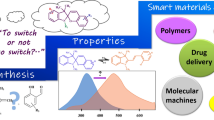
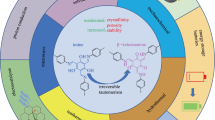
Abstract
This account gives an overview of our recent work in the area of conjugated azomethines derived from 2-aminothiophenes. It will be presented that mild reaction conditions can be used to selectively prepare symmetric and unsymmetric conjugated azomethines. It further will be demonstrated that azomethines consisting of various 5-membered aryl heterocycles lead to chemically, reductively, hydrolytically, and oxidatively robust compounds. The optical and electrochemical properties of these materials can be tuned contingent on the degree of conjugation, type of aryl heterocycle, and by including various electronic groups. The end result is materials having colors spanning 250 nm across the visible spectrum. These colors further can be tuned via electrochemical or chemical doping. The resulting doped states have high color contrasts from their corresponding neutral states. The collective opto-electronic properties and the means to readily tune them, make thiophenoazomethine derivatives interesting materials for potential use in a gamut of applications.
Similar content being viewed by others
References
Chiang CK, Fincher CRJ, Park YW, Heeger AJ, Shirakawa H, Louis EJ, Gau SC, MacDiarmid AG. Electrical conductivity in doped polyacetylene. Phys Rev Lett, 1977, 39(17): 1098–1101
Morin JF, Leclerc M, Adès D, Siove A. Polycarbazoles: 25 years of progress. Macromol Rapid Commun, 2005, 26(10): 761–778
Klärner G, Müller M, Morgenroth F, Wehmeier M, Soczka-Guth T, Müllen K. Conjugated oligomers and polymers —New routes, new structures. Synthetic Met, 1997, 84(1–3): 297–301
Rasmussen SC, Schwiderski RL, Mulholland ME. Thieno[3,4-b]pyrazines and their applications to low band gap organic materials. Chem Commun, 2011, 47(41): 11394–11410
Mishra A, Ma CQ, Bäuerle P. Functional oligothiophenes: Molecular design for multidimensional nanoarchitectures and their applications. Chem Rev, 2009, 109(3): 1141–1276
Schlüter AD. The tenth anniversary of suzuki polycondensation (spc). J Polym Sci Pol Chem, 2001, 39(10): 1533–1556
Chemli M, Haj Said A, Fave JL, Barthou C, Majdoub M. Synthesis and chemical modification of new luminescent substituted poly(p-phenylene) polymers. J Appl Polym Sci, 2012, 125(5): 3913–3919
Senkovskyy V, Tkachov R, Beryozkina T, Komber H, Oertel U, Horecha M, Bocharova V, Stamm M, Gevorgyan SA, Krebs FC, Kiriy A. “Hairy” poly(3-hexylthiophene) particles prepared via surface-initiated kumada catalyst-transfer polycondensation. J Am Chem Soc, 2009, 131(45): 16445–16453
Tam TL, Tan HHR, Ye W, Mhaisalkar SG, Grimsdale AC. One-pot synthesis of 4,8-dibromobenzo[1,2-d;4,5-d’]bistriazole and synthesis of its derivatives as new units for conjugated materials. Org Lett, 2011, 14(2): 532–535
Van den Bergh K, De Winter J, Gerbaux P, Verbiest T, Koeckelberghs G. Ni-catalyzed polymerization of poly(3-alkoxythiophene)s. Macromol Chem Phys, 2011, 212(4): 328–335
Pei J, Wen S, Zhou Y, Dong Q, Liu Z, Zhang J, Tian W. A low band gap donor-acceptor copolymer containing fluorene and benzothiadiazole units: Synthesis and photovoltaic properties. New J Chem, 2011, 35(2): 385–393
Cheng YJ, Luh TY. Synthesizing optoelectronic heteroaromatic conjugated polymers by cross-coupling reactions. J Organomet Chem, 2004, 689(24): 4137–4148
Berrouard P, Najari A, Pron A, Gendron D, Morin P-O, Pouliot J-R, Veilleux J, Leclerc M. Synthesis of 5-alkyl[3,4-c]thienopyrrole-4,6-dione-based polymers by direct heteroarylation. Angew Chem Int Ed, 2012, 51(9): 2068–2071
Hofmann S, Thomschke M, Lüssem B, Leo K. Top-emitting organic light-emitting diodes. Opt Express, 2011, 19(S6): A1250–A1264
Chen S, Deng L, Xie J, Peng L, Xie L, Fan Q, Huang W. Recent developments in top-emitting organic light-emitting diodes. Adv Mater, 2010, 22(46): 5227–5239
Zhong C, Duan C, Huang F, Wu H, Cao Y. Materials and devices toward fully solution processable organic light-emitting diodes. Chem Mater, 2010, 23(3): 326–340
Zhang F, Wu D, Xu Y, Feng X. Thiophene-based conjugated oligomers for organic solar cells. J Mater Chem, 2011, 21(44): 17590–17600
