Abstract
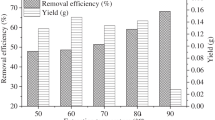
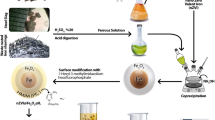
This research work aims to develop a biomaterial entrapped with iron nanoparticles by green synthesis method in which biomass act as both reducing and capping agent. Iron nanoparticles embedded in Citrus limetta peels were characterised using ICP-MS for determination of metal loading, XRD, XPS for crystallinity and oxidation states, TEM followed by FESEM-EDS for particle size and morphology. Sizes of nanoparticles were found to be in the range of 4–70 nm. Batch experiments were conducted to study the effect of different parameters such as contact time, amount of biomaterial and volume of chromium(VI) solution for 2500 mg L−1 of Cr(VI). Complete reduction was attained for a contact time of 5 min with 1.5 g of biomaterial for initial concentration of 2500 mg L−1. The experimental results inferred that 1 g of biomaterial completely reduced 33 mg of hexavalent Cr to trivalent Cr. XRD and XPS revealed that iron nanoparticles are in amorphous form while XPS confirms Fe0 state. The transition of Fe0 to Fe2+/Fe3+ during the treatment with chromium solution confirms the reduction of Cr6+ to Cr3+.










Similar content being viewed by others
References
Bhattacharyya KG, Gupta SS (2008) Adsorption of a few heavy metals on natural and modified kaolinite and montmorillonite: a review. Adv Colloid Interf Sci 140:114–131. https://doi.org/10.1016/j.cis.2007.12.008
Chen L, Ji T, Mu L, Shi Y, Brisbin L, Guo Z, Khan MA, Young DP, Zhu J (2016) Facile synthesis of mesoporous carbon nanocomposites from natural biomass for efficient dye adsorption and selective heavy metal removal. RSC Adv 6:2259–2269. https://doi.org/10.1039/c5ra19616g
Costa M, Klein C (2006) Toxicity and carcinogenicity of chromium compounds in humans. CRC Crit Rev Toxicol 36:155–163. https://doi.org/10.1080/10408440500534032
Dharnaik AS, Ghosh PK (2014) Hexavalent chromium [Cr(VI)] removal by the electrochemical ion-exchange process. Environ Technol (United Kingdom) 35:2272–2279. https://doi.org/10.1080/09593330.2014.902108
Ebrahiminezhad A, Zare-Hoseinabadi A, Berenjian A, Ghasemi Y (2017) Green synthesis and characterization of zero-valent iron nanoparticles using stinging nettle (Urtica dioica) leaf extract. Green Process Synth 6. https://doi.org/10.1515/gps-2016-0133
Fahmy HM, Mohamed FM, Marzouq MH, Mustafa ABED, Alsoudi AM, Ali OA, Mohamed MA, Mahmoud FA (2018) Review of green methods of iron nanoparticles synthesis and applications. Bionanoscience 8:491–503. https://doi.org/10.1007/s12668-018-0516-5
Gaikwad MS, Balomajumder C (2018) Removal of Cr(VI) and fluoride by membrane capacitive deionization with nanoporous and microporous Limonia acidissima (wood apple) shell activated carbon electrode. Sep Purif Technol 195:305–313. https://doi.org/10.1016/j.seppur.2017.12.006
Gan PP, Li SFY (2012) Potential of plant as a biological factory to synthesize gold and silver nanoparticles and their applications. Rev Environ Sci Biotechnol 11:169–206. https://doi.org/10.1007/s11157-012-9278-7
Garg UK, Kaur MP, Sud D, Garg VK (2009) Removal of hexavalent chromium from aqueous solution by adsorption on treated sugarcane bagasse using response surface methodological approach. Desalination 249:475–479. https://doi.org/10.1016/j.desal.2008.10.025
