Abstract
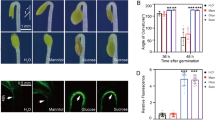
Plants are often subjected to unfavorable growing conditions, such as a limited supply of nutrients. Macroautophagy (autophagy) is an intracellular recycling process that targets unwanted proteins or damaged organelles, thus providing the nitrogen and carbon needed for survival and new plant growth during different phases of development and nutrient deprivation. During the formation of autophagosomes, AuTophaGy-related protein 7 (ATG7) is involved in both the ATG8 and ATG12 conjugation pathways. We isolated two ATG7 orthologs—MdATG7a and MdATG7b—from ‘Golden Delicious’ apple (Malus domestica Borkh.). Both were differentially responsive to leaf senescence, nitrogen depletion, and oxidative stresses at the transcriptional level. Heterologous expression of MdATG7b in Arabidopsis accelerated vegetative growth and facilitated the transition into reproductive phase, while also increasing the number of lateral branches, alleviating the negative effects of salt stress on seed germination and conferring tolerance to nitrogen- and carbon starvation. These results suggest that, as one of the important autophagy-related genes, MdATG7b enhances plant tolerance to nitrogen and carbon limitations, and it might be used as an important candidate for breeding crops with improved tolerance to nutrient-starvation stress.







Similar content being viewed by others
Abbreviations
- AD:
-
Adenylation domain
- ATG:
-
AuTophaGy-related protein
- C:
-
Carbon
- CDS:
-
Coding DNA sequence
- DTT:
-
Dithiothreitol
- ER:
-
Endoplasmic reticulum
- Kan:
-
Kanamycin
- LD:
-
Long days
- MV:
-
Methyl viologen
- N:
-
Nitrogen
- ORF:
-
Open reading frame
- qRT-PCR:
-
Quantitative real-time PCR
- SD:
-
Short days
References
Avila-Ospina L, Moison M, Yoshimoto K, Masclaux-Daubresse C (2014) Autophagy, plant senescence, and nutrient recycling. J Exp Bot 65:3799–3811. doi:10.1093/jxb/eru039
Avin-Wittenberg T, Bajdzienko K, Wittenberg G, Alseekh S, Tohge T, Bock R, Giavalisco P, Fernie AR (2015) Global analysis of the role of autophagy in cellular metabolism and energy homeostasis in Arabidopsis seedlings under carbon starvation. Plant Cell 27:306–322. doi:10.1105/tpc.114.134205
Bai T, Li C, Ma F, Shu H, Han M, Wang K (2008) Physiological responses and analysis of tolerance of apple rootstocks to root-zone hypoxia stress. Sci Agric Sin 41:4140–4148
Bassham DC (2009) Function and regulation of macroautophagy in plants. Biochim Biophys Acta 1793:1397–1403. doi:10.1016/j.bbamcr.2009.01.001
Breeze E, Harrison E, McHattie S, Hughes L, Hickman R, Hill C, Kiddle S, Kim YS, Penfold CA, Jenkins D, Zhang C, Morris K, Jenner C, Jackson S, Thomas B, Tabrett A, Legaie R, Moore JD, Wild DL, Ott S, Rand D, Beynon J, Denby K, Mead A, Buchanan-Wollaston V (2011) High-resolution temporal profiling of transcripts during Arabidopsis leaf senescence reveals a distinct chronology of processes and regulation. Plant Cell 23:873–894. doi:10.1105/tpc.111.083345
Brouquisse R, Gaudillère JP, Raymond P (1998) Induction of a carbon-starvation-related proteolysis in whole maize plants submitted to light/dark cycles and to extended darkness. Plant Physiol 117:1281–1291. doi:10.1104/pp.117.4.1281
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116. doi:10.1007/BF02670468
Chapin FS III, Walter CH, Clarkson DT (1988) Growth response of barley and tomato to nitrogen stress and its control by abscisic acid, water relations and photosynthesis. Planta 173:352–366. doi:10.1007/BF00401022
Chung T, Suttangkakul A, Vierstra RD (2009) The ATG autophagic conjugation system in maize: ATG transcripts and abundance of the ATG8-lipid adduct are regulated by development and nutrient availability. Plant Physiol 149:220–234. doi:10.1104/pp.108.126714
