Abstract
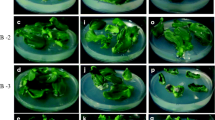
Enhanced numbers of multiple shoots were induced from shoot tip explants of cucumber. The effects of amino acids (leucine, isoleucine, methionine, threonine, and tryptophan) and polyamines (spermidine, spermine, and putrescine) along with benzyladenine (BA) on multiple shoot induction were investigated. A Murashige and Skoog (MS) medium containing a combination of BA (4.44 μM), leucine (88 μM), and spermidine (68 μM) induced the maximum number of shoots (36.6 shoots per explant) compared to BA (4.44 μM) alone or BA (4.44 μM) with leucine (88 μM). The regenerated shoots were elongated on the same medium. Elongated shoots were transferred to the MS medium fortified with BA (4.44 μM), leucine (88 μM), and putrescine (62 μM) for root induction. Rooted plants were hardened and successfully established in soil with a 90% survival rate.
Similar content being viewed by others
References
Alabadi A.; Aguero M. S.; Perez-Amador M. A.; Carbonell J. Arginase, arginine decarboxylase, ornithine decarboxylase and polyamines in tomato ovaries. Changes in unpollinated ovaries and parthenocarpic fruits induced by auxin or gibberellin. Plant Physiol. 112: 1237–1244; 1996.
Altman A. Retardation of radish leaf senescence by polyamines. Physiol. Plant. 54: 189–193; 1982.
Altman A.; Levin N. Interaction of polyamines and nitrogen nutrition in plants. Physiol. Plant. 89: 653–658; 1993.
Aziz H. A.; McCown B. H. Hormonal response of shoot and callus cultures of cucumber (Cucumis sativus L.). Sci. Hortic. 20: 540; 1985.
Bagni N.; Torrigiani P. Polyamines: A new class of growth substances. In: KarssenC. M.; Van LoonL. C.; VreugdenhilD. (eds) Progress in plant growth regulation. Kluwer, Dordrecht, The Netherlands, pp 264–275; 1982.
Bais H. P.; Ravishankar G. A. Role of polyamines in the ontogeny and their biotechnological applications. Plant Cell Tiss. Org. Cult. 69: 1–34; 2002.
Bastola D. R.; Minocha S. C. Increased putrescine biosynthesis through transfer of mouse ornithine cDNA in carrot promotes somatic embryogenesis. Plant Physiol. 109: 63–71; 1995.
Basu A.; Sethi U.; Guha-Mukherjee S. Regulation of cell proliferation and morphogenesis by aminoacids in Brassica tissue cultures and its correlation with threonine deaminase. Plant Cell Rep. 8: 333–335; 1989.
Chi G. L.; Lin W. S.; Lee J. E. E.; Pua E. C. Role of polyamines on de novo shoot morphogenesis from cotyledons of Brassica campestris spp. Pekinensis (Lour) Olsson in vitro. Plant Cell Rep. 13: 323–329; 1994.
Chowdhry N.; Tyagi A. K.; Maheshwari N.; Maheshwari S. C. Effect of L-proline and L-tryptophan on somatic embryogenesis and plantlet regeneration of rice (Oryza sativa L. cv. Pusa 169). Plant Cell Tiss. Org. Cult. 32: 357–361; 1993.
Compton M. E.; Gray D. J.; Elmstrom G. W. A simple protocol for micropropagating diploid and tetraploid watermelon using shoot tip explants. Plant Cell Tiss. Org. Cult. 3: 211–217; 1993.
Compton M. E.; Pierson B.; Staub J. E. Micropropagation for recovery of Cucumis hystrix. Plant Cell Tiss. Org. Cult. 64: 63–67; 2001.
Couee I.; Hummel I.; Sulmon C.; Gouesbet G.; Amrani A. E. Involvement of polyamines in root development. Plant Cell Tiss. Org. Cult. 76: 1–10; 2004.
Davies P. J. The plant hormones: Their nature, occurrence, and functions. In: DaviesP. J. (ed) Plant hormones and their role in plant growth and development. Martinus Nijhoff, Boston, MA, pp 1–12; 1987.
De Klerk G. J.; Hanecakova J.; Jasik J. The role of cytokinins in rooting of stem slices cut from apple microcuttings. Plant Biosys. 135: 79–84; 2001.
