Abstract
Objective
Retinal nerve fiber layer thickness (RNFL) is a biomarker of neuroaxonal loss and index of visual function in multiple sclerosis (MS). We aimed to assess the correlation between radiomic features and RNFL, visual acuity (VA) at patients’ presentation, visual outcome (VO), and clinical diagnosis.
Methods
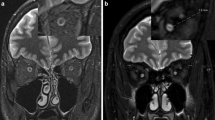
We reviewed imaging and clinical data of 25 patients with a first episode of optic neuritis (ON) (14 females, 11 males; 5 bilateral ON; 7 left ON; 13 right ON). All patients underwent a complete ophthalmological assessment, including visual acuity and RNFL, neurological evaluation, orbits MRI. Segmentation of the optic nerves was performed through 3D slicer open software to get radiomics analysis. All patients underwent a complete neuro-ophthalmological follow-up at 6 months to assess the VO, classified as: complete recovery, partial recovery, deficit persistence/relapse, or visual worsening and were diagnosed as MS or clinically isolated syndrome.
Results
We observed significant correlations between radiomic features and RNFL and between radiomic features and VA. Regression model analysis identified 1 radiomic feature with significant association with VO (Gray Level non-uniformity Normalized, p = 0.004) and 6 radiomic features with significant correlation with diagnosis (High Gray Level Zone Emphasis, p < 0.001; Entropy, p < 0.001, for T1 segmentation; Mean Absolute Deviation, p < 0.001; Coarseness < 0.001; Small Area Low Gray Level Emphasis, p < 0.001; Contrast, p = 0.008, for STIR segmentation).
Conclusion
Orbits MRI analysis at the first episode of ON has the potential to assess the visual function and VO in ON patients, and predict MS development.


Similar content being viewed by others
References
Petzold A, Wattjes M, Costello F et al (2014) The investigation of acute optic neuritis: a review and proposed protocol. Nat Rev Neurol 10(8):447–458. https://doi.org/10.1038/nrneurol.2014.108
Protti A, Spreafico C, Frigerio R et al (2004) Optic neuritis: diagnostic criteria application in clinical practice. Neurol Sci 25(Suppl 3):S296–S297
Ghezzi A, Martinelli V, Rodegher M et al (2000) The prognosis of idiopathic optic neuritis. Neurol Sci 21(4 Suppl 2):S865–S869
Huang-Link YM, Al-Hawasi A, Lindehammar H (2015) Acute optic neuritis: retinal ganglion cell loss precedes retinal nerve fiber thinning. Neurol Sci 36(4):617–620. https://doi.org/10.1007/s10072-014-1982-3
Kolappan M, Henderson AP, Jenkins TM et al (2009) Assessing structure and function of the afferent visual pathway in multiple sclerosis and associated optic neuritis. J Neurol 256(3):305–319. https://doi.org/10.1007/s00415-009-0123-z
Tas A, Altun S, Yolcu U et al (2015) Acute optic neuritis: retinal ganglion cell loss precedes retinal nerve fiber thinning. Neurol Sci 36(7):1277. https://doi.org/10.1007/s10072-014-2011-2
Hirst CL, Ingram G, Pickersgill TP et al (2012) Temporal evolution of remission following multiple sclerosis relapse and predictors of outcome. Mult Scler 18(8):1152–1158. https://doi.org/10.1177/1352458511433919
Cellina M, Floridi C, Rosti C et al (2019) MRI of acute optic neuritis (ON) at the first episode: can we predict the visual outcome and the development of multiple sclerosis (MS)? Radiol Med. https://doi.org/10.1007/s11547-019-01073-1
Hickman SJ, Toosy AT, Miszkiel KA et al (2004) Visual recovery following acute optic neuritis–a clinical, electrophysiological and magnetic resonance imaging study. J Neurol 251(8):996–1005. https://doi.org/10.1007/s00415-004-0477-1
Kupersmith MJ, Mandel G et al (2011) Baseline, one and three month changes in the peripapillary retina nerve fiber layer in acute optic neuritis: relation to baseline vision and MRI. J Neurol Sci 308(1–2):117–123. https://doi.org/10.1016/j.jns.2011.05.039
Jenkins TM, Toosy AT, Ciccarelli O (2010) Neuroplasticity predicts outcome of optic neuritis independent of tissue damage. Ann Neurol 67(1):99–113. https://doi.org/10.1002/ana.21823
Berg S, Kaschka I, Utz KS et al (2015) Baseline magnetic resonance imaging of the optic nerve provides limited predictive information on short-term recovery after acute optic neuritis. PLoS ONE 10(1):e0113961. https://doi.org/10.1371/journal.pone.0113961
McKee JB, Elston J, Evangelou N et al (2015) Amiloride clinical trial in optic neuritis (ACTION) protocol: a randomised, double blind, placebo controlled trial. BMJ Open 5(11):e009200. https://doi.org/10.1136/bmjopen-2015-009200
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures. Data Radiol 278(2):563–577. https://doi.org/10.1148/radiol.2015151169
Zhang Y (2012) MRI texture analysis in multiple sclerosis. Int J Biomed Imaging. https://doi.org/10.1155/2012/762804
Yip SS, Aerts HJ (2016) Applications and limitations of radiomics. Phys Med Biol 61(13):R150-66. https://doi.org/10.1088/0031-9155/61/13/R150
Zhang Y, Metz LM, Scott JN et al (2014) MRI texture heterogeneity in the optic nerve predicts visual recovery after acute optic neuritis. Neuroimage Clin 14(4):302–307. https://doi.org/10.1016/j.nicl.2014.01.003
