Abstract
Purpose of Review
The purpose of the following review is to summarize the most recent understanding of migraine pathophysiology, as well as of basic and clinical science pharmacologic literature regarding the development of calcitonin gene receptor peptide (CGRP) antagonists as a novel therapeutic modality for the treatment of migraine headaches. A review is provided of erenumab, the first of its class FDA approved CGRP antagonist.
Recent Findings
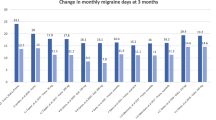
Despite its high prevalence, the occurrence and treatment of migraine headaches is poorly understood. Erenumab and CGRP antagonists as a whole significantly reduce the average number of migraine days experienced in migraine sufferers.
Summary
CGRP antagonists appear to significantly improve treatment outcomes in patients who suffer from episodic and chronic migraines. Erenumab is the first CGRP antagonist to be FDA approved for public use; however, further development of biologics in this class is underway.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bajwa ZH, Wootton RJ, Wippold FJ II. Evaluation of headache in adults. UpToDate. 2018.
Society IH. The International Classification of Headache Disorders, 3rd edn. 2018.
Cutrer FM, Bajwa ZH. Pathophysiology, clinical manifestations, and diagnosis of migraine in adults. UpToDate. 2017.
Russo A. Calcitonin gene-related peptide (CGRP): a new target for migraine. Annu Rev Pharmacol Toxicol. 2015;55:533–52.
Giffin N, Lipton R, Silberstein J, Olesen S, Goadsby P. The migraine postdrome: an electronic diary study. Neurology. 2016;87:309–13.
Hadjikhani N, Sanchez Del Rio M, Wu O, Schwartz D, Bakker D, Fischl B, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci U S A. 2001;98:4687–92.
Gursoy-Ozdemir Y, Qiu J, Matsuoka N, Bolay H, Bermpohl D, Jin H, et al. Cortical spreading depression activates and upregulates MMP-9. J Clin Invest. 2004;113:1447–55.
Karatas H, Erdener S, Gursoy-Ozdemir Y, Lule S, Eren-Koçak E, Sen Z, et al. Spreading depression triggers headache by activating neuronal Panx1 channels. Science. 2013;339:1092–5.
Lipton R, Stewart W, Diamond S, Diamond M, Reed M. Prevalence and burden of migraine in the United States: data from the American Migraine Study II. Headache. 2001;41:646–57.
Lipton R, Bigal M, Diamond M, Freitag F, Reed M, Stewart W, et al. Migraine prevalence, disease burden, and the need for preventive therapy. Neurology. 2007;68:343–9.
Stewart W, Wood C, Reed M, Roy J, Lipton R, Group AA. Cumulative lifetime migraine incidence in women and men. Cephalagia. 2008;28:1170–8.
Martin V, Behbehani M. Toward a rational understanding of migraine trigger factors. Med Clin North Am. 2001;85:911–41.
Kelman L. The triggers or precipitants of the acute migraine attack. Cephalagia. 2007;27:394–402.
Bajwa ZH, Smith JH. Preventive treatment of migraine in adults. UpToDate. 2018.
Goadsby P, Lipton R, Ferrari M. Migraine—current understanding and treatment. N Engl J Med. 2002;346:257–70.
Bajwa ZH, Smith JH. A cute treatment of migraine in adults. UpToDate. 2018.
Breeze A, Harvey T, Bazzo R, Campbell I. Solution structure of human calcitonin gene-related peptide by 1H NMR and distance geometry with restrained molecular dynamics. Biochemistry. 1991;30:575–82.
Sexton P, Christopoulos G, Christopoulos A, Nylen E, Snider R Jr, Becker K. Procalcitonin has bioactivity at calcitonin receptor family complexes: potential mediator implications in sepsis. Crit Care Med. 2008;36:1637–40.
Muddhrry PK, Ghatki MA, Spokks RA, Jonhs PM, Pierson AM, Hamid Q, et al. Differential expression of α-CGRP and β-CGRP by primary sensory neurons and enteric autonomic neurons of the rat. Neuroscience. 1988;25:195–205.
Poyner D, Sexton P, Marshall I, Smith D, Quirion R, Born W, et al. International Union of Pharmacology. XXXII. The mammalian calcitonin gene-related peptides, adrenomedullin, amylin, and calcitonin receptors. Pharm Rev. 2002;54:233–46.
Zhang Z, Winborn C, Marquez de Prado B, Russo A. Sensitization of calcitonin gene-related peptide receptors by receptor activity-modifying protein-1 in the trigeminal ganglion. J Neurosci. 2007;27:2693–703.
Archbold J, Flanagan J, Watkins H, Gingell J, Hay D. Structural insights into RAMP modification of secretin family G protein-coupled receptors: implications for drug development. Trends Pharmacol Sci. 2011;32:591–600.
Edvinsson L. The trigeminovascular pathway: role of CGRP and CGRP receptors in migraine. Headache. 2017;57:47–55.