Xue J. Perspectives on organic photovoltaics. Polymer Rev, 2010, 50(4): 411–419
Beaujuge PM, Reynolds JR. Color control in π-conjugated organic polymers for use in electrochromic devices. Chem Rev, 2010, 110(1): 268–320
Mortimer RJ, Dyer AL, Reynolds JR. Electrochromic organic and polymeric materials for display applications. Displays, 2006, 27(1): 2–18
Haubner K, Jaehne E, Adler HJP, Koehler D, Loppacher C, Eng LM, Grenzer J, Herasimovich A, Scheiner S. Assembly, structure, and performance of an ultra-thin film organic field-effect transistor (OFET) based on substituted oligothiophenes. Org Electron, 2009, 75–94
Yumusak C, Sariciftci NS. Organic electrochemical light emitting field effect transistors. Appl Phys Lett, 2010, 97(3): 033302
Aleshin AN, Shcherbakov IP, Petrov VN, Titkov AN. Solution-processed polyfluorene-zno nanoparticles ambipolar light-emitting field-effect transistor. Org Electron, 2011, 12(8): 1285–1292
Okamoto K, Luscombe CK. Controlled polymerizations for the synthesis of semiconducting conjugated polymers. Polym Chem, 2011, 2(11): 2424–2434
Starčević K, Boykin DW, Karminski-Zamola G. New amidino-benzimidazolyl thiophenes: Synthesis and photochemical synthesis. Heteroat Chem, 2003, 14(3): 218–222
Younes AH, Zhang L, Clark RJ, Davidson MW, Zhu L. Electronic structural dependence of the photophysical properties of fluorescent heteroditopic ligands-implications in designing molecular fluorescent indicators. Org Biomol Chem, 2010, 8(23): 5431–5441
Karacsony O, Deschamps JR, Trammell SA, Nita R, Knight DA. Synthesis of a 2,2′-bipyridyl functionalized oligovinylene-phenylene using heck and horner-wadsworth-emmons reactions and X-ray crystal structure of e-(4-(4-bromostyryl)phenyl)(methyl)sulfane. Molecules, 2012, 17: 5724–5732
Patil PS, Haram NS, Pal RR, Periasamy N, Wadgaonkar PP, Salunkhe MM. Synthesis, spectroscopy, and electrochemical investigation of new conjugated polymers containing thiophene and 1,3,4-thiadiazole in the main chain. J Appl Polym Sci, 2012, 125(3): 1882–1889
Jenekhe SA, Yang CJ, Vanherzeele H, Meth JS. Cubic nonlinear optics of polymer thin films. Effects of structure and dispersion on the nonlinear optical properties of aromatic schiff base polymers. Chem Mater, 1991, 3(6): 985–987
Schab-Balcerzak E, Grucela-Zajac M, Krompiec M, Niestroj A, Janeczek H. New low band gap compounds comprised of naphthalene diimide and imine units. Synth Met, 2012, 162(5–6): 543–553
Bolduc A, Dufresne S, Skene WG. Chemical doping of edot azomethine derivatives: Insight into the oxidative and hydrolytic stability. J Mater Chem, 2012, 22(11): 5053–5064
Jursic BS. Suitability of furan, pyrrole and thiophene as dienes for diels-alder reactions viewed through their stability and reaction barriers for reactions with acetylene, ethylene and cyclopropene. An am1 semiempirical and b3lyp hybrid density functional theory study. Theochem, 1998, 454(2–3): 105–116
Bourgeaux M, Skene WG. Photophysics and electrochemistry of conjugated oligothiophenes prepared by using azomethine connections. J Org Chem, 2007, 72(23): 8882–8892
Yang CJ, Jenekhe SA. Conjugated aromatic poly(azomethines). 1. Characterization of structure, electronic spectra, and processing of thin films from soluble complexes. Chem Mater, 1991, 3(5): 878–887
da Silva CM, da Silva DL, Martins CVB, de Resende MA, Dias ES, Magalhães TFF, Rodrigues LP, Sabino AA, Alves RB, de Fátima Â. Synthesis of aryl aldimines and their activity against fungi of clinical interest. Chem Biol Drug Des, 2011, 78(5): 810–815
Matharu BK, Sharma JR, Manrao MR. Aldimines: Synthesis and effect of molecule dimension on antifungal potential. J Indian Counc Chem, 2006, 23: 47–50
Rani N, Sharma JR, Manrao MR. Synthesis and comparative fungitoxicity of benzalbenzylamines and benzalanilines. Pestic Res J, 2006, 18: 129–132
Niazi S, Javali C, Paramesh M, Shivaraja S. Study of influence of linkers and substitutions on antimicrobial activity of some schiff bases. Int J Pharm Pharm Sci, 2010, 2: 108–112
Hania MM. Synthesis of some imines and investigation of their biological activity. E-J Chem, 2009, 6: 629–632