Chung T, Allison RP, Vierstra RD (2010) ATG8 lipidation and ATG8-mediated autophagy in Arabidopsis require ATG12 expressed from the differentially controlled ATG12A and ATG12B loci. Plant J 62:483–493. doi:10.1111/j.1365-313X.2010.04166.x
Doelling JH, Walker JM, Friedman EM, Thompson AR, Vierstra RD (2002) The APG8/12-activating enzyme APG7 is required for proper nutrient recycling and senescence in Arabidopsis thaliana. J Biol Chem 277:33105–33114. doi:10.1074/jbc.M204630200
Escobar-Gutiérrez AJ, Daudet F, Gaudillère J, Maillard P, Frossard JS (1998) Modeling of allocation and balance of carbon in walnut (Juglans regia L.) seedling during heterotrophy-autotrophy transition. J Theor Biol 194:29–47. doi:10.1006/jtbi.1998.0740
Farmer LM, Rinaldi MA, Young PG, Danan CH, Burkhart SE, Bartel B (2013) Disrupting autophagy restores peroxisome function to an Arabidopsis lon2 mutant and reveals a role for the LON2 protease in peroxisomal matrix protein degradation. Plant Cell 25:4085–4100. doi:10.1105/tpc.113.113407
Guiboileau A, Sormani R, Meyer C, Masclaux-Daubresse C (2010) Senescence and death of plant organs: nutrient recycling and developmental regulation. CR Biol 333:382–391. doi:10.1016/j.crvi.2010.01.016
Hanaoka H, Noda T, Shirano Y, Kato T, Hayashi H, Shibata D, Tabata S, Ohsumi Y (2002) Leaf senescence and starvation-induced chlorosis are accelerated by the disruption of an Arabidopsis autophagy gene. Plant Physiol 129:1181–1193. doi:10.1104/pp.011024
He CC, Klionsky DJ (2009) Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet 43:67–93. doi:10.1146/annurev-genet-102808-114910
Hofius D, Schultz-Larsen T, Joensen J, Tsitsigiannis DI, Petersen NH, Mattsson O, Jorgensen LB, Jones JDG, Mundy J, Petersen M (2009) Autophagic components contribute to hypersensitive cell death in Arabidopsis. Cell 137:773–783. doi:10.1016/j.cell.2009.02.036
Htwe NMPS, Tanigawa H, Ishibashi Y, Zheng S, Yuasa T, Iwaya-Inoue M (2009) Nutrient starvation differentially regulates the autophagy-related gene GmATG8i in soybean seedlings. Plant Biotechnol 26:317–326. http://doi.org/10.5511/plantbiotechnology.26.317
Jordan MO (2015) C depletion and tree dieback in young peach trees: a possible consequence of N shortage? Ann Forest Sci 72:529–537. doi:10.1007/s13595-015-0466-9
Klionsky DJ, Emr SD (2000) Autophagy as a regulated pathway of cellular degradation. Science 290:1717–1721. doi:10.1126/science.290.5497.1717
Klionsky DJ, Codogno P, Cuervo AM, Deretic V, Elazar Z, Fueyo-Margareto J, Gewirtz DA, Kroemer G, Levine B, Mizushima N, Rubinsztein DC, Thumm M, Tooze SA (2010) A comprehensive glossary of autophagy-related molecules and processes. Autophagy 6:438–448. doi:10.4161/auto.6.4.12244
Kurusu T, Koyano T, Hanamata S, Kubo T, Noguchi Y, Yagi C, Nagata N, Yamamoto T, Ohnishi T, Okzaki Y, Kitahata N, Ando D, Ishikawa M, Wada S, Miyao A, Hirochika H, Shimada H, Makino A, Saito K, Ishida H, Kinoshita T, Kurata N, Kuchitsu K (2014) OsATG7 is required for autophagy-dependent lipid metabolism in rice postmeiotic anther development. Autophagy 10:878–888. doi:10.4161/auto.28279
Kwon SI, Cho HJ, Park OK (2010) Role of Arabidopsis RabG3b and autophagy in tracheary element differentiation. Autophagy 6:1187–1189. doi:10.4161/auto.6.8.13429
Lee I, Schindelin H (2008) Structural insights into E1-catalyzed ubiquitin activation and transfer to conjugating enzymes. Cell 134:268–278. doi:10.1016/j.cell.2008.05.046
Li F, Vierstra RD (2012) Autophagy: a multifaceted intracellular system for bulk and selective recycling. Trends Plant Sci 17:526–537. doi:10.1016/j.tplants.2012.05.006
