Abstract
Objectives
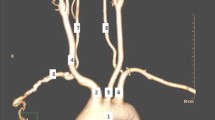
The present study analyzed the prevalence of variations of the aortic arch branching in Japanese population, comparing patients with aortic arch disease with healthy controls.
Methods
Between from October 1999 and December 2015, 815 Japanese patients with aortic arch disease defined as aortic arch aneurysm (diameter ≥ 45 mm) and aortic dissection (group A) underwent aortic arch surgery in our institution. As a control group, 1506 traumatic screened patients were enrolled (group C).
Results
Aortic arch anomaly was diagnosed in 140 patients (17.2%) in the group A and in 222 patients (14.7%) in the group C (p = 0.125). Significant differences were found in the incidence of aberrant subclavian artery (A: 14 patients, 1.7%, vs. C: 8 patients, 0.5%, p = 0.006). Significantly more patients with aortic arch aneurysm in the group A had anomalies of the aortic arch compared with the group C (p = 0.009), including bovine aortic arch (p = 0.049) and aberrant subclavian artery (p < 0.001). In term of aneurysm location, bovine arch was detected in more patients with proximal arch aneurysm (15.7%, p = 0.043), whereas aberrant subclavian artery was in more patients with distal location (3.7%, p < 0.001). No difference was found in aortic arch anomaly in patients with acute or chronic dissection.
Conclusion
Aberrant subclavian artery was a significant maker of aortic arch disease in Japanese populations. Bovine arch was a risk maker of proximal arch aneurysm, and aberrant subclavian artery was a risk factor of distal arch aneurysm.


Similar content being viewed by others
References
Okita Y. Surgery for thoracic aortic disease in japan: Evolving strategies toward the growing enemies. Gen Thorac Cardiovasc Surg. 2015;63(4):185–96.
Natsis KI, Tsitouridis IA, Didagelos MV, Fillipidis AA, Vlasis KG, Tsikaras PD. Anatomical variations in the branches of the human aortic arch in 633 angiographies: clinical significance and literature review. Surg Radiol Anat. 2009;31(5):319–23.
Jakanani GC, Adair W. Frequency of variations in aortic arch anatomy depicted on multidetector CT. Clin Radiol. 2010;65(6):481–7.
Müller M, Schmitz BL, Pauls S, et al. Variations of the aortic arch—a study on the most common branching patterns. Acta Radiol. 2011;52(7):738–42.
Celikyay ZRY, Koner AE, Celikyay F, Denız C, Acu B, Firat MM. Frequency and imaging findings of variations in human aortic arch anatomy based on multidetector computed tomography data. Clin Imaging. 2013;37(6):1011–9.
Ergun E, Simsek B, Kosar PN, Yilmaz BK, Turgut AT. Anatomical variations in branching pattern of arcus aorta: 64-slice CTA appearance. Surg Radiol Anat. 2013;35(6):503–9.
Karacan A, Turkvatan A, Karacan K. Anatomical variations of aortic arch branching: evaluation with computed tomographic angiography. Cardiol Young. 2014;24(3):485–93.
Dumfarth J, Chou AS, Ziganshin BA, et al. Atypical aortic arch branching variants: a novel marker for thoracic aortic disease. J Thorac Cardiovasc Surg. 2015;149(6):1586–92.
Moorehead PA, Kim AH, Miller CP, Kashyap TV, Kendrick DE, Kashyap VS. Prevalence of bovine aortic arch configuration in adult patients with and without thoracic aortic pathology. Ann Vasc Surg. 2016;30:132–7.
Keith A. The modes of origin of the carotid and subclavian arteries from the arch of the aorta in some of the higher primates. J Anat Physiol. 1895;29(Pt 3):453.
Adachi B, Hasebe K. Das arteriensystem der japaner. Kaiserlich-japanische Universität zu Kyoto. In: kommission bei. Maruzen Company, Kyoto; 1928.
De Garis CF, Black IH, Riemenschneider EA. Patterns of the aortic arch in american white and negro stocks, with comparative notes on certain other mammals. J Anat. 1933;67(Pt 4):599.
