Abstract
Purpose of Review
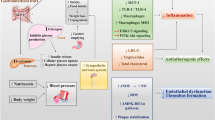
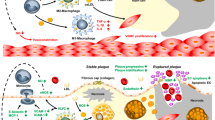
Mimetics and analogs that extend the half-life of native glucagon-like peptide-1 (GLP-1), i.e., glucagon-like peptide-1 receptor agonists (GLP-1 RAs), at therapeutic doses, are indicated as adjuncts to diet and exercise, to improve glycemic control in adults with type 2 diabetes mellitus (T2DM). In patients with T2DM, GLP-1 RAs not only affect improvements in impaired beta cell and alpha cell function, suppress appetite, and induce weight loss but also possess multiple cardiovascular protective properties that potentially have a beneficial impact on atherosclerotic cardiovascular disease (ASCVD) morbidity and mortality.
Recent Findings
Required to demonstrate CV safety, compared to standard-of-care antidiabetic therapies, GLP-1 RAs have revealed statistically significant non-inferiority (p < 0.001), among CV outcome trials (CVOTs) thus far completed. Once-daily liraglutide and once-weekly semaglutide demonstrated significant superiority (p = 0.01 and p = 0.02, respectively), reducing 3-point composite major adverse cardiovascular events (MACE) in extreme risk secondary prevention adults with T2DM. Once-weekly exenatide demonstrated only a non-significant (p = 0.06) favorable trend for CV superiority, possibly due to in-trial mishaps, including placebo drop-ins with other CV protective medications. The short half-life lixisenatide was neutral (p = 0.81) in reducing MACE, most likely due to ineffective once-daily dosing. Structural differences among GLP-1 mimetics and analogs may explain potency differences in both A1C reduction and weight loss that may parallel important cardiovascular protective properties of the GLP-1 RA class.
Summary
Significant superiority in reducing 3-point composite MACE in adults with T2DM with GLP-1 RAs has been limited to liraglutide and semaglutide. Careful attention to within-trial drop-in of cardioprotective antidiabetic agents assuring equipoise between placebo and investigational product groups might demonstrate significant MACE risk reduction with once-weekly exenatide. Maintenance of 24-h circulating levels, by an alternative administration method, may resurrect lixisenatide as a cardioprotective agent. Before a GLP-1 RA bioequivalence “class effect” claim for composite MACE risk reduction superiority can be fully discussed, we are obliged to wait for the pending results of CVOTs with other GLP-1 RAs, particularly albiglutide and dulaglutide, where steric hindrance may potentially inhibit full mimicry of pharmacologic GLP-1.
Similar content being viewed by others
Abbreviations
- ARR:
-
Absolute risk reduction
- BMI:
-
Body mass index
- CKD:
-
Chronic kidney disease
- CVD:
-
Cardiovascular disease
- CVOTs:
-
CV (safety) outcome trials
- DPP-4:
-
Dipeptidyl peptidase-4
- FDA:
-
Food and Drug Administration
- GLP-1 RAs:
-
Glucagon-like peptide-1 receptor agonists
- HR:
-
Hazard ratio
- MACE:
-
Major adverse cardiovascular events
- NF-MI:
-
Non-fatal myocardial infarction
- NNH:
-
Number needed to harm
- NNT:
-
Number needed to treat
- PEP:
-
Primary endpoint
- RCTs:
-
Randomized clinical trials
- RRR:
-
Relative risk reduction
- T2DM:
-
Type 2 diabetes mellitus
- UA:
-
Unstable angina
- ELIXA:
-
Evaluation of Lixisenatide in Acute Coronary Syndrome
- EXSCEL:
-
Effect of Once Weekly Exenatide On Cardiovascular Outcome in Type 2 Diabetes’
- FREEDOM-CVO:
-
Cardiovascular outcome safety study of ITCA 650, an injection-free exenatide osmotic mini-pump delivery system
- HARMONY:
-
Trial of the effect of albiglutide on major adverse cardiovascular (CV) events in patients with T2DM and established CV disease
- LEADER:
-
Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results
- REWIND:
-
Researching cardiovascular Events with a Weekly Incretin in Diabetes
- SUSTAIN-6:
-
Trial to Evaluate Cardiovascular and Other Long-term Outcomes with Semaglutide in Subjects with Type 2 Diabetes
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Kannel W, McGee DL. Diabetes and cardiovascular disease: the Framingham study. JAMA. 1979;241(19):2035–8.