Li F, Chung T, Pennington JG, Federico ML, Kaeppler HF, Kaeppler SM, Otegui MS, Vierstra RD (2015) Autophagy recycling plays a central role in maize nitrogen remobilization. Plant Cell 27:1389–1408. doi:10.1105/tpc.15.00158
Liu T (2008) Expression pattern and functional analysis of an autophagy-related gene MgATG4 in Magnaporthe oryzae. Dissertation, Zhejiang University
Liu Y, Bassham DC (2012) Autophagy: pathways for self-eating in plant cells. Annu Rev Plant Biol 63:215–237. doi:10.1146/annurev-arplant-042811-105441
Liu Y, Schiff M, Czymmek K, Talloczy Z, Levine B, Dinesh-Kumar SP (2005) Autophagy regulates programmed cell death during the plant innate immune response. Cell 121:567–577. doi:10.1016/j.cell.2005.03.007
Liu Y, Xiong Y, Bassham DC (2009) Autophagy is required for tolerance of drought and salt stress in plants. Autophagy 5:954–963. doi:10.4161/auto.5.7.9290
Liu Y, Burgos JS, Deng Y, Srivastava R, Howell SH, Bassham DC (2012) Degradation of the endoplasmic reticulum by autophagy during endoplasmic reticulum stress in Arabidopsis. Plant Cell 24:4635–4651. doi:10.1105/tpc.112.101535
Ma L, Li Y, Chen Y, Li X (2016) Improved drought and salt tolerance of Arabidopsis thaliana by ectopic expression of a cotton (Gossypium hirsutum) CBF gene. Plant Cell Tissue Organ Cult (PCTOC) 124:583–598. doi:10.1007/s11240-015-0917-x
Minina EA, Sanchez-Vera V, Moschou PN, Suarez MF, Sundberg E, Weih M, Bozhkov PV (2013) Autophagy mediates caloric restriction-induced lifespan extension in Arabidopsis. Aging Cell 12:327–329. doi:10.1111/acel.12048
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873. doi:10.1101/gad.1599207
Mizushima N, Noda T, Yoshimori T, Tanaka Y, Ishii T, George MD, Klionsky DJ, Ohsumi M, Ohsumi Y (1998) A protein conjugation system essential for autophagy. Nature 395:395–398. doi:10.1038/26506
Mizushima N, Yoshimori T, Ohsumi Y (2011) The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol 27:107–132. doi:10.1146/annurev-cellbio-092910-154005
Noda NN, Inagaki F (2015) Mechanisms of autophagy. Annu Rev Biophys 44:101–122. doi:10.1146/annurev-biophys-060414-034248
Noda NN, Satoo K, Fujioka Y, Kumeta H, Ogura K, Nakatogawa H, Ohsumi Y, Inagaki F (2011) Structural basis of Atg8 activation by a homodimeric E1, Atg7. Mol Cell 44:462–475. doi:10.1016/j.molcel.2011.08.035
Patel MK, Joshi M, Mishra A, Jha B (2015) Ectopic expression of SbNHX1 gene in transgenic castor (Ricinus communis L.) enhances salt stress by modulating physiological process. Plant Cell Tissue Organ Cult (PCTOC) 122:477–490. doi:10.1007/s11240-015-0785-4
Phillips AR, Suttangkakul A, Vierstra RD (2008) The ATG12-conjugating enzyme ATG10 is essential for autophagic vesicle formation in Arabidopsis thaliana. Genetics 178:1339–1353. doi:10.1534/genetics.107.086199
Ravikumar B, Sarkar S, Davies JE, Futter M, Garcia-Arencibia M, Green-Thompson ZW, Jimenez-Sanchez M, Korolchuk VI, Lichtenberg M, Luo S, Massey DC, Menzies FM, Moreau K, Narayanan U, Renna M, Siddiqi FH, Underwood BR, Winslow AR, Rubinsztein DC (2010) Regulation of mammalian autophagy in physiology and pathophysiology. Physiol Rev 90:1383–1435. doi:10.1152/physrev.00030.2009
Ren C, Liu J, Gong Q (2014) Functions of autophagy in plant carbon and nitrogen metabolism. Front Plant Sci 5:301. doi:10.3389/fpls.2014.00301
Rose TL, Bonneau L, Der C, Marty-Mazars D, Marty F (2006) Starvation-induced expression of autophagy-related genes in Arabidopsis. Biol Cell 98:53–67. doi:10.1042/BC20040516
Shibata M, Oikawa K, Yoshimoto K, Kondo M, Mano S, Yamada K, Hayashi M, Sakamoto W, Ohsumi Y, Nishimura M (2013) Highly oxidized peroxisomes are selectively degraded via autophagy in Arabidopsis. Plant Cell 25:4967–4983. doi:10.1105/tpc.113.116947