Ozkay Y, Incesu Z, Isikdag I, Yesilkaya M. Antiproliferative effects of some n-benzylideneanilines. Cell Biochem Funct, 2008, 26: 102–106
Han SY, Inoue H, Terada T, Kamoda S, Saburi Y, Sekimata K, Saito T, Kobayashi M, Shinozaki K, Yoshida S, Asami T. N-benzylideneaniline and N-benzylaniline are potent inhibitors of lignostilbene-α,β-dioxygenase, a key enzyme in oxidative cleavage of the central double bond of lignostilbene. J Enzyme Inhib Med Chem, 2003, 18: 279–283
Dronia H, Gruss U, Gaegele G, Friedrich T, Weiss H. Structure-activity analysis of fluorinated 1-n-arylamino-1-arylmethane-phosphonic acid esters as inhibitors of the nadh:Ubiquinone oxidoreductase (complex i). J Comput-Aided Mol Des, 1996, 10: 100–106
Pasayat S, Dash SP, Saswati, Majhi PK, Patil YP, Nethaji M, Dash HR, Das S, Dinda R. Mixed-ligand aroylhydrazone complexes of molybdenum: Synthesis, structure and biological activity. Polyhedron, 2012, 38(1): 198–204
Patel RN, Singh A, Shukla KK, Sondhiya VP, Patel DK, Singh Y, Pandey R. Design, synthesis, and characterization of a series of biologically active copper(II) schiff-base coordination compounds. J Coord Chem, 2012, 65: 1381–1397
Kerneghan PA, Halperin SD, Bryce DL, Maly KE. Postsynthetic modification of an imine-based microporous organic network. Can J Chem, 2011, 89(5): 577–582, S577/571-S577/516
Uribe-Romo FJ, Doonan CJ, Furukawa H, Oisaki K, Yaghi OM. Crystalline covalent organic frameworks with hydrazone linkages. J Am Chem Soc, 2011, 133(30): 11478–11481
Pandey P, Katsoulidis AP, Eryazici I, Wu Y, Kanatzidis MG, Nguyen ST. Imine-linked microporous polymer organic frameworks. Chem Mater, 2010, 22(17): 4974–4979
Uribe-Romo FJ, Hunt JR, Furukawa H, Klöck C, O’Keeffe M, Yaghi OM. A crystalline imine-linked 3-d porous covalent organic framework. J Am Chem Soc, 2009, 131(13): 4570–4571
Pattakaran RLR, Burkanudeen AR. Synthesis and characterization of epoxy-containing schiff-base and phenylthiourea groupes for improved thermal conductivity. Polym-Plast Technol, 2012, 51: 140–145
Zhang XH, Huang LH, Chen S, Qi GR. Improvement of thermal properties and flame retardancy of epoxy-amine thermosets by introducing bisphenol containing azomethine moiety. Express Polym Lett, 2007, 1(5): 326–332
Ayesha Kausar SZ, Ahmad Z, Muhammad IS. Novel processable and heat resistant poly(phenylthiourea azomethine imide)s: Synthesis and characterization. Polym Degrad Stab, 2010, 95: 1826–1833
Bourque AN, Dufresne S, Skene WG. Thiophene-phenyl azomethines with varying rotational barriers-model compounds for examining imine fluorescence deactivation. J Phys Chem C, 2009, 113(45): 19677–19685
Dong Y, Bolduc A, McGregor N, Skene WG. Push-pull aminobithiophenes-highly fluorescent stable fluorophores. Org Lett, 2011, 13(7): 1844–1847
Dufresne S, Perez Guarin SA, Bolduc A, Bourque AN, Skene WG. Conjugated fluorene-thiophenes prepared from azomethine connections part i. The effect of electronic and aryl groups on the spectroscopic and electrochemical properties. Photochem Photobiol Sci, 2009, 8(6): 796–804
Knipping É, Roche IU, Dufresne S, McGregor N, Skene WG. Selective fluorescence turn-on of a prefluorescent azomethine with Zn2+. Tetrahedron Lett, 2011, 52(34): 4385–4387
Farcas A, Jarroux N, Ghosh I, Guégan P, Nau WM, Harabagiu V. Polyrotaxanes of pyrene-triazole conjugated azomethine and α-cyclodextrin with high fluorescence properties. Macromol Chem Phys, 2009, 210(17): 1440–1449
Sibel Derinkuyu KE, Oter O, Ergun Y. Ph-driver fluorescent switch behavior of azomethine dyes in solid matrix materials. Spectrosc Lett, 2010, 43: 500–512
Liu JL, Xu S, Yan B. Photoactive hybrids with the functionalized schiff-base derivatives covalently bonded inorganic silica network: Sol-gel synthesis, characterization and photoluminescence. Colloids Surf A, 2011, 373(1–3): 116–123
Sek D, Grabiec E, Janeczek H, Jarzabek B, Kaczmarczyk B, Domanski M, Iwan A. Structure-properties relationship of linear and star-shaped imines with triphenylamine moieties as hole-transporting materials. Opt Mater, 2010, 32(11): 1514–1525
