Abstract
Objective
To study the effects and possible underlying mechanism of Qufeng Tongluo Prescription (祛风通络方, QFTL) on the regulation of mesangial cells (MCs) proliferation and apoptosis.
Methods
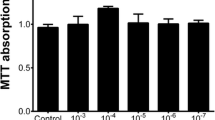
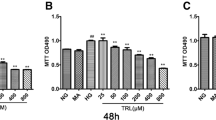
The MCs used in this experiment have undergone five passages induced by lipopolysaccharide (LPS). Changes in the proliferation, apoptosis, cell cycle regulatory proteins and mRNA expression levels of the MCs after administration of Benazepril or QFTL were measured by methyl thiazolyl tetrazolium (MTT) reduction assay, flow cytometry, Western blot and quantitative real-time polymerase chain reaction (qRT-PCR), respectively.
Results
The addition of Benazepril or QFTL serum inhibited LPS-induced MC proliferation after treatment for 24, 48 and 72 h, respectively (P<0.05 or P<0.01). Moreover, the inhibitory effect is more significant in the QFTL group at 48 h (P<0.05). Compared with the control group, LPS-induced cell proliferation decreased the number of cells in G1 phase versus cells in S and G2/M phases, while the addition of QFTL and Benazepril serum increased the ratio of cells at G1 phase (P<0.05 or P<0.01) to cells at S phase (P<0.01), implicating the cell cycle inhibition effect exerted by QFTL. LPS decreased the level of MC apoptosis, compared with the control group (P<0.05), while QFTL and Benazepril serum increased the level of MC apoptosis (P<0.01). Moreover, the difference between the QFTL group and the Benazepril group was statistically significant (P<0.01). Compared with the control group, the protein and mRNA expression levels of cylinD1, cyclin dependent kinase 2 (CDK2) and p21 were significantly increased (P<0.05 or P<0.01), p27 was decreased but with no statistical significance (P>0.05); After being treated with QFTL and Benazepril serum, the protein and mRNA expression levels of cylinD1, CDK2, p21 were decreased and p27 increased significantly (P<0.05 or P<0.01); Compared with the Benazepril group, QFTL show better effects on protein and mRNA expression levels of cylinD1, CDK2 (P<0.05 or P<0.01) and p21 protein expression (P<0.05).
Conclusion
QFTL inhibits MCs proliferation, promotes MCs apoptosis through an underlying mechanism of down-regulating the protein and mRNA expression levels of cylinD1, CDK2, p21 and up-regulation of the expression level of p27.
Similar content being viewed by others
References
Patel K, Harding P, Haney LB, Glass WF. Regulation of the mesangial cell myofibroblast phenotype by actin polymerization. J Cell Physiol 2003;195:435–445.
Herrera GA. Plasticity of mesangial cells: a basis for understanding pathological alterations. Ultrastruct Pathol 2006;30:471–479.
Schocklmann HO, Lang S, Sterzel RB. Regulation of mesangial cell proliferation. Kidney Int 1999;56:1199–1207.
Striker U, Doi T, Elliot S, Striker GE. The contribution of glomerular cells to progressive glomeruloscierosis. Semin Nephrol 1989;19:318–328.
Jia H, Qi X, Fang S, Jin Y, Han X, Wang Y, et al. Carnosine inhibits high glucose-induced mesangial cell proliferation through mediating cell cycle progression. Regul Pept 2009;154(1-3):69–76.
Ka SM, Cheng CW, Shui HA, Wu WM, Chang DM, Lin YC, et al. Mesangial cells of lupus-prone mice are sensitive to chemokine production. Arthritis Res Ther 2007;9(4):R67.
Tamouza H, Vende F, Tiwari M, Arcos-Fajardo M, Vrtovsnik F, Benhamou M, et al. Transferring receptor engagement by polymeric IgA1 induces receptor expression and mesangial cell proliferation: role in IgA nephropathy. Contrib Nephrol 2007;157:144–147.
Xue Y, Tie CR, Li J, Tian T, Li QX. Ligustrazine inhibits lipopolysaccharide-induced proliferation by affecting p27, Bcl-2 expression in rat mesangial cells. Eur J Pharmacol 2011;665(1-3):8–12.