Shin JH, Yoshimoto K, Ohsumi Y, Jeon JS, An G (2009) OsATG10b, an autophagosome component, is needed for cell survival against oxidative stresses in rice. Mol Cells 27:67–74. doi:10.1007/s10059-009-0006-2
Slavikova S, Ufaz S, Avin-Wittenberg T, Levanony H, Galili G (2008) An autophagy-associated Atg8 protein is involved in the responses of Arabidopsis seedlings to hormonal controls and abiotic stresses. J Exp Bot 59:4029–4043. doi:10.1093/jxb/ern244
Taherbhoy AM, Tait SW, Kaiser SE, Williams AH, Deng A, Nourse A, Hammel M, Kurinov I, Rock CO, Green DR, Schulman BA (2011) Atg8 transfer from Atg7 to Atg3: a distinctive E1-E2 architecture and mechanism in the autophagy pathway. Mol Cell 44:451–461. doi:10.1016/j.molcel.2011.08.034
Tamirisa S, Reddy VD, Rao KV (2014) Ectopic expression of pigeonpea cold and drought regulatory protein (CcCDR) in yeast and tobacco affords multiple abiotic stress tolerance. Plant Cell Tissue Organ Cult (PCTOC) 119:489–499. doi:10.1007/s11240-014-0549-6
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739. doi:10.1093/molbev/msr121
Thomas H (2013) Senescence, ageing and death of the whole plant. New Phytol 197:696–711. doi:10.1111/nph.12047
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. doi:10.1093/nar/22.22.4673
Thompson AR, Doelling JH, Suttangkakul A, Vierstra RD (2005) Autophagic nutrient recycling in Arabidopsis directed by the ATG8 and ATG12 conjugation pathways. Plant Physiol 138:2097–2110. doi:10.1104/pp.105.060673
Tu M, Wang X, Huang L, Guo R, Zhang H, Cai J, Wang X (2016) Expression of a grape bZIP transcription factor, VqbZIP39, in transgenic Arabidopsis thaliana confers tolerance of multiple abiotic stresses. Plant Cell Tissue Organ Cult (PCTOC) 125:537–551. doi:10.1007/s11240-016-0969-6
van der Graaff E, Schwacke R, Schneider A, Desimone M, Flugge UI, Kunze R (2006) Transcription analysis of Arabidopsis membrane transporters and hormone pathways during developmental and induced leaf senescence. Plant Physiol 141:776–792. doi:10.1104/pp.106.079293
Wang Y, Yu B, Zhao J, Guo J, Li Y, Han S, Huang L, Du Y, Hong Y, Tang D, Liu Y (2013) Autophagy contributes to leaf starch degradation. Plant Cell 25:1383–1399. doi:10.1105/tpc.112.108993
Wang Y, Cai S, Yin L, Shi K, Xia X, Zhou Y, Yu J, Zhou J (2015) Tomato HsfA1a plays a critical role in plant drought tolerance by activating ATG genes and inducing autophagy. Autophagy 11:2033–2047. doi:10.1080/15548627.2015.1098798
Wang P, Sun X, Jia X, Wang N, Gong X, Ma F (2016) Characterization of an autophagy-related gene MdATG8i from apple. Front Plant Sci 7:720. doi:10.3389/fpls.2016.00720
Weigl K, Flachowsky H, Peil A, Hanke MV (2015) Heat mediated silencing of MdTFL1 genes in apple (Malus × domestica). Plant Cell Tissue Organ Cult (PCTOC) 123:511–521. doi:10.1007/s11240-015-0855-7
Wu J, Ji J, Wang G, Wu G, Diao J, Li Z, Chen X, Chen Y, Luo L (2015) Ectopic expression of the Lycium barbarum β-carotene hydroxylase gene (chyb) enhances drought and salt stress resistance by increasing xanthophyll cycle pool in tobacco. Plant Cell Tissue Organ Cult (PCTOC) 121:559–569. doi:10.1007/s11240-015-0725-3
Xia K, Liu T, Ouyang J, Wang R, Fan T, Zhang M (2011) Genome-wide identification, classification, and expression analysis of autophagy-associated gene homologues in rice (Oryza sativa L.). DNA Res 18:363–377. doi:10.1093/dnares/dsr024
Xia T, Xiao D, Liu D, Chai W, Gong Q, Wang N (2012) Heterologous expression of ATG8c from soybean confers tolerance to nitrogen deficiency and increases yield in Arabidopsis. PLoS One 7:e37217. doi:10.1371/journal.pone.0037217