Geng B, Jin Z, Li T, Qi X (2009) Kinetics of hexavalent chromium removal from water by chitosan-Fe0 nanoparticles. Chemosphere 75:825–830. https://doi.org/10.1016/j.chemosphere.2009.01.009
Hoag GE, Collins JB, Holcomb JL, Hoag JR, Nadagouda MN, Varma RS (2009) Degradation of bromothymol blue by “greener” nano-scale zero-valent iron synthesized using tea polyphenols. J Mater Chem 19:8671–8677. https://doi.org/10.1039/b909148c
Johnson P, Loganathan C, Krishnan V, Sakayanathan P, Raji V, Vijayan S, Sathishkumar P, Murugesan K, Palvannan T (2017) Plant extract as environmental-friendly green catalyst for the reduction of hexavalent chromium in tannery effluent. Environ Technol (United Kingdom) 3330:1–8. https://doi.org/10.1080/09593330.2017.1329355
Karthikeyan T, Rajgopal S, Miranda LR (2005) Chromium(VI) adsorption from aqueous solution by Hevea Brasilinesis sawdust activated carbon. J Hazard Mater 124:192–199. https://doi.org/10.1016/j.jhazmat.2005.05.003
Kim T, Kim T, Choe W, Kim MK, Jung YJ, Zoh KD (2018) Removal of heavy metals in electroplating wastewater by powdered activated carbon ( PAC ) and sodium diethyldithiocarbamate-modified PAC. Environ Eng Res 23:301–308
Kumari V, Sasidharan M, Bhaumik A (2015) Mesoporous BaTiO3@SBA-15 derived via solid state reaction and its excellent adsorption efficiency for the removal of hexavalent chromium from water. Dalt Trans 44:1924–1932. https://doi.org/10.1039/c4dt03180f
Li XQ, Elliott DW, Zhang WX (2006) Zero-valent iron nanoparticles for abatement of environmental pollutants: materials and engineering aspects. Crit Rev Solid State Mater Sci 31:111–122. https://doi.org/10.1080/10408430601057611
Li Z, Kirk Jones H, Zhang P, Bowman RS (2007) Chromate transport through columns packed with surfactant-modified zeolite/zero valent iron pellets. Chemosphere 68:1861–1866. https://doi.org/10.1016/j.chemosphere.2007.03.011
López-Téllez G, Barrera-Díaz CE, Balderas-Hernández P, Morales GR, Bilyeu B (2011) Removal of hexavalent chromium in aquatic solutions by iron nanoparticles embedded in orange peel pith. Chem Eng J 173:480–485. https://doi.org/10.1016/j.cej.2011.08.018
Machado S, Pinto SL, Grosso JP, Nouws HPA, Albergaria JT, Delerue-Matos C (2013) Green production of zero-valent iron nanoparticles using tree leaf extracts. Sci Total Environ 445–446:1–8. https://doi.org/10.1016/j.scitotenv.2012.12.033
Madhavi V, Prasad TNVKV, Reddy AVB, Reddy R, Madhavi G (2013) Application of phytogenic zerovalent iron nanoparticles in the adsorption of hexavalent chromium. Spectrochim Acta A Mol Biomol Spectrosc 116:17–25. https://doi.org/10.1016/j.saa.2013.06.045
Mary Mangaiyarkarasi MS, Vincent S, Janarthanan S, Subba rao T, Tata BVR (2011) Bioreduction of Cr(VI) by alkaliphilic Bacillus subtilis and interaction of the membrane groups. Saudi J Biol Sci 18:157–167. https://doi.org/10.1016/j.sjbs.2010.12.003
Miretzky P, Cirelli AF (2010) Cr(VI) and Cr(III) removal from aqueous solution by raw and modified lignocellulosic materials: a review. J Hazard Mater 180:1–19. https://doi.org/10.1016/j.jhazmat.2010.04.060