Yen HJ, Liou GS. Novel blue and red electrochromic poly(azomethine ether)s based on electroactive triphenylamine moieties. Org Electron, 2010, 11(2): 299–310
Is OD, Koyuncu FB, Koyuncu S, Ozdemir E. A new imine coupled pyrrole-carbazole-pyrrole polymer: Electro-optical properties and electrochromism. Polymer, 2010, 51(8): 1663–1669
Gao Z, Yu Y, Xu Y, Li S. Synthesis and characterization of a liquid crystalline epoxy containing azomethine mesogen for modification of epoxy resin. J Appl PolymSci, 2007, 105(4): 1861–1868
Mallikharjuna Rao Darla SV. Synthesis and characterisation of azomethine class thermotropic liquid crystals and their application in non-linear optics. Liq Cryst, 2012, 39(1): 63–70
Iwan A, Bilski P, Janeczek H, Jarzabek B, Domanski M, Rannou P, Sikora A, Pociecha D, Kaczmarczyk B. Thermal, optical, electrical and structural study of new symmetrical azomethine based on poly(1,4-butanediol)bis(4-aminobenzoate). J Mol Struct, 2010, 963(2–3): 175–182
Iwan A, Palewicz M, Sikora A, Chmielowiec J, Hreniak A, Pasciak G, Bilski P. Aliphatic-aromatic poly(azomethine)s with ester groups as thermotropic materials for opto(electronic) applications. Synth Met, 2010, 160(17–18): 1856–1867
Hindson JC, Ulgut B, Friend RH, Greenham NC, Norder B, Kotlewski A, Dingemans TJ. All-aromatic liquid crystal triphenylamine-based poly(azomethine)s as hole transport materials for opto-electronic applications. J Mater Chem, 2010, 20(5): 937–944
Bürgi HB, Dunitz JD. Crystal and molecular structures of benzylideneaniline, benzylideneaniline-p-carboxylic acid and p-methylbenzylidene-p-nitroaniline. Helv Chim Acta, 1970, 53(7): 1747–1764
Hoekstra A, Meertens P, Vos A. Refinement of the crystal structure of trans-stilbene (TSB). The molecular structure in the crystalline and gaseous phases. Acta Crystallogr, Sect B: Struct Sci, 1975, 31(12): 2813–2817
Bartholomew GP, Bu X, Bazan GC. Preferential cocrystallization among distyrylbenzene derivatives. Chem Mater, 2000, 12: 2311–2318
Zhu S, Zhu S, Jin G, Li Z. Strong phenyl-perfluorophenyl π-π stacking and C-H…F-C hydrogen bonding interactions in the crystals of the corresponding aromatic aldimines. Tetrahedron Lett, 2005, 46(15): 2713–2716
Mallet C, Allain M, Leriche P, Frere P. Competition between π-π or furan-perfluorophenyl stacking interactions in conjugated compounds prepared from azomethine connections. CrystEngComm, 2011, 13(19): 5833–5840
Roncali J. Conjugated poly(thiophenes): Synthesis, functionalization, and applications. Chem Rev, 1992, 92(4): 711–738
McCullough RD, Tristram-Nagle S, Williams SP, Lowe RD, Jayaraman M. Self-orienting head-to-tail poly(3-alkylthiophenes): New insights on structure-property relationships in conducting polymers. J Am Chem Soc, 1993, 115(11): 4910–4911
Facchetti A. Electroactive oligothiophenes and polythiophenes for organic field effect transistors. Handbook of Thiophene-Based Materials: Applications in Organic Electronics and Photonics, 2009, 1: 595–646
Lu K, Liu Y. Polythiophenes: Important conjugated semiconducting polymers for organic field-effect transistors. Curr Org Chem, 2010, 14: 2017–2033
Gigli G, Barbarella G, Favaretto L, Cacialli F, Cingolani R. High-efficiency oligothiopene-based light-emitting diodes. Appl Phys Lett, 1999, 75: 439–441
Amb CM, Dyer AL, Reynolds JR. Navigating the color palette of solution-processable electrochromic polymers. Chem Mater, 2011, 23: 397–415
Gunbas G, Toppare L. Electrochromic conjugated polyheterocycles and derivatives-highlights from the last decade towards realization of long lived aspirations. Chem Commun, 2012, 48: 1083–1101
Bench R, Duflos J, Dupas G, Bourguignon J, Queguiner G. Synthesis and study of chiral nadh models in the thieno[2,3-b]pyridine series. J Heterocycl Chem, 1989, 26(6): 1595–1600
Chirakadze GG, Geliashvili EE, Gagolishvili MS. Synthesis and properties of thiophene containing azo dyes and pigments. Izv Akad Nauk Gruz, Ser Khim, 1999, 25: 203–209
Ivanova VN. Nitrogenous compounds of phenylated derivatives of thiophene. I. Zh Obshch Khim, 1958, 28: 1232–1238