Desai H. V.; Mehta A. R. Changes in polyamine levels during shoot formation, root formation and callus induction in cultured Passiflora leaf discs. J. Plant. Physiol. 119: 45–53; 1985.
Dey O. K. S. M.; Kalia S.; Ghose S.; Guha-Mukherjee S. Biochemical basis of differentiation in plant tissue culture. Curr. Sci. 74: 591–596; 1998.
Eapen S.; George L. Plant regeneration from leaf discs of peanut and pigeonpea: Influence of benzyladenine, indoleacetic acid and indoleacetic acid-amino acid conjugates. Plant Cell Tiss. Org. Cult. 353: 223–227; 1993.
Flores H. E.; Filner P. Polyamine catabolism in higher plants: Characterization of pyrroline dehydrogenase. Plant Growth Regul. 3: 277–291; 1985.
Galston A. W. Polyamines as modulators of plant development. Bioscience. 33: 382–388; 1983.
Galston A. W.; Kaur-Sawhney R. Polyamines as endogenous growth regulators. In: DaviesP. J. (ed) Plant hormones and their role in plant growth and development. Martinus Nijhoff, Dordrecht, pp 280–295; 1987.
Galston A. W.; Kaur-Sawhney R. Polyamines in plant physiology. Plant Physiol. 94: 406–410; 1990.
Galston A. W.; Kaur-Sawhney R.; Altabella T.; Tiburcio A. F. Plant polyamines in reproductive activity and response to abiotic stress. Bot. Acta. 110: 197–207; 1997.
Geneve R. L.; Hackett W. P. Ethylene evolution and endogenous polyamine levels during adventitious root formation in English ivy. In: FloresH. E.; ArtecaR. N.; ShanonJ. C. (eds) Polyamine and ethylene: Biochemistry, physiology and interactions. Amer. Soc. Plant Physiologists, Rockville, pp 332–334; 1990.
Gomez K. A.; Gomez K. A. Statistical procedures for agricultural research with emphasis of rice. International Rice Research Institute, Los Banos, Phillipines; 1976.
Hare P. D.; Cress W. A.; van Staden J. The effects of exogenous proline and proline analogues on in vitro shoot organogenesis in Arabidopsis. Plant Growth Reg. 342: 203–207; 2001.
Hoagland, D. R.; Arnon, D. I. The water culture method for growing plants without soil. California Agric. Exp. Sta. Bull., no. 347; 1950.
John S. J.; Guha-Mukherjee S. In: TewaryK. K.; SinghalG. S. (eds) Plant molecular biology and biotechnology. Narosa, New Delhi, pp 17–28; 1997.
Kaur-Sawhney R.; Tiburcio A. F.; Galston A. W. Polyamine-mediated control of organogenesis in thin layer explants of tobacco. Plant Physiol. 80: 37; 1986.
Li Z. L.; Burritt D. J. Changes in endogenous polyamines during the formation of somatic embryos from isogenic lines of Dactylis glomerata L. with different regenerative capacities. Plant Growth Regul. 40: 65–74; 2003.
Martin-Tanguy J. Metabolism and function of polyamines in plants: Recent development (new approaches). Plant Growth Regul. 34: 135–148; 2001.
Misra A. K.; Bhatnagar S. P. Direct shoot regeneration from the leaf explant of cucumber (Cucumis sativus L.). Phytomorphology. 45: 47–55; 1995.
Murashige T.; Skoog F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Physiol. 15: 473–497; 1962.
Muyuan Z.; Abing X.; Miaobao Y.; Chunnong H.; Zhilong Y.; Linji W.; Jianjun Y. Effects of amino acids on callus differentiation in barley anther culture. Plant Cell Tiss. Org. Cult. 22: 201–204; 1990.
Nag S.; Saha K.; Chowdhuri M. Role of auxin and polyamine in adventitious root formation at the base of mung bean cuttings. Indian J. Plant Physiol. 4: 247–255; 1999.
Rao A. M.; Padma Sree K.; Kavi Kishor P. B. Enhanced plant regeneration in grain and sweet sorghum by asparagine, proline and cefotaxime. Plant Cell Rep. 15: 72–75; 1995.