Fruchart JC, Sacks F, Hermans MP, Assmann G, Brown WV, Ceska R, et al. The residual risk reduction initiative: a call to action to reduce residual vascular risk in patients with dyslipidemia. Am J Cardiol. 2008;102(10 Suppl):1K–34K.
Lin F-J, Tseng W-K, Yin W-H, Yeh H-I, Chen J-W, Wu C-C. Residual risk factors to predict major adverse cardiovascular events in atherosclerotic cardiovascular disease patients with and without diabetes mellitus. Sci Rep. 2017;7:9179. 1–9
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–53.
Reichard P, Nilsson BY, Rosenqvist U. The effect of long-term intensified insulin treatment on the development of microvascular complications of diabetes mellitus. N Engl J Med. 1993;329(5):304–9.
Aronson D. Hyperglycemia and the pathobiology of diabetic complications. Adv Cardiol. 2008;45:1–16.
Stefano GB, Challenger S, Kream RM. Hyperglycemia-associated alterations in cellular signaling and dysregulated mitochondrial bioenergetics in human metabolic disorders. Eur J Nutr. 2016;55(8):2339–45.
Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res. 2016;118(11):1808–29.
Stratton IM, Adler AI, Neil HAW, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321:405–12.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577–89.
DeFronzo RA. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes. 2009;58:773–95.
US Department of Human and Health Services Food and Drug Administration (2008) Guidance for industry: diabetes mellitus—evaluating cardiovascular risk in new antidiabetic therapies to treat type 2 diabetes. https://www.fda.gov/downloads/Drugs/Guidances/ucm071627.pdf
• Nauck MA, Meier JJ, Cavender MA, Abd El Aziz M, Drucker D. Cardiovascular actions and clinical outcomes with glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Circulation 2017;136(9):849–870. A recent extensive review of the potential cardiovascular benefits of DPP-4 inhibitors and pharmacological GLP-1 and its receptor agonists.
• Drucker DJ. The cardiovascular biology of glucagon-like peptide-1. Cell Metab. 2016;24(1):15–30. A recent extensive review of the potential cardiovascular benefits of pharmacological GLP-1 and its receptor agonists
Alhadeff AL, Rupprecht LE, Hayes MR. GLP-1 neurons in the nucleus of the solitary tract project directly to the ventral tegmental area and nucleus accumbens to control for food intake. Endocrinology. 2012;153(2):647–58.
Chang AM, Jakobsen G, Sturis J, Smith MJ, Bloem CJ, An B, et al. The GLP-1 derivative NN2211 restores beta-cell sensitivity to glucose in type 2 diabetic patients after a single dose. Diabetes. 2003;52(7):1786–91.
Farilla L, Bulotta A, Hirshberg B, Li Calzi S, Khoury N, Noushmehr H, et al. Glucagon-like peptide 1 inhibits cell apoptosis and improves glucose responsiveness of freshly isolated human islets. Endocrinol. 2003;144:5149–51.
Hyson D, Rutledge JC, Berglund L. Postprandial lipemia and cardiovascular disease. Curr Atheroscler Rep. 2003;5:437–44.
Meier JJ, Gethmann A, Götze O, Gallwitz B, Holst JJ, Schmidt WE, et al. Glucagon-like peptide 1 abolishes the postprandial rise in triglyceride concentrations and lowers levels of non-esterified fatty acids in humans. Diabetologia. 2006;49:452–8.
Matikainen N, Söderlund S, Björnson E, Pietiläinen K, Hakkarainen A, Lundbom N, et al. Liraglutide treatment improves postprandial lipid metabolism and cardiometabolic risk factors in humans with adequately controlled type 2 diabetes: a single-centre randomised controlled study. Diabetes Obes Metab. 2018; https://doi.org/10.1111/dom.13487.
Nordestgaard BG. Triglyceride-rich lipoproteins and atherosclerotic cardiovascular disease: new insights from epidemiology, genetics, and biology. Circ Res. 2016;118(4):547–63.
• Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JFE, Nauck MA, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311–22. The liraglutide LEADER CVOT results
• Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;37(5):1834–44. The semaglutide SUSTAIN-6 CVOT results
• Holman RR, Bethel MA, Mentz RJ, Thompson VP, Lokhnygina Y, Buse JB, et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2017;377:1228–39. The once-weekly exenatide EXSCEL CVOT results
• Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015;373:2247–57. The lixisenatide ELIXA CVOT results
A study to evaluate cardiovascular outcomes in patients with type 2 diabetes treated with ITCA 650. ClinicalTrials.gov website. clinicaltrials.gov/ct2/show/NCT01455896.
