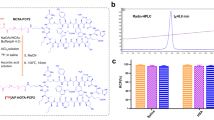
Abstract
Purpose
The poor tissue penetration of visible light has been a major barrier for optical imaging, photoactivatable conversions, and photodynamic therapy for in vivo targets with depths beyond 10 mm. In this report, as a proof-of-concept, we demonstrated that a positron emission tomography (PET) radiotracer, 2-deoxy-2-[18F]fluoro-d-glucose (18FDG), could be used as an alternative light source for photoactivation.
Procedures
We utilized 18FDG, which is a metabolic activity-based PET probe, as a source of light to photoactivate caged luciferin in a breast cancer animal model expressing luciferase.
Results
Bioluminescence produced from luciferin allowed for the real-time monitoring of Cherenkov radiation-promoted uncaging of the substrate.
Conclusion
The proposed method may provide a very important option for in vivo photoactivation, in particular for activation of photosensitizers for photodynamic therapy and eventually for combining radioisotope therapy and photodynamic therapy.




Similar content being viewed by others
References
MacCormack MA (2006) Photodynamic therapy. Adv Dermatol 22:219–258
Celli JP, Spring BQ, Rizvi I et al (2010) Imaging and photodynamic therapy: mechanisms, monitoring, and optimization. Chem Rev 110:2795–2838
Dolmans DE, Fukumura D, Jain RK (2003) Photodynamic therapy for cancer. Nat Rev Cancer 3:380–387
Robertson R, Germanos MS, Li C, Mitchell GS, Cherry SR, Silva MD (2009) Optical imaging of Cerenkov light generation from positron-emitting radiotracers. Phys Med Biol 54:N355–N365
Liu H, Ren G, Miao Z et al (2010) Molecular optical imaging with radioactive probes. PLoS One 5:e9470
Dothager RS, Goiffon RJ, Jackson E, Harpstrite S, Piwnica-Worms D (2010) Cerenkov radiation energy transfer (CRET) imaging: a novel method for optical imaging of PET isotopes in biological systems. PLoS One 5:e13300
Li C, Mitchell GS, Cherry SR (2010) Cerenkov luminescence tomography for small-animal imaging. Opt Lett 35:1109–1111
Liu H, Zhang X, Xing B, Han P, Gambhir SS, Cheng Z (2010) Radiation-luminescence-excited quantum dots for in vivo multiplexed optical imaging. Small 6:1087–1091
Ruggiero A, Holland JP, Lewis JS, Grimm J (2010) Cerenkov luminescence imaging of medical isotopes. J Nucl Med 51:1123–1130
Spinelli AE, D’Ambrosio D, Calderan L, Marengo M, Sbarbati A, Boschi F (2010) Cerenkov radiation allows in vivo optical imaging of positron emitting radiotracers. Phys Med Biol 55:483–495
Hu Z, Liang J, Yang W et al (2010) Experimental Cerenkov luminescence tomography of the mouse model with SPECT imaging validation. Opt Express 18:24441–24450
Lewis MA, Kodibagkar VD, Oz OK, Mason RP (2010) On the potential for molecular imaging with Cerenkov luminescence. Opt Lett 35:3889–3891
Mayer G, Heckel A (2006) Biologically active molecules with a “light switch”. Angew Chem Int Ed Engl 45:4900–4921
Adams SR, Tsien RY (1993) Controlling cell chemistry with caged compounds. Annu Rev Physiol 55:755–784
Yu H, Li J, Wu D, Qiu Z, Zhang Y (2010) Chemistry and biological applications of photo-labile organic molecules. Chem Soc Rev 39:464–473
Lee HM, Larson DR, Lawrence DS (2009) Illuminating the chemistry of life: design, synthesis, and applications of “caged” and related photoresponsive compounds. ACS Chem Biol 4:409–427
Fueger BJ, Czernin J, Hildebrandt I et al (2006) Impact of animal handling on the results of 18F-FDG PET studies in mice. J Nucl Med 47:999–1006
Abouzied MM, Crawford ES, Nabi HA (2005) 18F-FDG imaging: pitfalls and artifacts. J Nucl Med Technol 33:145–155
Hung JC (2002) Comparison of various requirements of the quality assurance procedures for (18)F-FDG injection. J Nucl Med 43:1495–1506
Shao Q, Jiang T, Ren G, Cheng Z, Xing B (2009) Photoactivable bioluminescent probes for imaging luciferase activity. Chem Commun (Camb) 27:4028–4030
Yang J, Thomason DB (1993) An easily synthesized, photolyzable luciferase substrate for in vivo luciferase activity measurement. Biotechniques 15:848–850
O’Connor AE, Gallagher WM, Byrne AT (2009) Porphyrin and nonporphyrin photosensitizers in oncology: preclinical and clinical advances in photodynamic therapy. Photochem Photobiol 85:1053–1074
Berg K, Selbo PK, Weyergang A et al (2005) Porphyrin-related photosensitizers for cancer imaging and therapeutic applications. J Microsc 218:133–147
Kaiser PK (2007) Verteporfin photodynamic therapy and anti-angiogenic drugs: potential for combination therapy in exudative age-related macular degeneration. Curr Med Res Opin 23:477–487
Pshenichnov I, Larionov A, Mishustin I, Greiner W (2007) PET monitoring of cancer therapy with 3He and 12 C beams: a study with the GEANT4 toolkit. Phys Med Biol 52:7295–7312
Conflict of Interest Statement
A.M. declares the payment for lectures outside the submitted work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
SI Fig. 1
The LC–MS spectra of a water solution of DMNP-luciferin irradiated with UV 365 nm light for 5 min. Insert mass spectra of the released luciferin (left) and the unreacted DMNP-luciferin (right). (PDF 27 kb)
SI Fig. 2
a Bioluminescence images of mice bearing luciferase-expressing tumors injected with luciferin. b Quantitative analysis of the images in (a). (PDF 602 kb)
SI Fig. 3
Full-time course optical signal from tumor sites of control mice injected with 18FDG only. (PDF 58 kb)
Rights and permissions
About this article
Cite this article
Ran, C., Zhang, Z., Hooker, J. et al. In Vivo Photoactivation Without “Light”: Use of Cherenkov Radiation to Overcome the Penetration Limit of Light. Mol Imaging Biol 14, 156–162 (2012). https://doi.org/10.1007/s11307-011-0489-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-011-0489-z