Williams GD, Edmonds HW. Variations in the arrangement of the branches arising from the aortic arch in American whites and Negroes (a second study). Anat Rec. 1935;62(2):139–46.
McDonald JJ, Anson BJ. Variations in the origin of arteries derived from the aortic arch, in American whites and Negroes. Am J Phys Anthropol. 1940;27(1):91–107.
Shuford W, Sybers R, Milledge R, Brinsfield D. The cervical aortic arch. Am J Roentgenol. 1972;116(3):519–27.
Haughton V, Rosenbaum A. The normal and anomalous aortic arch and brachiocephalic arteries. Radiol Skull Brain. 1974;2:1145–63.
Ehren H, Wells TR, Landing BH. Association of common origin of the carotid arteries with anomalous origin of the left coronary artery from the pulmonary artery. Pediatr Pathol. 1985;4(1–2):59–66.
Hornick M, Moomiaie R, Mojibian H, et al. ‘Bovine’ aortic arch—a marker for thoracic aortic disease. Cardiology. 2012;123(2):116–24.
Malone CD, Urbania TH, Crook SE, Hope MD. Bovine aortic arch: a novel association with thoracic aortic dilation. Clin Radiol. 2012;67(1):28–31.
Wanamaker KM, Amadi CC, Mueller JS, Moraca RJ. Incidence of aortic arch anomalies in patients with thoracic aortic dissections. J Card Surg. 2013;28(2):151–4.
Kirby ML, Waldo KL. Role of neural crest in congenital heart disease. Circulation. 1990;82:332–40.
Katz JC, Chakravarti S, Ko HH, et al. Common origin of the innominate and carotid arteries: prevalence, nomenclature, and surgical implications. J Am Soc Echocardiogr. 2006;19:1446–8.
Tanaka A, Milner R, Ota T. Kommerell’s diverticulum in the current era: a comprehensive review. Gen Thorac Cardiovasc Surg. 2015;63(5):245–59.
Kim KM, Cambria RP, Isselbacher EM, et al. Contemporary surgical approaches and outcomes in adults with Kommerell diverticulum. Ann Thorac Surg. 2014;98(4):1347–54.
Murzi M, Mariani M, Tiwari KK, et al. Aberrant right subclavian artery aneurysm in coexistence with a common carotid trunk. Ann Thorac Surg. 2009;88(1):e8.
Park N, Hashmi ZA, Kim J, et al. Aortic arch aneurysm associated with arch vessel anomalies: truncus bicaroticus and retroesophageal right subclavian artery. J Card Surg. 2009;24(4):476–9.
Ikeno Y, Matsumori M, Yokawa K, et al. A case of type A acute aortic dissection with a common carotid trunk. Gen Thorac Cardiovasc Surg. 2018. https://doi.org/10.1007/s11748-018-0953-7
Poellinger A, Lembcke AE, Elgeti T, Filimonov S, Enzweiler CN. The cervical aortic arch a rare vascular anomaly. Circulation. 2008;117(20):2716–7.
Dasari TW, Paliotta M. Cervical aortic arch. N Engl J Med. 2014;371(26):e38.
Pham T, Martin C, Elefteriades J, Sun W. Biomechanical characterization of ascending aortic aneurysm with concomitant bicuspid aortic valve and bovine aortic arch. Acta Biomater. 2013;9:7927–36.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yuki Ikeno has no conflict of interest. Yutaka Koide has no conflict of interest. Takashi Matsueda has no conflict of interest. Katsuhiro Yamanaka has no conflict of interest. Takeshi Inoue has no conflict of interest. Satoshi Ishihara has no conflict of interest. Shinichi Nakayama has no conflict of interest. Hiroshi Tanaka has no conflict of interest. Koji Sugimoto has no conflict of interest. Yutaka Okita has no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ikeno, Y., Koide, Y., Matsueda, T. et al. Anatomical variations of aortic arch vessels in Japanese patients with aortic arch disease. Gen Thorac Cardiovasc Surg 67, 219–226 (2019). https://doi.org/10.1007/s11748-018-1001-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11748-018-1001-3