Puterova Z, Krutošíková A, Végh D. Gewald reaction: Synthesis, properties and applications of substituted 2-aminothiophenes. ARKIVOC, 2010, i: 209–246
Buchstaller H-P, Siebert CD, Lyssy RH, Frank I, Duran A, Gottschlich R, Noe CR. Synthesis of novel 2-aminothiophene-3-carboxylates by variations of the gewald reaction. Monats Chem, 2001, 132(2): 279–293
Sabnis RW, Rangnekar DW, Sonawane ND. 2-Aminothiophenes by the gewald reaction. J Heterocycl Chem, 1999, 36(2): 333–345
Bourgeaux M, Vomscheid S, Skene WG. Optimized synthesis and simple purification of 2,5-diaminothiophene-3,4-dicarboxylic acid diethyl ester. Synth Commun, 2007, 37: 3551–3558
Gewald K. Heterocycles from ch-acidic nitriles. Vii. 2-Aminothiophene from a-oxo mercaptans and methylene-active nitriles. Chem Ber, 1965, 98(11): 3571–3577
Gewald K. Methods for the synthesis of 2-aminothiophenes and their reactions (review). Chem Hetero Comp, 1976, 12(10): 1077–1090
Gewald K, Gruner M, Hain U, Süptitz G. Zur ringumwandlung von 2-amino-thiophen-3-carbonsäureestern: Pyridon-und pyridazinonderivate. Monats Chem, 1988, 119(8–9): 985–992
Gewald VK, Kleinert M, Thiele B, Hentschel M. Zur basenkatalysierten reaktion von methylenaktiven nitrilen mit schwefel. J Prak Chem, 1972, 314(2): 303–314
Angell RM, Atkinson FL, Brown MJ, Chuang TT, Christopher JA, Cichy-Knight M, Dunn AK, Hightower KE, Malkakorpi S, Musgrave JR, Neu M, Rowland P, Shea RL, Smith JL, Somers DO, Thomas SA, Thompson G, Wang R. N-(3-cyano-4,5,6,7-tetrahydro-1-benzothien-2-yl)amides as potent, selective, inhibitors of jnk2 and jnk3. Bioorg Med Chem Lett, 2007, 17: 1296–1301
Bowers S, Truong AP, Neitz RJ, Neitzel M, Probst GD, Hom RK, Peterson B, Galemmo RA, Jr., Konradi AW, Sham HL, Toth G, Pan H, Yao N, Artis DR, Brigham EF, Quinn KP, Sauer JM, Powell K, Ruslim L, Ren Z, Bard F, Yednock TA, Griswold-Prenner I. Design and synthesis of a novel, orally active, brain penetrant, tri-substituted thiophene based jnk inhibitor. Bioorg Med Chem Lett, 2011, 21: 1838–1843
De SK, Barile E, Chen V, Stebbins JL, Cellitti JF, Machleidt T, Carlson CB, Yang L, Dahl R, Pellecchia M. Design, synthesis, and structure-activity relationship studies of thiophene-3-carboxamide derivatives as dual inhibitors of the c-jun n-terminal kinase. Bioorg Med Chem, 2011, 19: 2582–2588
Grembecka J, He S, Shi A, Purohit T, Muntean AG, Sorenson RJ, Showalter HD, Murai MJ, Belcher AM, Hartley T, Hess JL, Cierpicki T. Menin-mll inhibitors reverse oncogenic activity of mll fusion proteins in leukemia. Nat Chem Biol, 2012, 8: 277–284
Aurelio L, Christopoulos A, Flynn BL, Scammells PJ, Sexton PM, Valant C. The synthesis and biological evaluation of 2-amino-4,5,6,7,8,9-hexahydrocycloocta[b]thiophenes as allosteric modulators of the a1 adenosine receptor. Bioorg Med Chem Lett, 2011, 21: 3704–3707
Kumar V, Madan AK. Prediction of the agonist allosteric enhancer activity of thiophenes with respect to human a1 adenosine receptors using topological indices. Pharm Chem J, 2007, 41: 140–145
Nikolakopoulos G, Figler H, Linden J, Scammells PJ. 2-Aminothiophene-3-carboxylates and carboxamides as adenosine a1 receptor allosteric enhancers. Bioorg Med Chem, 2006, 14: 2358–2365
Gaber HM, Bagley MC, Sherif SM. Antimicrobial investigations on synthetic p-tolylazo derivatives of thienopyrimidinone based on an ortho-functionalized thiophene nucleus. Eur J Chem, 2010, 1: 115–123
Panchamukhi SI, Mulla JAS, Shetty NS, Khazi MIA, Khan AY, Kalashetti MB, Khazi IAM. Benzothieno[3,2-e][1,2,4]triazolo [4,3-c]pyrimidines: Synthesis, characterization, antimicrobial activity, and incorporation into solid lipid nanoparticles. Arch Pharm (Weinheim, Ger), 2011, 344: 358–365
Shams HZ, Mohareb RM, Helal MH, Mahmoud AE-S. Design and synthesis of novel antimicrobial acyclic and heterocyclic dyes and their precursors for dyeing and/or textile finishing based on 2-n-acylamino-4,5,6,7-tetrahydro-benzo[b]thiophene systems. Mole-cules, 2011, 16: 6271–6305
Hallas G, Choi JH. Synthesis and spectral properties of azo dyes derived from 2-aminothiophenes and 2-aminothiazoles. Dyes Pigm, 1999, 42(3): 249–265
