Abstract
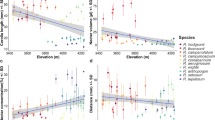
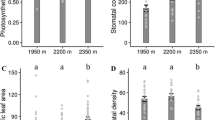
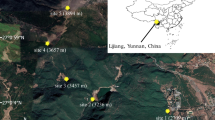
Phenotypic plasticity and/or pollinator-mediated selection may be responsible for the changes in floral traits of plants when they are forced to live in new conditions. Although the two events could be independent, we hypothesized that phenotypic plasticity in floral traits might help to coordinate plant-pollinator interactions and enhance plant reproductive success in changing habitats. To test this hypothesis, we investigated floral traits and pollination on three natural populations of a lousewort (Pedicularis siphonantha) ranging at different elevations, as well as two downward transplanted populations in Shangeri-La County and Deqin County, northwest Yunnan, China. The results indicated that floral traits, i.e. phenology, longevity, display size, corolla tube length and pollen production differed significantly among populations. Moreover, or the two transplanted populations, floral traits diverged from their original populations, but converged to their host populations. Although the phenotypic plasticity in floral traits might be a rapid response to abiotic factor such as warmer environment, the changes in floral traits were found to be well adapted to pollination environment of the host population. Compared with plants of their original habitats in higher elevation, the transplanted individuals advanced flowering time, shortened flower longevity, reduced floral display size and pollen production, received higher visiting frequency and yielded more seeds. These findings suggested that phenotypic plasticity of floral traits might help plants adjust their resource allocation strategy between preand post-pollination stages in response to harsh or temperate conditions, which might correspondingly meet a pollinator-poor or hyphen rich environment. This would be beneficial for the widely-distributed species to adapt to various environmental changes.
Similar content being viewed by others
References
Anderson JT, Gezon ZJ (2015) Plasticity in functional traits in the context of climate change: a case study of the subalpine forb Boechera stricta (Brassicaceae). Global Change Biology 21(4): 1689–1703. https://doi.org/10.1111/gcb.12770
Arroyo MTK, Dudley LS, Jespersen G, et al. (2013) Temperature-driven flower longevity in a high-alpine species of Oxalis influences reproductive assurance. New Phytologist 200(4): 1260–1268. https://doi.org/10.1111/nph.12443
Arroyo MTK, Munoz MS, Henriquez C, et al. (2006) Erratic pollination, high selfing levels and their correlates and consequences: an altitudinally widespread above-tree-line species in the high Andes of Chile. Acta Oecologica 30(2): 248–257. https://doi.org/10.1016/j.actao.2006.05.006
Benadi G, Hovestadt T, Poethke HJ, Bluthgen N (2014) Specialization and phenological synchrony of plant-pollinator interactions along an altitudinal gradient. Journal of Animal Ecology 83(3): 639–650. https://doi.org/10.1111/1365-2656.12158
Brys R, Jacquemyn H (2012) Effects of human-mediated pollinator impoverishment on floral traits and mating patterns in a short-lived herb: an experimental approach. Functional Ecology 26(1): 189–197. https://doi.org/10.1111/j.1365-2435.2011.01923.x
Cruden RW (1977) Pollen-ovule ratios: a conservative indicator of breeding systems in flowering plants. Evolution 31(1): 32–46. https://doi.org/10.2307/2407542
Dai WK, Kadiori EL, Wang QF, Yang CF (2017) Pollen limitation, plasticity in floral traits, and mixed mating system in an alpine plant Pedicularis siphonantha (Orobanchaceae) from different altitudes. Journal of Systematics and Evolution 55(9):192–199. https://doi.org/10.1111/jse.12240
Dorken ME, Pannell JR (2009) Hermaphroditic sex allocation evolves when mating opportunities change. Current Biology 19(6): 514–517. https://doi.org/10.1016/j.cub.2009.03.042
Eaton DAR, Fenster CB, Hereford J, et al. (2012) Floral diversity and community structure in Pedicularis (Orobanchaceae). Ecology 93(sp8): S182–S194. https://doi.org/10.1890/11-0501.1
Eckert CG, Kalisz S, Geber MA, et al. (2010) Plant mating systems in a changing world. Trends in Ecology and Evolution 25(1): 35–43. https://doi.org/10.1016/j.tree.2009.06.013
Ensslin A, Fischer M (2015) Variation in life-history traits and their plasticities to elevational transplantation among seed families suggests potential for adaptative evolution of 15 tropical plant species to climate change. American Journal of Botany 102(8): 1371–1379. https://doi.org/10.3732/ajb.1400518