Walker CS, Conner AC, Poyner DR, Hay DL. Regulation of signal transduction by calcitonin gene-related peptide receptors. Trends Pharmacol Sci. 2010;31:476–83.
Walker C, Sajedeh E, Bower RL, Wilderman A, Insel PA, Edvinsson L, et al. A second trigeminal CGRP receptor: function and expression of the AMY1 receptor. Ann Clin Transl Neurol. 2015;2:595–608.
Walker C, Hay D. CGRP in the trigeminovascular system: a role for CGRP, adrenomedullin andamylin receptors? Br J Pharmocol. 2013;170:1293–307.
McCulloch J, Uddman R, Kingman T, Edvinsson L. Calcitonin gene-related peptide: functional role in cerebrovascular regulation. Proc Natl Acad Sci U S A. 1986;83:5731–5.
Goadsby P, Edvinsson L, Ekman R. Vasoactive peptide release in the extracerebral circulation of humans during migraine headache. Ann Neurol. 1990;28:183–7.
Goadsby P, Edvinsson L. The trigeminovascular system and migraine: studies characterizing cerebrovascular and neuropeptide changes seen in humans and cats. Ann Neurol. 1993;33:48–56.
Lassen L, Haderslev P, Jacobsen V, Iversen H, Sperling B, Olesen J. CGRP may play a causative role in migraine. Cephalagia. 2002;22:54–61.
Lennerz J, Rühle V, Ceppa E, Neuhuber W, Bunnett N, Grady E, et al. Calcitonin receptor-like receptor (CLR), receptor activity-modifying protein 1 (RAMP1), and calcitonin gene-related peptide (CGRP) immunoreactivity in the rat trigeminovascular system: differences between peripheral and central CGRP receptor distribution. J Comp Neurol. 2008;507:1277–99.
Russo A, Kuburas A, Kaiser E, Raddant A, Recober A. A potential preclinical migraine model: CGRP-sensitized mice. Mol Cell Pharmocol. 2009;1:264–70.
Coull J, Beggs S, Boudreau D, Boivin D, Tsuda M, Inoue K, et al. BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature. 2005;438:1017–21.
Souslova V, Cesare P, Ding Y, Akopian A, Stanfa L, Suzuki R, et al. Warm-coding deficits and aberrant inflammatory pain in mice lacking P2X3 receptors. Nature. 2000;407:1015–7.
MacKenzie ET, Edvinsson LSB. Functional bases for a central serotonergic involvement in classic migraine: a speculative view. Cephalalgia. 1985;5:69–781.
Cady RK, Vause CV, Ho TW, Bigal ME, Durham PL. Elevated saliva calcitonin gene-related peptide levels during acute migraine predict therapeutic response to rizatriptan. Headache. 2009;49:1258–66.
•• Goadsby PJ, Reuter U, Hallström Y, Broessner G, Bonner JH, Zhang F, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123–32 Important STRIVE phase 3 trial demonstrating efficacy of erenumab for the prevention of episodic migraines.
Edvinsson L. CGRP receptor antagonists and antibodies against CGRP and its receptor in migraine treatment. Br J Clin Pharmacol. 2015;80:193–9.
Hostetler ED, Joshi AD, Sanabria-Bohorquez S, Fan H, Zeng Z, Purcell M, et al. In vivo quantification of calcitonin gene-related peptide receptor occupancy by telcagepant in rhesus monkey and human brain using the positron emission tomography tracer [11C]MK-4232. J Pharmacol Exp Ther. 2013;347:478–86.
Pellesi L, Guerzoni S, Pini LA. Spotlight on anti-CGRP monoclonal antibodies in migraine: the clinical evidence to date. Clin Pharmacol Drug Dev. 2017;6:534–47.
Lipton RB, Brennan A, Palmer S, Hatswell AJ, Porter JK, Sapra S, et al. Estimating the clinical effectiveness and value-based price range of erenumab for the prevention of migraine in patients with prior treatment failures: a US societal perspective. J Med Econ. Informa UK Ltd. 2018;21:666–75.
Vu T, Ma P, Chen JS, de Hoon J, Van Hecken A, Yan L, et al. Pharmacokinetic-pharmacodynamic relationship of erenumab (AMG 334) and capsaicin-induced dermal blood flow in healthy and migraine subjects. Pharm Res Pharm Res. 2017;34:1784–95.
de Hoon J, Van Hecken A, Vandermeulen C, Yan L, Smith B, Chen JS, et al. Phase I, randomized, double-blind, placebo-controlled, single-dose, and multiple-dose studies of erenumab in healthy subjects and patients with migraine. Clin Pharmacol Ther. 2018;103:815–25.
Giamberardino MA, Affaitati G, Costantini R, Cipollone F, Martelletti P. Calcitonin gene-related peptide receptor as a novel target for the management of people with episodic migraine: current evidence and safety profile of erenumab. J Pain Res. 2017;10:2751–60.
•• Dodick DW, Ashina M, Brandes JL, Kudrow D, Lanteri-Minet M, Osipova V, et al. ARISE: the phase 2 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026–37 The ARISE Phase 3 trial of erenumab for the prevention of episodic migraines.