Hallas G, Choi JH. Synthesis and properties of novel aziridinyl azo dyes from 2-aminothiophenes-Part 1: Synthesis and spectral properties. Dyes Pigm, 1999, 40(2–3): 99–117
Hallas G, Choi JH. Synthesis and properties of novel aziridinyl azo dyes from 2-aminothiophenes-Part 2: Application of some disperse dyes to polyester fibres. Dyes Pigm, 1999, 40(2–3): 119–129
Hallas G, Towns AD. Dyes derived from aminothiophenes. Part 1: Synthesis of some heterocyclic disperse dyes using the gewald reaction. Dyes Pigm, 1996, 32(3): 135–149
Hallas G, Towns AD. A comparison of the properties of some 2-aminothiophene-derived disperse dyes. Dyes Pigm, 1996, 31(4): 273–289
Hallas G, Towns AD. Dyes derived from aminothiophenes. Part 4: Synthesis of some nitro-substituted thiophene-based azo disperse dyes. Dyes Pigm, 1997, 33(4): 319–336
Hallas G, Towns AD. Dyes derived from aminothiophenes-Part 2. Spectroscepic properties of some disperse dyes derived from 2-aminothiophenes. Dyes Pigm, 1997, 33(3): 205–213
Hallas G, Towns AD. Dyes derived from aminothiophenes. Part 6: Application of some nitro-substituted thiophene-based azo disperse dyes to hydrophobic fibres. Dyes Pigm, 1997, 35(1): 45–55
Hallas G, Towns AD. Dyes derived from aminothiophenes-Part 3. Application of some disperse dyes derived from 2-aminothiophenes to hydrophobic fibres. Dyes Pigm, 1997, 33(3): 215–228
El-Shekeil A, Abu-Bakr AO. Dc electrical conductivity of the direct electrochemically synthesized polythiophene metal complexes. J Macromol Sci Part A: Pure Appl Chem, 2011, 48: 233–240
El-Shekeil A, Al-Khader M, Abu-Bakr AO. Synthesis, characterization and dc electrical conductivity of some oligmer mixed metal complexes. Synth Met, 2004, 143(2): 147–152
El-Dossoki FI. Electric conductance and semi-empirical studies on two thiophene derivatives/metal cation complexation. J Mol Liq, 2008, 142: 53–56
Skene WG, Dufresne S, Trefz T, Simard M. (e)-Diethyl 2-amino-5-(2-thienylmethyleneamino)thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2006, 62(6): o2382–o2384
Dufresne S, Bourgeaux M, Skene WG. Diethyl 2,5-bis((e)-thiophen-2-ylmethyleneamino)thiophene-3,4-dicarboxylate triad. Acta Crystallogr, Sect E: Struct Rep Online, 2006, 62(12): o5602–o5604
Wong BM, Cordaro JG. Electronic properties of vinylene-linked heterocyclic conducting polymers: Predictive design and rational guidance from dft calculations. J Phys Chem C, 2011, 115(37): 18333–18341
Qing F, Sun Y, Wang X, Li N, Li Y, Li X, Wang H. A novel poly(thienylenevinylene) derivative for application in polymer solar cells. Polym Chem, 2011, 2(9): 2102–2106
Gergely J, Morgan JB, Overman LE. Stereocontrolled synthesis of functionalized cis-cyclopentapyrazolidines by 1,3-dipolar cycloaddition reactions of azomethine imines. J Org Chem, 2006, 71: 9144–9152
Bourgeaux M, Skene WG. Photophysics and electrochemistry of conjugated oligothiophenes prepared by using azomethine connections. J Org Chem, 2007, 72(23): 8882–8892
Guarìn SAP, Bourgeaux M, Dufresne S, Skene WG. Photophysical, crystallographic, and electrochemical characterization of symmetric and unsymmetric self-assembled conjugated thiopheno azomethines. J Org Chem, 2007, 72(7): 2631–2643
Mielke J, Leyssner F, Koch M, Meyer S, Luo Y, Selvanathan S, Haag R, Tegeder P, Grill L. Imine derivatives on Au(111): Evidence for “inverted” thermal isomerization. ACS Nano, 2011, 5(3): 2090–2097
Luo Y, Utecht M, Dokić J, Korchak S, Vieth H-M, Haag R, Saalfrank P. Cis-trans isomerisation of substituted aromatic imines: A comparative experimental and theoretical study. ChemPhysChem, 2011, 2311–2321
Traven’ V, Ivanov I, Panov A, Safronova O, Chibisova T. Solvent-induced E/Z (C=N)-isomerization of imines of some hydroxy-substituted formylcoumarins. Rus Chem Bull, 2008, 57(9): 1989–1995
Selli E. Photochemistry of n-benzylideneanilinium cations in concentrated sulfuric acid solutions. J Photochem Photobiol A, 1996, 101(2–3): 185–188
Geissler G, Fust W, Krüger B, Tomaschewski G. Azomethinimine. Vii. Photochemisches und thermisches verhalten azarylsubstituierter pyrazolidon-(3)-azomethinimine. J Prak Chem, 1983, 325(2): 205–210