Funk JO, Galloway DA. Inhibiting CDK inhibitors: New lessons from DNA tumor viruses. Trends Biochem Sci 1998;23:337–341.
Zhang YL, Shi YH, Liu YL, Dong H, Liu MD, Li Y, et al. Growth pattern switch of renal cells and expression of cell cycle related proteins at the early stage of diabetic nephropathy. Biochem Biophys Res Commun 2007;363:159–164.
Wu XL, Sun WS, Zhang WG, Qiao CL. Pharmacodynamic study of Tongluo Treatment on glomerular sclerosis in rats. J Hebei Univ (Chin) 2007;28(1):12–14.
Wu XL, Sun WS, Zhang WG, Wang Z. Mechanism of the effect of Tongluo Treatment against glomerular sclerosis in rats. J South Med Univ (Chin) 2008;28:1198–1201.
Schlöndorff D, Banas B. The mesangial cell revisited: no cell is an island. J Am Soc Nephrol 2009;20:1179–1187.
Lee HS, Song CY. Differential role of mesangial cells and podocytes in TGF-beta-induced mesangial matrix synthesis in chronic glomerular disease. Histol Histopathol 2009;24:901–908.
Picken MM. The role of mesangial homeostasis in glomerular injury progression: hope for mesangial sclerosis reversal. Kidney Int 2009;75:574–576.
Andrea CS, Bruce MH. The regulation of mesangial cell proliferation. Nephron Exp Nephrol 2008;108(4):74–79.
Stockand JD, Sansom SC. Glomerular mesangial cells: electrophysiology and regulation of contraction. Physiol Rev 1998;78:723–744.
Olivetti G, Anversa P, Rigamonti W, Vitali-Mazza L, Loud AV. Morphometry of the renal corpuscle during normal postnatal growth and compensatory hypertrophy: a light microscope study. J Cell Biol 1977;75(2 Pt 1):573–585.
Schocklmann HO, Lang S, Sterzel RB. Regulation of mesangial cell proliferation. Kidney Int 1999;56:1199–1207.
Lang F, Shumilina E, Ritter M, Gulbins E, Vereninov A, Huber SM. Ion channels and cell volume in regulation of cell proliferation and apoptotic cell death. Contrib Nephrol 2006;152:142–160.
Watson S, Cailhier JF, Hughes J, Savill J. Apoptosis and glomerulonephritis. Curr Dir Autoimmun 2006;9:188–204.
Ueda N, Kaushal GP, Shah SV. Apoptotic mechanisms in acute renal failure. Am J Med 2000;108:403–415.
Rana A. Sathyanarayana P, Lieberthal W. Role of apoptosis of renal tubular cells in acute renal failure: therapeutic implications. Apoptosis 2001;6:83–102.
Shankland SJ, Wolf G. Cell cycle regulatory proteins in renal disease: role in hypertrophy, proliferation, and apoptosis. Am J Physiol Renal Physiol 2000;278:F515–F529.
Shankland SJ. Cell-cycle control and renal disease. Kidney Int 1997;52:294–297.
van der Boog PJ, van Kooten C, de Fijter JW, Daha MR. Role of macromolecular IgA in IgA nephropathy. Kidney Int 2005;67:813–821.
Xu LX, Zhao MH. Aberrantly glycosylated IgA1 are closed associated with pathologic phenotype of IgA nephropathy. Kidney Int 2005;68:167–172.
Ding JX, Xu LX, Lu JC, Zhao MH, Zhang H, Wang HY. Aberrant sialylation of serum IgA1 was associated with prognosis of patients with IgA nephropathy. Clin Immunol 2007;125:268–274.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 30801504, No. 30901926, No. 81100530 and No. 81070590)
Rights and permissions
About this article
Cite this article
Wu, Xl., An, P., Ye, By. et al. Qufeng Tongluo Prescription (祛风通络方) inhibits mesangial cell proliferation and promotes apoptosis through regulating cell cycle progression. Chin. J. Integr. Med. 19, 927–934 (2013). https://doi.org/10.1007/s11655-013-1655-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-013-1655-8