Ronchi V. N.; Caligo M. A.; Nizzolini M.; Luccarini G. Stimulation of carrot somatic embryogenesis by proline and serine. Plant Cell Rep. 3: 210–214; 1984.
Scaramagli S.; Biondi S.; Capitani P.; Gerola P.; Altamura M. M.; Torrigiani P. T. Polyamines conjugate levels and ethylene biosynthesis: Inverse relationship with vegetative bud formation in tobacco thin layers. Physiol. Plant. 105: 367–376; 1999.
Scholten H. J. Effect of polyamines on the growth and development of some horticultural crops in micropropagation. Sci. Hortic. 77: 83–88; 1998.
Selvaraj N.; Vengadesan G.; Vasudevan A.; Prem Anand R.; Ramesh Anbazhagan V.; Ganapathi A. Micropropagation of Cucumis sativus L. from field grown plants. In: MaynardD. N. (ed) Proceedings of the Cucurbitaceae Conference. Acta Hortic., Belgium, pp 149–156; 2002.
Sen J.; Kalia S.; Guha-Mukherjee S. Level of endogenous free amino acids during various stages of culture of Vigna mungo (L.) Hepper—somatic embryogenesis, organogenesis and plant regeneration. Curr. Sci. 82: 429–433; 2002.
Smith T. A. Polyamines. Ann. Rev. Plant Physiol. 36: 117–143; 1985.
Tang W.; Newton R. Polyamines promote root elongation and growth by increasing root cell division in regenerated Virginia pine (Pinus virginiana Mill.). Plant Cell Rep. 24: 581–589; 2005.
Tanimoto S.; Matsubara Y.; Ishioka N. Significance of spermidine in the initiation of adventitious buds in stem segments of Torenia. Plant Cell Physiol. 35: 1071–1077; 1994.
Tarenghi E.; Carre M.; Martin-Tanguy J. Effects of inhibitors of polyamine biosynthesis and of polyamines on strawberry microcutting growth and development. Plant Cell Tiss. Org. Cult. 42: 47–55; 1995.
Tian C. E.; Li R. G.; Guan H. Relationship between polyamines and morphogenesis in cotyledons of Cucumis melo L. cultured in vitro. Acta Bot. Sin. 36: 219–222; 1994.
Tiburcio A. F.; Kaur-Sawhney R.; Galston A. W. Polyamine biosynthesis during vegetative and floral bud differentiation in thin layer tobacco tissue cultures. Plant Cell Physiol. 29: 1241–1249; 1988.
Tonon G.; Kevers C.; Gaspar T. Changes in polyamines, auxins and peroxidase activity during in vitro rooting of Fraxinus angustifolia shoots: An auxin independent rooting model. Tree Physiol. 2110: 655–663; 2001.
Tupy J.; Hrabetova E.; Capkova V. Amino acids and bivalent cations in the growth of tobacco pollen in mass culture. Plant Sci. Lett. 30: 91–98; 1983.
Vasudevan A.; Selvaraj N.; Suresh Kumar S.; Ganapathi A. Multiple shoot induction from shoot tip explants of Cucumber (Cucumis sativus L.). Cucurbit Genet. Coop. Rep. 24: 8–12; 2001.
Walden R.; Cordeiro A.; Tiburcio A. F. Polyamines: Small molecules triggering pathways in plant growth and development. Plant Physiol. 113: 1009–1013; 1997.
Yamada Y.; Kumpaisal R.; Hashimoto T.; Sugimoto Y. Growth and aspartate kinase activity in wheat cell suspension culture: Effects of lysine analogs and aspartate-derived amino acids. Plant Cell Physiol. 27: 607–617; 1986.
Zhu C.; Chen Z. Role of polyamines in adventitious shoot morphogenesis from cotyledons of cucumber in vitro. Plant Cell Tiss. Org. Cult. 81: 45–53; 2005.
Acknowledgment
The authors are grateful to Department of Science and Technology (DST), Government of India, for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Gregory C. Phillips
Rights and permissions
About this article
Cite this article
Vasudevan, A., Selvaraj, N., Ganapathi, A. et al. Leucine and spermidine enhance shoot differentiation in cucumber (Cucumis sativus L.). In Vitro Cell.Dev.Biol.-Plant 44, 300–306 (2008). https://doi.org/10.1007/s11627-008-9135-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-008-9135-0