Russegger P. Photoisomerization about carbon-nitrogen double bonds. I. Kinetic and potential energy for ground and excited states of methylenimine. Chem Phys, 1978, 34(3): 329–339
Traven VF, Miroshnikov VS, Pavlov AS, Ivanov IV, Panov AV, Chibisova TyA. Unusual e/z-isomerization of 7-hydroxy-4-methyl-8-[(9h-fluoren-2-ylimino)methyl]-2h-1-benzopyran-2-one in acetonitrile. Mendeleev Commun, 17(2): 88–89
Dufresne S, Skene WG. Optoelectronic property tailoring of conjugated heterocyclic azomethines-the effect of pyrrole, thiophene and furans. J Phys Org Chem, 2011, 211–221
Roncali J. Conjugated poly(thiophenes): Synthesis, functionalization, and applications. Chem Rev, 1992, 92(4): 711–738
Lee CK, Yu JS, Lee HJ. Determination of aromaticity indices of thiophene and furan by nuclear magnetic resonance spectroscopic analysis of their phenyl esters. J Heterocyclic Chem, 2002, 39(6): 1207–1217
Dufresne S, Bolduc A, Skene WG. Towards materials with reversible oxidation and tuneable colours using heterocyclic conjugated azomethines. J Mater Chem, 2010, 20(23): 4861–4866
Bolduc A, Dufresne S, Skene WG. Edot-containing azomethine: An easily prepared electrochromically active material with tuneable colours. J Mater Chem, 2010, 20(23): 4820–4826
Dong Y, Navarathne D, Bolduc A, McGregor N, Skene WG. A,α′-n-boc-substituted bi- and terthiophenes: Fluorescent precursors for functional materials. J Org Chem, 2012, 77(22): 5429–5433
Lakowicz JR. Principles of Fluorescence Spectroscopy. New York: Springer, 2006
Bourgeaux M, Guarin SAP, Skene WG. Photophysical, crystallographic, and electrochemical characterization of novel conjugated thiopheno azomethines. J Mater Chem, 2007, 17(10): 972–979
Luo Y, Korchak S, Vieth HM, Haag R. Effective reversible photoinduced switching of self-assembled monolayers of functional imines on gold nanoparticles. Chem Phys Chem, 2011, 12(1): 132–135
Bléger D, Ciesielski A, Samorì P, Hecht S. Photoswitching vertically oriented azobenzene self-assembled monolayers at the solid-liquid interface. Chem Eur J, 2010, 16(48): 14256–14260
Bourque AN, Dufresne S, Skene WG. Conjugated fluorenes prepared from azomethines connections: The effect of alternating fluorenones and fluorenes on the spectroscopic and electrochemical properties. J Phys Chem C, 2009, 113(45): 19677–19685
Dufresne S, Skalski T, Skene WG. Insights into the effect of ketylimine, aldimine, and vinylene group attachment and regiosubstitution on the fluorescence deactivation of fluorene. Can J Chem, 2011, 89(2): 173–180
Dufresne S, Roche IU, Skalski T, Skene WG. Insights into the effect of the ketylimine group on the fluorescence deactivation of oligofluorenes. J Phys Chem C, 2010, 114(30): 13106–13112
Dufresne S, Bourque AN, Skene WG. (e)-5-(2-Thienylmethyleneamino)quinolin-8-ol. Acta Crystallogr, Sect E: Struct Rep Online, 2008, 64(1): o316
Skene WG, Dufresne S, Trefz T, Simard M. (e)-Diethyl 2-amino-5-(2-thienylmethyleneamino)thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2006, E62(6): o2382–o2384
Dufresne S, Bourgeaux M, Skene WG. Diethyl 2,5-bis[(e)-thiophen-2-ylmethyleneamino]thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2006, 62(12): o5602–o5604
Dufresne S, Skene WG. Diethyl 2,5-bis[(1e)-(1h-pyrrol-2-ylmethylidene)amino]thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2011, 67(9): o2302
Dufresne S, Skene WG. Diethyl 2-amino-5-[(e)-(1-methyl-1h-pyrrol-2-yl)methylideneamino]thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2010, 66(12): o3221
Dufresne S, Skene WG. Diethyl 2-amino-5-[(e)-(furan-2-ylmethylidene)amino]thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2010, 66(11): o3027
Dufresne S, Skene WG. Diethyl 2-[(1-methyl-1h-pyrrol-2-yl)methyleneamino]-5-(2-thienylmethyleneamino)thiophene-3,4-dica rboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2008, 64(5): o782
Dufresne S, Skene WG. Diethyl 2,5-bis[(e)-2-furylmethyleneamino] thiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2008, 64(4): o710
Dufresne S, Bolduc A, Skene WG. Diethyl 2,5-bis[(2,3-dihydroth-ieno[3,4-b][1,4]dioxin-5-yl)methylideneamino]thiophene-3,4-dicarbo xylate acetone monosolvate. Acta Crystallogr, Sect E: Struct Rep Online, 2011, 67(12): o3138