Fisher M, Petrie MC, Ambery PD, Donaldson J, Ye J, McMurray JJV. Cardiovascular safety of albiglutide in the Harmony programme: a meta-analysis. Lancet Diabetes Endocrinol. 2015;3(9):697–703.
• Green JB, Hernandez AF, D'Agostino RB, Granger CB, Janmohamed S, Jones NP, et al. Harmony outcomes: a randomized, double-blind, placebo-controlled trial of the effect of albiglutide on major cardiovascular events in patients with type 2 diabetes mellitus—rationale, design, and baseline characteristics. Am Heart J. 2018;12(203):30–8. The rationale and design of the albiglutide HARMONY CVOT
• Gerstein HC, Colhoun H, Dagenais GR, Diaz R, Lakshmanan M, Pais P, et al. Design and baseline characteristics of participants in the Researching cardiovascular Events with a Weekly INcretin in Diabetes (REWIND) trial on the cardiovascular effects of dulaglutide. Diabetes Obes Metab. 2018;20(1):42–9. The rationale and design of the dulaglutide REWIND CVOT
Jellinger PS, Handelsman Y, Rosenblit PD, Bloomgarden ZT, Fonseca VA, Garber AJ, et al. AACE/ACE guidelines: American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Supplement 2):1–87.
Baggio LL, Huang Q, Brown TJ, Drucker DJ. A recombinant human glucagon-like peptide (GLP)-1-albumin protein (albugon) mimics peptidergic activation of GLP-1 receptor-dependent pathways coupled with satiety, gastrointestinal motility, and glucose homeostasis. Diabetes. 2004;53(9):2492–500.
Jacobsen LV, Flint A, Olsen AK, Ingwersen SH. Liraglutide in type 2 diabetes mellitus: clinical pharmacokinetics and pharmacodynamics. Clin Pharmacokinet. 2016;55:657–72.
Lau J, Bloch P, Schäffer L, Pettersson I, Spetzler J, Kofoed J, et al. Discovery of the once-weekly glucagon-like peptide-1 (GLP-1) analogue semaglutide. J Med Chem. 2015;58(18):7370–80.
Blair HA, Keating GM. Albiglutide: a review of its use in patients with type 2 diabetes mellitus. Drugs. 2015;75:651–63.
Jimenez-Solem E, Rasmussen MH, Christensen M, Knop FK. Dulaglutide, a long-acting GLP-1 analog fused with an Fc antibody fragment for the potential treatment of type 2 diabetes. Curr Opin Mol Ther. 2010;12(6):790–7.
Koole C, Reynolds CA, Mobarec JC, Hick C, Sexton PM, Sakmar TP. Genetically-encoded photocrosslinkers determine the biological binding site of Exendin-4 in the N-terminal domain of the intact human glucagon-like peptide-1 receptor (GLP-1R). J Biol Chem. 2017;292:7131–44.
Werner U, Haschke G, Herling AW, Kramer W. Pharmacological profile of lixisenatide: a new GLP-1 receptor agonist for the treatment of type 2 diabetes. Regul Pept. 2010;164:58–64.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–28.
Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644–57.
Buse JB, Garber A, Rosenstock J, Schmidt WE, Brett JH, Videbæk N, et al. Liraglutide treatment is associated with a low frequency and magnitude of antibody formation with no apparent impact on glycemic response or increased frequency of adverse events: results from the Liraglutide Effect and Action in Diabetes (LEAD) trials. J Clin Endocrinol Metab. 2011;96:1695–702.
Knopp RH, D’Emden M, Smilde JG, for the ASPEN Study Group, et al. Efficacy and safety of atorvastatin in the prevention of cardiovascular end points in subjects with type 2 diabetes: the atorvastatin study for prevention of coronary heart disease endpoints in non-insulin-dependent diabetes mellitus (ASPEN). Diabetes Care. 2006;29:1478–85.
Sever PS, Dahlof B, Poulter NR, et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial-Lipid Lowering Arm (ASCOT-LLA): a multicentre randomized controlled trial. Lancet. 2003;361(9364):1149–58.
The ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in moderately hypercholesterolemic, hypertensive patients randomized to pravastatin vs usual care: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT-LLT). JAMA. 2002;288:2998–3007.