Depre C, Antalik L, Starling A, Koren M, Eisele O, Lenz RA, et al. A randomized, double-blind, placebo-controlled study to evaluate the effect of erenumab on exercise time during a treadmill test in patients with stable angina. Headache. 2018;58:715–23.
de Hoon J, Van Hecken A, Vandermeulen C, Herbots M, Kubo Y, Lee E, et al. Phase I, randomized, parallel-group, double-blind, placebo-controlled trial to evaluate the effects of erenumab (AMG 334) and concomitant sumatriptan on blood pressure in healthy volunteers. Cephalalgia. 2018;0:1–11.
Markham A. Pegvaliase: first global approval. Drugs. 2018;78:1157–61.
Reinke T. Aimovig for migraine prevention: the new kid may have trouble fitting in. Manag Care. 2018;27:10–1.
Traynor K. FDA approves licensing of erenumab-aooe to prevent migraine. Am J Heal Pharm. 2018;75:929–30.
Choy M. Pharmaceutical approval update. Pharm Ther. 2018;43:461–2.
•• Tepper S, Ashina M, Reuter U, Brandes JL, Doležil D, Silberstein S, et al. Safety and efficacy of erenumab for preventive treatment of chronic migraine: a randomised, double-blind, placebo-controlled phase 2 trial. Lancet Neurol. 2017;16:425–34 Excellent phase 2 randomized controlled trial demonstrating the safety and efficacy of erenumab for the treatment of chronic migraines.
Traynor J. μ-Opioid receptors and regulators of G protein signaling (RGS) proteins: from a symposium on new concepts in mu-opioid pharmacology. Drug Alcohol Depend. 2011;121:173–80.
• Dodick DW, Goadsby PJ, Silberstein SD, Lipton RB, Olesen J, Ashina M, et al. Safety and efficacy of ALD403, an antibody to calcitonin gene-related peptide, for the prevention of frequent episodic migraine: a randomised, double-blind, placebo-controlled, exploratory phase 2 trial. Lancet Neurol. Elsevier Ltd; 2014;13:1100–1107. Excellent phase 2 trial of Eptinezumab for the prevention of frequent episodic migraine.
• Halker Singh RB, Aycardi E, Bigal ME, Loupe PS, McDonald M, Dodick DW. Sustained reductions in migraine days, moderate-to-severe headache days and days with acute medication use for HFEM and CM patients taking fremanezumab: post-hoc analyses from phase 2 trials. Cephalalgia. 2018;0:1–9 A phase 2 trial of Fremanezumab for the treatment of episodic and chronic migraines.
• Stauffer VL, Dodick DW, Zhang Q, Carter JN, Ailani J, Conley RR. Evaluation of galcanezumab for the prevention of episodic migraine. JAMA Neurol. 2018;85054. Excellent review of Galcanezumab for the treatment of episodic migraines.
Raffaelli B, Reuter U. The biology of monoclonal antibodies: focus on calcitonin gene-related peptide for prophylactic migraine therapy. Neurotherapeutics. 2018;15:324–35.
Kaplon H, Reichert JM. Antibodies to watch in 2018. MAbs. 2018;10:183–203.
Walter S, Alibhoy A, Escandon R, Bigal ME. Evaluation of cardiovascular parameters in cynomolgus monkeys following IV administration of LBR-101, a monoclonal antibody against calcitonin gene-related peptide. MAbs. 2014;6:871–8.
Bigal ME, Walter S, Bronson M, Alibhoy A, Escandon R. Cardiovascular and hemodynamic parameters in women following prolonged CGRP inhibition using LBR-101, a monoclonal antibody against CGRP. Cephalalgia. 2014;34:968–76.
Bigal ME, Escandon R, Bronson M, Walter S, Sudworth M, Huggins JP, et al. Safety and tolerability of LBR-101, a humanized monoclonal antibody that blocks the binding of CGRP to its receptor: results of the phase 1 program. Cephalalgia. 2014;34:483–92.
Silberstein SD, Dodick DW, Bigal ME, Yeung PP, Goadsby PJ, Blankenbiller T, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017;377:2113–22.
Monteith D, Collins EC, Vandermeulen C, Van Hecken A, Raddad E, Scherer JC, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of the CGRP binding monoclonal antibody LY2951742 (galcanezumab) in healthy volunteers. Front Pharmacol. 2017;8:1–11.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Ivan Urits, Mark R. Jones, Kyle Gress, Karina Charipova, Jacob Fiocchi, and Omar Viswanath declare no conflict of interest. Dr. Kaye is a speaker for Depomed, Inc. and Merck, Inc.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Other Pain
Rights and permissions
About this article
Cite this article
Urits, I., Jones, M.R., Gress, K. et al. CGRP Antagonists for the Treatment of Chronic Migraines: a Comprehensive Review. Curr Pain Headache Rep 23, 29 (2019). https://doi.org/10.1007/s11916-019-0768-y
Published:
DOI: https://doi.org/10.1007/s11916-019-0768-y