Bourgeaux M, Vomsheid S, Skene WG. Hydrogen-bonded network of diethyl 2,5-diaminothiophene-3,4-dicarboxylate. Acta Crystallogr, Sect E: Struct Rep Online, 2006, 62(12): o5529–o5531
Ruban G, Zobel D. Crystal structure of trans-1,2-di-2-thienylethene. Acta Crystallogr, Sect B, 1975, B31: 2632–2634
Dogan F, Kaya I, Bilici A. Azomethine-based phenol polymer: Synthesis, characterization and thermal study. Synth Met, 2011, 161(1–2): 79–86
El-Shekeil AG, Al-Yusufy FA, Saknidy S. Dc conductivity of some polyazomethines. Polym Int, 1997, 42(1): 39–44
Iwan A, Sek D. Processible polyazomethines and polyketanils: From aerospace to light-emitting diodes and other advanced applications. Prog Polym Sci, 2008, 33(3): 289–345
Bourgeaux M, Skene WG. A highly conjugated p- and n-type polythiophenoazomethine: Synthesis, spectroscopic, and electrochemical investigation. Macromolecules, 2007, 40(6): 1792–1795
Hall HKJ, Padias AB, Williams PA, Gosau JM, Boone HW, Park DK. Novel polyaromatic quinone imines. Macromolecules, 1995, 28(1): 1–8
Bourgeaux M, Skene WG. A highly conjugated p- and n-type polythiophenoazomethine: Synthesis, spectroscopic, and electrochemical investigation. Macromolecules, 2007, 40(6): 1792–1795
Giuseppone N. Toward self-constructing materials: A systems chemistry approach. Acc Chem Res, 2012, DOI: 10.1021/ar2002655
Rue NM, Sun J, Warmuth R. Polyimine container molecules and nanocapsules. Israel J Chem, 2011, 51(7): 743–768
Ciesielski A, Samori P. Supramolecular assembly/reassembly processes: Molecular motors and dynamers operating at surfaces. Nanoscale, 2011, 3(4): 1397–1410
Lehn J. Dynamers: Dynamic molecular and supramolecular polymers. Aust J Chem, 2010, 63(4): 611–623
Meyer CD, Joiner CS, Stoddart JF. Template-directed synthesis employing reversible imine bond formation. Chem Soc Rev, 2007, 36(11): 1705–1723
Barik S, Bishop S, Skene WG. Spectroelectrochemical and electrochemical investigation of a highly conjugated all-thiophene polyazomethine. Mater Chem Phys, 2011, 129(1–2): 529–533
Ryan B, McCann G. Novel sub-ceiling temperature rapid depolymerization-repolymerization reactions of cyanoacrylate polymers. Macromol Rapid Commun, 1996, 17: 217–227
Wong BM, Cordaro JG. Electronic properties of vinylene-linked heterocyclic conducting polymers: Predictive design and rational guidance from dft calculations. J Phys Chem C, 2011, 115(37): 18333–18341
Bentkowska H. The effect of factors breaking the siloxane bond on the repolymerization course of siloxanes. I. Effect of hydrogen chloride on cyclic and linear poly(diethylsiloxane). Rocz Chem, 1963, 37: 717–721
Barik S, Skene WG. Selective chain-end postpolymerization reactions and property tuning of a highly conjugated and all-thiophene polyazomethine. Macromolecules, 2010, 43(24): 10435–10441
Author information
Authors and Affiliations
Corresponding author
Additional information
After completing her B.Sc. degree in Chemistry at the Université de Montréal, BOLDUC Andréanne (left) joined Skene’s group as a M. Sc. student in 2008 and then transferred directly into the Ph.D. program. Her thesis focusses on preparing and examining new conjugated azomethines for different plastic electronic applications. MALLET Charlotte (center) completed her Ph.D. studies in 2010 at the Universitéd’Angers, France. She joined Skene’s group as a postdoctoral fellow in 2011, where she works on conjugated benzothiadiazole derivatives for fluorescence applications. SKENE William (right) is an associate professor at U de M. His research interests are the design, synthesis, and characterization of easily prepared conjugated materials for plastic electronics and the structure-property studies of these materials.
Rights and permissions
About this article
Cite this article
Bolduc, A., Mallet, C. & Skene, W.G. Survey of recent advances of in the field of π-conjugated heterocyclic azomethines as materials with tuneable properties. Sci. China Chem. 56, 3–23 (2013). https://doi.org/10.1007/s11426-012-4778-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-012-4778-4