Keech A, Simes RJ, Barter P, for The FIELD study investigators, et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomized controlled trial. Lancet. 2005;366(9500):1849–61.
The AIM-HIGH Investigators, Boden WE, Probstfield JL, Anderson T, Chaitman BR, Desvignes-Nickens P, et al. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011;365:2255–67.
HPS2-THRIVE Collaborative Group. Effects of extended-release niacin with laropiprant in high-risk patients. NEJM. 2014;371(3):203–12.
Buse JB, Sesti G, Schmidt WE, Montanya E, Chang CT, Xu Y, et al. Switching to once-daily liraglutide from twice-daily exenatide further improves glycemic control in patients with type 2 diabetes using oral agents. Diabetes Care. 2010;33(6):1300–3.
Blevins T, Pullman J, Malloy J, Yan P, Taylor K, Schulteis C, et al. DURATION-5: exenatide once weekly resulted in greater improvements in glycemic control compared with exenatide twice daily in patients with type 2 diabetes. J Clin Endocrinol Metab. 2011;96(5):1301–10.
Buse JB, Nauck M, Forst T, Sheu WH, Shenouda SK, Heilmann CR, et al. Exenatide once weekly versus liraglutide once daily in patients with type 2 diabetes (DURATION-6): a randomised, open-label study. Lancet. 2013;381(9861):117–24.
Ahmann AJ, Capehorn M, Charpentier G, Dotta F, Henkel E, Lingvay I, et al. Efficacy and safety of once-weekly semaglutide versus exenatide ER in subjects with type 2 diabetes (SUSTAIN 3): a 56-week, open-label, randomized clinical trial. Diabetes Care. 2018;41(2):258–66.
Dungan KM, Povedano ST, Forst T, González JG, Atisso C, Sealls W, et al. Once-weekly dulaglutide versus once-daily liraglutide in metformin-treated patients with type 2 diabetes (AWARD-6): a randomised, open-label, phase 3, non-inferiority trial. Lancet. 2014;384(9951):1349–57.
Kayaniyil S, Lozano-Ortega G, Bennett HA, Johnsson K, Shaunik A, Grandy S, et al. A network meta-analysis comparing exenatide once weekly with other GLP-1 receptor agonists for the treatment of type 2 diabetes mellitus. Diabetes Ther. 2016;7:27–43.
Baggio LL, Huang Q, Cao X, Drucker DJ. An albumin-exendin-4 conjugate engages central and peripheral circuits regulating murine energy and glucose homeostasis. Gastroenterology. 2008;134:1137–47.
Pratley RE, Aroda VR, Lingvay I, Lüdemann J, Andreassen C, Navarria A, et al. Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): a randomised, open-label, phase 3b trial. Lancet Diabetes Endocrinol. 2018;6(4):275–86.
Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8:728–42.
Jia X, Alam M, Ye Y, Bajaj M, Birnbaum Y. GLP-1 receptor agonists and cardiovascular disease: a meta-analysis of recent cardiac outcome trials. Cardiovasc Drugs Ther. 2018;32:65–72.
Bethel MA, Patel RA, Merrill P, Lokhnygina Y, Buse JB, Mentz RJ, et al. Cardiovascular outcomes with glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes: a meta-analysis. Lancet Diabetes Endocrinol. 2018;6:105–13.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Yixing Li declares that he has no conflicts of interest.
Paul D. Rosenblit reports the following: Clinical Research Trial site for Amgen, AstraZeneca, Boehringer Ingelheim, Bristol-Myers-Squib, Glaxo SmithKline, Ionis, Lexicon, Novo Nordisk, and Sanofi; Speaking/Teaching Honoraria from Akcea, Amgen, Janssen, Novo Nordisk, and Merck; and Advisory Board Honoraria from Akcea, Amarin, Amgen, Novo Nordisk, and Sanofi-Regeneron.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Diabetes and Cardiovascular Disease
Rights and permissions
About this article
Cite this article
Li, Y., Rosenblit, P.D. Glucagon-Like Peptide-1 Receptor Agonists and Cardiovascular Risk Reduction in Type 2 Diabetes Mellitus: Is It a Class Effect?. Curr Cardiol Rep 20, 113 (2018). https://doi.org/10.1007/s11886-018-1051-2
Published:
DOI: https://doi.org/10.1007/s11886-018-1051-2