suppl 7:vii155–66. An actualized clinical guideline based on current evidence.
Thavendiranathan P, Grant AD, Negishi T, Plana JC, Popović ZB, Marwick TH. Reproducibility of echocardiographic techniques for sequential assessment of LV ejection fraction and volumes: application to patients undergoing cancer chemotherapy. J Am Coll Cardiol. 2013;61:77–84.
Yancy CW, Jessup M, Bozkurt B, et al. ACCF/AHA Guideline for the Management of Heart Failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013. doi:10.1161/CIR.0b013e31829e8776.
Scott JM, Khakoo A, Mackey JR, Haykowsky MJ, Douglas PS, Jones LW. Modulation of anthracycline-induced cardiotoxicity by aerobic exercise in breast cancer: current evidence and underlying mechanisms. Circulation. 2011;124:642–50. Good review on the beneficial effects of exercise on anthracycline-induced cardiotoxicity.
Chicco A, Hydock D, Schneider C, Hayward R. Low-intensity exercise training during doxorubicin treatment protects against cardiotoxicity. J Appl Physiol. 2006;100:519–27.
Smith L, Cornelius V, Plummer C, et al. Cardiotoxicity of anthracycline agents for the treatment of cancer: systematic review and meta-analysis of randomised controlled trials. BMC Cancer. 2010;10:337. Good systematic review on anthracycline cardiac toxicity showing that there is not sufficiently robust evidence to support clear evidence-based recommendations on different anthracycline treatment regimens or for routine use of cardiac protective agents or liposomal formulations, and that there is a need to improve cardiac monitoring in oncology trials.
Van Dalen E, Michiels E, Caron H, et al. Different anthracycline derivates for reducing cardiotoxicity in cancer patients. Cochrane Database Syst Rev. 2010;3, CD005006. Cochrane meta-analysis of all clinical trials comparing doxorubicin vs epirubicin, and conventional doxorubicin vs liposomal-encapsulated doxorubicin.
Van Dalen E, Caron H, Dickinson H, et al. Cardioprotective interventions for cancer patients receiving anthracyclines. Cochrane Database Syst Rev. 2011;6, CD003917. Cochrane meta-analysis of clinical trials performed until 2010 to prevent chemotherapy-induced cardiac toxicity. Only dexrazoxane is efficacious in preventing heart damage in patients treated with anthracyclines, and that there is no evidence of a lower response rate to cancer therapy. Negative effects on progression-free survival were not identified.
Lipshultz SE, Rifai N, Dalton VM, et al. The effect of dexrazoxane on myocardial injury in doxorubicin-treated children with acute lymphoblastic leukemia. N Engl J Med. 2004;351:145–53.
Lipshultz SE, Scully RE, Lipsitz SR, et al. Assessment of dexrazoxane as a cardioprotectant in doxorubicin-treated children with high-risk acute lymphoblastic leukaemia: long-term follow-up of a prospective, randomised, multicentre trial. Lancet Oncol. 2010;11:950–61.
Tebbi CK, London WB, Friedman D, Villaluna D, De Alarcon PA, Constine LS, et al. Dexrazoxane-associated risk for acute myeloid leukemia/myelodysplastic syndrome and other secondary malignancies in pediatric Hodgkin's disease. J Clin Oncol. 2007;25:493–500.
Salzer WL, Devidas M, Carroll WL, Winick N, Pullen J, Hunger SP, et al. Long-term results of the pediatric oncology group studies for childhood acute lymphoblastic leukemia 1984–2001: a report from the children's oncology group. Leukemia. 2010;24:355–70.
Toko H, Oka T, Zou Y, et al. Angiotensin II type 1a receptor mediates doxorubicin-induced cardiomyopathy. Hypertens Res. 2000;25:597–603.
Vaynblat M, Shah HR, Bhaskaran D, Ramdev G, Davis WJ, Cunningham JN, et al. Simultaneous angiotensin converting enzyme inhibition moderates ventricular dysfunction caused by doxorubicin. Eur J Heart Fail. 2002;4:583–6.
Sacco G, Bigioni M, Evangelista S, Goso C, Manzini S, Maggi CA. Cardioprotective effects of zofenopril, a new angiotensin-converting enzyme inhibitor, on doxorubicin-induced cardiotoxicity in the rat. Eur J Pharmacol. 2001;414:71–8.
Silber JH, Cnaan A, Clark BJ, et al. Enalapril to prevent cardiac function decline in long-term survivors of pediatric cancer exposed to anthracyclines. J Clin Oncol. 2004;22:820–8.
Cardinale D, Colombo A, Sandri M, et al. Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation. 2006;114:2474–81. First study to prove the efficacy of a troponin-guided strategy of myocardial prevention with enalapril in patients in high-risk patients submitted to high-dose chemotherapy.