Xiong Y, Contento AL, Bassham DC (2005) AtATG18a is required for the formation of autophagosomes during nutrient stress and senescence in Arabidopsis thaliana. Plant J 42:535–546. doi:10.1111/j.1365-313X.2005.02397.x
Xiong Y, Contento AL, Nguyen PQ, Bassham DC (2007) Degradation of oxidized proteins by autophagy during oxidative stress in Arabidopsis. Plant Physiol 143:291–299. doi:10.1104/pp.106.092106
Yang ZF, Klionsky DJ (2009) An overview of the molecular mechanism of autophagy. Curr Top Microbiol Immunol 335:1–32. doi:10.1007/978-3-642-00302-8_1
Yang X, Srivastava R, Howell SH, Bassham DC (2016) Activation of autophagy by unfolded proteins during endoplasmic reticulum stress. Plant J 85:83–95. doi:10.1111/tpj.13091
Yorimitsu T, Nair U, Yang Z, Klionsky DJ (2006) Endoplasmic reticulum stress triggers autophagy. J Biol Chem 281:30299–30304. doi:10.1074/jbc.M607007200
Yoshimoto K, Jikumaru Y, Kamiya Y, Kusano M, Consonni C, Panstruga R, Ohsumi Y, Shirasu K (2009) Autophagy negatively regulates cell death by controlling NPR1-dependent salicylic acid signaling during senescence and the innate immune response in Arabidopsis. Plant Cell 21:2914–2927. doi:10.1105/tpc.109.068635
Yoshimoto K, Takano Y, Sakai Y (2010) Autophagy in plants and phytopathogens. FEBS Lett 584:1350–1358. doi:10.1016/j.febslet.2010.01.007
Yoshimoto K, Shibata M, Kondo M, Oikawa K, Sato M, Toyooka K, Shirasu K, Nishimura M, Ohsumi Y (2014) Organ-specific quality control of plant peroxisomes is mediated by autophagy. J Cell Sci 127:1161–1168. doi:10.1242/jcs.139709
Zhang X, Henriques R, Lin SS, Niu QW, Chua NH (2006) Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat Protoc 1:641–646. doi:10.1038/nprot.2006.97
Zhao Q, Sun C, Liu D, Hao Y, You C (2015) Ectopic expression of the apple Md-miR172e gene alters flowering time and floral organ identity in Arabidopsis. Plant Cell Tissue Organ Cult (PCTOC) 123:535–546. doi:10.1007/s11240-015-0857-5
Zhou X, Zhao P, Wang W, Zou J, Cheng T, Peng X, Sun M (2015) A comprehensive, genome-wide analysis of autophagy-related genes identified in tobacco suggests a central role of autophagy in plant response to various environmental cues. DNA Res 22:245–257. doi:10.1093/dnares/dsv012
Acknowledgments
This work was supported by the Young Scientists Fund of the National Natural Science Foundation of China (31601735) and by the earmarked fund for the China Agriculture Research System (CARS-28).
Author contributions
P. W. and F. W. M. conceived and designed the experiments; P. W. and X. S. performed the experiments; N. W. and X. J. helped in collecting data; P. W. wrote the manuscript; F. W. M. critically revised the article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Ping Wang and Xun Sun have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11240_2016_1070_MOESM2_ESM.tif
Supplementary Fig. 2 Yeast complementary test in atg7 mutant with MdATG7a and MdATG7b under nitrogen-starvation conditions. (TIF 844 KB)
11240_2016_1070_MOESM3_ESM.tif
Supplementary Fig. 3 Immunoblot analysis of the accumulation of MdATG7b-GFP fusion protein in 2-week-old Arabidopsis seedlings of WT and MdATG7b OE lines (OE-2, -4, -6, -12) with anti-GFP antibody. Protein extract loading amount was referenced by immunoblot analysis with an anti-Actin antibody. (TIF 174 KB)
Rights and permissions
About this article
Cite this article
Wang, P., Sun, X., Wang, N. et al. Ectopic expression of an autophagy-associated MdATG7b gene from apple alters growth and tolerance to nutrient stress in Arabidopsis thaliana . Plant Cell Tiss Organ Cult 128, 9–23 (2017). https://doi.org/10.1007/s11240-016-1070-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-1070-x