Narayanan KB, Sakthivel N (2010) Biological synthesis of metal nanoparticles by microbes. Adv Colloid Interf Sci 156:1–13. https://doi.org/10.1016/j.cis.2010.02.001
Nayak A, Bhushan B, Gupta V, Sharma P (2017) Journal of colloid and interface science chemically activated carbon from lignocellulosic wastes for heavy metal wastewater remediation: effect of activation conditions. J Colloid Interface Sci 493:228–240. https://doi.org/10.1016/j.jcis.2017.01.031
Njagi EC, Huang H, Stafford L, Genuuino H, Galindo HM, Collins JB, Hoag GE, Suib SL (2011) Biosynthesis of iron and silver nanoparticles at room temperature using aqueous sorghum bran extracts. Langmuir 27:264–271. https://doi.org/10.1021/la103190n
Ray S, Kanti Ray M (2009) Bioremediation of heavy metal toxicity-with special reference to chromium. Al Ameen J Med Sei 2:57–63
Richardson Y, Blin J, Volle G, Motuzas J, Julbe A (2010) In situ generation of Ni metal nanoparticles as catalyst for H2-rich syngas production from biomass gasification. Appl Catal A Gen 382:220–230. https://doi.org/10.1016/j.apcata.2010.04.047
Sathishkumar M, Sneha K, Yun YS (2010) Immobilization of silver nanoparticles synthesized using Curcuma longa tuber powder and extract on cotton cloth for bactericidal activity. Bioresour Technol 101:7958–7965. https://doi.org/10.1016/j.biortech.2010.05.051
Song Z, Li W, Liu W, Yang Y, Wang N, Wang H, Gao H (2015) Novel magnetic lignin composite sorbent for chromium(VI) adsorption. RSC Adv 5:13028–13035. https://doi.org/10.1039/c4ra15546g
Thakur S, Karak N (2014) One-step approach to prepare magnetic iron oxide/reduced graphene oxide nanohybrid for efficient organic and inorganic pollutants removal. Mater Chem Phys 144:425–432. https://doi.org/10.1016/j.matchemphys.2014.01.015
Vivek Narayanan N, Ganesan M (2009) Use of adsorption using granular activated carbon (GAC) for the enhancement of removal of chromium from synthetic wastewater by electrocoagulation. J Hazard Mater 161:575–580. https://doi.org/10.1016/j.jhazmat.2008.03.113
Yin W, Li Y, Wu J, Chen G, Jiang G, Li P, Gu J, Liang H, Liu C (2017) Enhanced Cr(VI) removal from groundwater by Fe0-H2O system with bio-amended iron corrosion. J Hazard Mater 332:42–50. https://doi.org/10.1016/j.jhazmat.2017.02.045
Yuan P, Liu D, Fan M, Yang D, Zhu R, Ge F, Zhu J, He H (2010) Removal of hexavalent chromium [Cr(VI)] from aqueous solutions by the diatomite-supported/unsupported magnetite nanoparticles. J Hazard Mater 173:614–621. https://doi.org/10.1016/j.jhazmat.2009.08.129
Zhou S, Li Y, Chen J, Liu Z, Wang Z, Na P (2014) Enhanced Cr(VI) removal from aqueous solutions using Ni/Fe bimetallic nanoparticles: characterization, kinetics and mechanism. RSC Adv 4:50699–50707. https://doi.org/10.1039/c4ra08754b
Zhou L, Li R, Zhang G, Wang D, Cai D, Wu Z (2018) 2018 Zero-valent iron nanoparticles supported by functionalized waste rock woolfor efficient removal of hexavalent chromium. Chem Eng J 339:85–96. https://doi.org/10.1016/j.cej.2018.01.132
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dalal, U., Reddy, S.N. A novel nano zero-valent iron biomaterial for chromium (Cr6+ to Cr3+) reduction. Environ Sci Pollut Res 26, 10631–10640 (2019). https://doi.org/10.1007/s11356-019-04528-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04528-0