Nakamae H, Tsumura K, Terada Y, et al. Notable effects of angiotensin II receptor blocker Valsartan on acute cardiotoxic changes after standard chemotherapy with cyclophosphamide, doxorubicin, vincristine and prednisolone. Cancer. 2005;104:2492–8.
Cadeddu C, Piras A, Mantovani G, et al. Protective effects of the angiotensin II receptor blocker telmisartan on epirubicin-induced inflammation, oxidative stress, and early ventricular impairment. Am Heart J. 2010;160:487e.1–7.
Santos DL, Moreno AJ, Leino RL, Froberg MK, Wallace KB. Carvedilol protects against doxorubicin-induced mitochondrial cardiomyopathy. Toxicol Appl Pharmacol. 2002;185:218–27.
Spallarossa P, Garibaldi S, Altieri P, Fabbi P, Manca V, Nasti S, et al. Carvedilol prevents doxorubicin-induced free radical release and apoptosis in cardiomyocytes in vitro. J Mol Cell Cardiol. 2004;37:837–46.
Kalay N, Basar E, Ozdogru I, et al. Protective effects of carvedilol against anthracycline-induced cardiomyopathy. J Am Coll Cardiol. 2006;48:2258–62. Study demonstrating the cardioprotective effects of carvedilol on anthracycline-induced LV dysfunction.
El-Shitany NA, Tolba OA, El-Shanshory MR, El-Hawary EE. Protective effect of carvedilol on adriamicyn-induced LV dysfunction in children with acute lymphoblastic leukemia. J Card Fail. 2012;18:607–13.
Georgakopoulos P, Roussou P, Matsakas E, et al. Cardioprotective effect of metoprolol and enalapril in doxorubicin-treated lymphoma patients: a prospective parallel-group, randomized, controlled study with 36 month follow-up. Am J Hematol. 2010;85:894–907.
Bosch X, Rovira M, Sitges M, et al. Enalapril and carvedilol for preventing chemotherapy-induced LV systolic dysfunction in patients with malignant hemopathies: the OVERCOME trial (preventiOn of LV dysfunction with Enalapril and caRvedilol in patients submitted to intensive ChemOtherapy for the treatment of Malignant hEmopathies). J Am Coll Cardiol. 2013;61:2355–62. First study performed in patients with malignant hemopathies using enalapril and carvedilol at the time of hospital admission as a primordial prevention strategy, and showing a beneficial effect on chemotherapy-induced LV systolic dysfunction. Patients with acute leukemia treated with more intense chemotherapy regimens and including anthracyclines benefited the most from this prevention strategy.
Riad A, Bien S, Westermann D, et al. Pretreatment with statin attenuates the cardiotoxicity of doxorubicin in mice. Cancer Res. 2009;69:695–9.
Seicean S, Seicean A, Plana JC, Budd GT, Marwick TH. Effect of statin therapy on the risk for incident heart failure in patients with breast cancer receiving anthracycline chemotherapy: an observational clinical cohort study. J Am Coll Cardiol. 2012;60:2384–90.
Acar Z, Kale A, Turgut M, et al. Efficiency of atorvastatin in the protection of anthracycline-induced cardiomyopathy. J Am Coll Cardiol. 2011;58:988–9.
Blanco J, Leisenring W, Gonzalez-Covarrubias V, et al. Genetic polymorphisms in the carbonyl reductase 3 gene CBR3 and the NAD(P)H: quinone oxidoreductase 1 gene NQO1 in patients who developed anthracycline-related congestive heart failure after childhood cancer. Cancer. 2008;112:2789–95.
Visscher H, Ross CJD, Rassekh SR, et al. Pharmacogenomic prediction of anthracycline-induced cardiotoxicity in children. J Clin Oncol. 2012;30:1422–8. One of the biggest genetic studies performed to find predictors of anthracycline-induced cardiotoxicity. 3000 single-nucleotide polymorphisms were evaluated in 156 patients, and the results replicated in two other cohort of patients.
Compliance with Ethics Guidelines
Conflict of Interest
Dr. Laura Carola Magnano, Dr. Nuria Martínez Cibrian, Dr. Xavier Andrade González , and Dr. Xavier Bosch each declare no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Cardio-oncology
Rights and permissions
About this article
Cite this article
Magnano, L.C., Martínez Cibrian, N., Andrade González, X. et al. Cardiac Complications of Chemotherapy: Role of Prevention. Curr Treat Options Cardio Med 16, 312 (2014). https://doi.org/10.1007/s11936-014-0312-7
Published:
DOI: https://doi.org/10.1007/s11936-014-0312-7