Garcia-Camacho R, Totland O (2009) Pollen limitation in the alpine: a meta-analysis. Arctic Antarctic and Alpine Research 41(1): 103–111. https://doi.org/10.1657/1523-0430-41.1.103
Grassein, F, Lavorel S, Till-Bottraud I (2013) The importance of biotic interactions and local adaptation for plant response to environmental changes: Field evidence along an elevational gradient. Global Change Biology 20(5): 1452–1460. https://doi.org/10.1111/gcb.12445
Huang SQ, Fenster CB (2007) Absence of long-proboscid pollinators for long-corolla-tubed Himalayan Pedicularis species: Implications for the evolution of corolla length. International Journal of Plant Science 168(3): 325–331. https://doi.org/10.1086/510209
Kim E, Donohue K (2013) Local adaptation and plasticity of Erysimum capitatum to elevation: its implications for responses to climate change. Journal of Ecology 101(3): 796–805. https://doi.org/10.1111/1365-2745.12077
Michalski SG, Durka W (2009) Pollination mode and life form strongly affect the relation between mating system and pollen to ovule ratios. New Phytologist 183(2): 470–479. https://doi.org/10.1111/j.1469-8137.2009.02861.x
Nicotra AB, Atkin OK, Bonser SP, et al. (2010) Plant phenotypic plasticity in a changing climate. Trends in Plant Science 15(12): 684–692. https://doi.org/10.1016/j.tplants.2010.09.008
Preite V, Stocklin J, Armbruster GFJ, Scheepens JF (2015) Adaptation of flowering phenology and fitness-related traits across environmental gradients in the widespread Campanula rotundifolia. Evolutionary Ecology 29(2): 249–267. https://doi.org/10.1007/s10682-015-9754-y
Queller DC (1984) Pollen-ovule ratios and hermaphrodite sexual allocation strategies. Evolution 38: 1148–1151. https://doi.org/10.2307/2408447
Scheepens JF, Stocklin J (2013) Flowering phenology and reproductive fitness along a mountain slope: maladaptive responses to transplantation to a warmer climate in Campanula thyrsoides. Oecologia 171(3): 679–691. https://doi.org/10.1007/s00442-012-2582-7
Steiner BL, Armbruster GFJ, Scheepens JF, Stöcklin J (2012) Distribution of bulbil-and seed-producing plants of Poa alpina (Poaceae) and their growth and reproduction in common gardens suggest adaptation to different elevations. American Journal of Botany 99(12): 2035–2044. https://doi.org/10.3732/ajb.1200213
Thomann M, Imbert E, Cheptou PO (2015) Is rapid evolution of reproductive traits in Adonis annua consistent with pollinator decline? Acta Oecologica 69: 161–166. https://doi.org/10.1016/j.actao.2015.10.007
Totland O (2001) Environment-dependent pollen limitation and selection on floral traits in an alpine species. Ecology 82(8): 2233–2244. https://doi.org/10.2307/2680228
Tsoong PC (1955) A new system for the genus Pedicularis. Acta Phytotax Sin 4: 71–147.
Yang CF, Guo YH (2004) Pollen size-number trade-off and pollen-pistil relationships in Pedicularis (Orobanchaceae). Plant Systematics and Evolution 247(3): 177–185. https://doi.org/10.1007/s00606-004-0165-8
Yang CF, Sun SG, Guo YH (2005) Resource limitation and pollen source (self and outcross) affecting seed production in two louseworts, Pedicularis siphonantha and P. longiflora (Orobanchaceae). Botanical Journal of Linnean Society 147(1): 83–89. https://doi.org/10.1111/j.1095-8339.2005.00363.x
Yang CF, Wang QF (2015) Nectarless flowers with deep corolla tubes in Pedicularis: Does long pistil length provide an arena for male competition? Botanical Journal of Linnean Society 179(3): 526–532. https://doi.org/10.1111/boj.12331
Yu WB, Li DZ, Wang H (2012) Highly efficient pollination by bumblebees ensures seed production in Pedicularis lachnoglossa (Orobanchaceae), an early-flowering Himalayan plant. Journal of Systematics and Evolution 50(3): 218–226. https://doi.org/10.1111/j.1759-6831.2012.00180.x
Zhao ZG, Wang YK (2015) Selection by pollinators on floral traits in generalized Trollius ranuncuoides (Ranunculaceae) along Altitudinal Gradients. Plos One 10: e0118299. https://doi.org/10.1371/journal.pone.0118299
Acknowledgements
The authors are grateful to JIN Xiao-fang, WU Chui-xu, YANG Jian, YANG Jun and YE Zhong-ming for their assistance in field, LIAO Kuo, JIN Xiao-fang and YE Zhong-ming for helpful discussion and suggestion. This work was supported by the National Natural Science Foundation of China (Grant No. 31370263 and 31770255).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Dai, WK., Amboka, G.M., Kadiori, E.L. et al. Phenotypic plasticity of floral traits and pollination adaption in an alpine plant Pedicularis siphonantha D. Don when transplanted from higher to lower elevation in Eastern Himalaya. J. Mt. Sci. 14, 1995–2002 (2017). https://doi.org/10.1007/s11629-017-4481-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11629-017-4481-1