Cellina M, Fetoni V, Ciocca M et al (2018) Anti-myelin oligodendrocyte glycoprotein antibodies: Magnetic resonance imaging findings in a case series and a literature review. Neuroradiol J 31(1):69–82. https://doi.org/10.1177/1971400917698856
Fedorov A, Beiche R, Kalpathy Cramer J et al (2012) 3D Slicer as an Image Computing Platform for the Quantitative Imaging Network. Magn Reson Imag 30(9):1323–1341. https://doi.org/10.1016/j.mri.2012.05.001
Pieper S, Halle M, Kikinis R (2004) 3D Slicer. IEEE Int Symp Biomed Imag Nano Macro 1:632–635
Brusa A, Jones SJ, Plant GT (2001) Long-term remyelination after optic neuritis: A 2-year visual evoked potential and psychophysical serial study. Brain 124(Pt 3):468–479
Thompson AJ, Banwell BL, Barkhof F et al (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17(2):162–173. https://doi.org/10.1016/S1474-4422(17)30470-2
Jenkins T, Ciccarelli O, Toosy A et al (2010) Dissecting structure-function interactions in acute optic neuritis to investigate neuroplasticity. Hum Brain Mapp 31(2):276–286. https://doi.org/10.1002/hbm.20863
Swanton JK, Fernando KT, Dalton CM et al (2010) Early MRI in optic neuritis: the risk for clinically definite multiple sclerosis. Mult Scler 16(2):156–165. https://doi.org/10.1177/1352458509353650
Jankowska-Lech I, Wasyluk J, Palasik W et al (2018) Peripapillary retinal nerve fiber layer thickness measured by optical coherence tomography in different clinical subtypes of multiple sclerosis. Mult Scler Relat Disord 5(27):260–268. https://doi.org/10.1016/j.msard.2018.11.003
Waldman AT, Hiremath G, Avery RA et al (2013) Monocular and binocular low-contrast visual acuity and optical coherence tomography in pediatric multiple sclerosis. Mult Scler Relat Disord 3(3):326–334. https://doi.org/10.1016/j.msard.2013.10.008
Cettomai D, Hiremath G, Ratchford J et al (2010) Associations between retinal nerve fiber layer abnormalities and optic nerve examination. Neurology 75(15):1318–1325. https://doi.org/10.1212/WNL.0b013e3181f735bd
Costello F, Coupland S, Hodge W et al (2006) Quantifying axonal loss after optic neuritis with optical coherence tomography. Ann Neurol 59(6):963–969. https://doi.org/10.1002/ana.20851
Costello F, Burton JM (2018) Retinal imaging with optical coherence tomography: a biomarker in multiple sclerosis? Eye Brain 10:47–63. https://doi.org/10.2147/EB.S139417
Soelberg K, Skejoe HPB, Grauslund J et al (2018) Magnetic resonance imaging findings at the first episode of acute optic neuritis. Mult Scler Relat Disord 20:30–36. https://doi.org/10.1016/j.msard.2017.12.018
Harrison LC, Raunio M, Holli KK et al (2010) MRI texture analysis in multiple sclerosis: toward a clinical analysis protocol. Acad Radiol 17(6):696–707. https://doi.org/10.1016/j.acra.2010.01.005
Zhang Y, Moore GR, Laul C et al (2013) Pathological correlates of magnetic resonance imaging texture heterogeneity in multiple sclerosis. Ann Neurol 74(1):91–99. https://doi.org/10.1002/ana.23867
Zhang Y, Zhu H, Mitchell JR et al (2009) T2 MRI texture analysis is a sensitive measure of tissue injury and recovery resulting from acute inflammatory lesions in multiple sclerosis. Neuroimage 47(1):107–111. https://doi.org/10.1016/j.neuroimage.2009.03.075
Zhang Y, Traboulsee A, Zhao Y et al (2011) Texture analysis differentiates persistent and transient T1 black holes at acute onset in multiple sclerosis: a preliminary study. Mult Scler 17(5):532–540. https://doi.org/10.1177/1352458510395981
Cadavid D, Balcer L, Galetta S et al (2017) RENEW Study Investigators Safety and efficacy of opicinumab in acute optic neuritis (RENEW): a randomised, placebo-controlled, phase 2 trial. Lancet Neurol 16(3):189–199. https://doi.org/10.1016/S1474-4422(16)30377-5
Takahashi H, Okuda S, Tamura M et al (2019) Prophylactic treatment with intravenous immunoglobulin attenuates experimental optic neuritis in mice. Biol Pharm Bull 42(2):173–178. https://doi.org/10.1248/bpb.b18-00458
Tourbah A, Gout O, Vighetto A et al (2018) MD1003 (high-dose pharmaceutical-grade biotin) for the treatment of chronic visual loss related to optic neuritis in multiple sclerosis: a randomized, double-blind. Placebo-Controlled Study CNS Drugs 32(7):661–672. https://doi.org/10.1007/s40263-018-0528-2
Zhang Y, Zhu Y, Shi X et al (2018) Soft tissue sarcomas: preoperative predictive histopathological grading based on radiomics of MRI. Acad Radiol. https://doi.org/10.1016/j.acra.2018.09.025
Lecler A, Duron L, Balvay D et al (2019) Combining multiple magnetic resonance imaging sequences provides independent reproducible radiomics features. Sci Rep 9(1):2068. https://doi.org/10.1038/s41598-018-37984-8
Funding
No funding has been received for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Human or animals rights
This retrospective study involving human participants has been approved by our Institutional Review Board.
Informed consent
Informed written consent was provided by patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cellina, M., Pirovano, M., Ciocca, M. et al. Radiomic analysis of the optic nerve at the first episode of acute optic neuritis: an indicator of optic nerve pathology and a predictor of visual recovery?. Radiol med 126, 698–706 (2021). https://doi.org/10.1007/s11547-020-01318-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-020-01318-4