Abstract
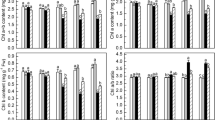
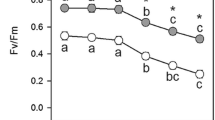
Reactive oxygen species (ROS) and calcium (Ca2+), two crucial intracellular signaling molecules, have been reported to play important roles in chlorophyll biosynthesis. In this study, we aimed to investigate whether disturbance of chlorophyll synthesis affects chloroplast ROS and Ca2+ homeostases. Chlorophyll biosynthesis was inhibited at the Mg branch by virus-induced gene silencing (VIGS) of CHLI gene encoding the Mg chelatase CHLI subunit in pea (Pisum sativum). Subsequently, ROS and intracellular free Ca2+ concentration ([Ca2+]i) in these chlorophyll-deficient pea plants were evaluated by histochemical and fluorescent staining assays. The results showed that the superoxide anion and hydrogen peroxide were predominantly generated in chloroplasts of the yellow leaves of pea VIGS-CHLI plants. The expression of genes encoding chloroplast antioxidant enzymes (CuZn-superoxide dismutase, ascorbate peroxidase, glutathione reductase, phospholipid glutathione peroxidase, peroxiredoxin and thioredoxins) were also decreased in the leaves of VIGS-CHLI plants compared with the control plants. Additionally, the [Ca2+]i were significantly reduced in the yellow leaves of VIGS-CHLI plants compared with the green leaves of VIGS-GFP control plants. The expression of genes encoding Ca2+ signaling related proteins (thylakoid Ca2+ transporter, calmodulins and calcineurin B-like protein) was down-regulated in yellow VIGS-CHLI leaves. These results indicate that inhibition of chlorophyll biosynthesis at the Mg branch by silencing CHLI affects chloroplast ROS homeostasis and Ca2+ signaling and down-regulates the expression of ROS scavenging genes and Ca2+ signaling related genes.





Similar content being viewed by others
References
Abbasi BH, Khan M, Guo B, Bokhari SA, Khan MA (2011) Efficient regeneration and antioxidative enzyme activities in Brassica rapa var. turnip. Plant Cell Tissue Organ Cult 105:337–344
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. doi:10.1146/annurev.arplant.55.031903.141701
Asada K (2006) Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol 141:391–396. doi:10.1104/pp.106.082040
Balsera M et al (2009) Characterization of Tic110, a channel-forming protein at the inner envelope membrane of chloroplasts, unveils a response to Ca2+ and a stromal regulatory disulfide bridge. J Biol Chem 284:2603–2616. doi:10.1074/jbc.M807134200
Chigri F, Hormann F, Stamp A, Stammers DK, Bolter B, Soll J, Vothknecht UC (2006) Calcium regulation of chloroplast protein translocation is mediated by calmodulin binding to Tic32. Proc Natl Acad Sci USA 103:16051–16056. doi:10.1073/pnas.0607150103
Dietz KJ (2011) Peroxiredoxins in plants and cyanobacteria. Antioxid Redox Signal 15:1129–1159. doi:10.1089/ars.2010.3657
Dietz KJ, Pfannschmidt T (2011) Novel regulators in photosynthetic redox control of plant metabolism and gene expression. Plant Physiol 155:1477–1485. doi:10.1104/pp.110.170043
Fey V, Wagner R, Bräutigam K, Pfannschmidt T (2005) Photosynthetic redox control of nuclear gene expression. J Exp Bot 56:1491–1498. doi:10.1093/jxb/eri180
Hoffmann A, Hammes E, Plieth C, Desel C, Sattelmacher B, Hansen UP (2005) Effect of CO2 supply on formation of reactive oxygen species in Arabidopsis thaliana. Protoplasma 227:3–9. doi:10.1007/s00709-005-0133-3
Ifuku K, Ishihara S, Sato F (2010) Molecular functions of oxygen-evolving complex family proteins in photosynthetic electron flow. J Integr Plant Biol 52:723–734. doi:10.1111/j.1744-7909.2010.00976.x
Ikegami A et al (2007) The CHLI1 subunit of Arabidopsis thaliana magnesium chelatase is a target protein of the chloroplast thioredoxin. J Biol Chem 282:19282–19291. doi:10.1074/jbc.M703324200
Kong S-G, Wada M (2011) New insights into dynamic actin-based chloroplast photorelocation movement. Mol Plant 4:771–781. doi:10.1093/mp/ssr061
Li J, Wang D-Y, Li Q, Xu Y-J, Cui K-M, Zhu Y-X (2004) PPF1 inhibits programmed cell death in apical meristems of both G2 pea and transgenic Arabidopsis plants possibly by delaying cytosolic Ca2+ elevation. Cell Calcium 35:71–77. doi:10.1016/j.ceca.2003.07.003
Libik-Konieczny M, Konieczny R, Ewa Surówka E, Ireneusz Ślesak I, Michalec Ż, Rozpądek P, Miszalski Z (2012) Pathways of ROS homeostasis regulation in Mesembryanthemum crystallinum L. calli exhibiting differences in rhizogenesis. Plant Cell Tissue Organ Cult 110:123–131. doi:10.1007/s11240-012-0136-7
Luo T et al (2012) Thioredoxin redox regulates ATPase activity of magnesium chelatase CHLI subunit and modulates redox-mediated signaling in tetrapyrrole biosynthesis and homeostasis of reactive oxygen species in pea plants. Plant Physiol 159:118–130
Luo T et al (2013) Virus-induced gene silencing of pea CHLI and CHLD affects tetrapyrrole biosynthesis, chloroplast development and the primary metabolic network. Plant Physiol Biochem 65:17–26. doi:10.1016/j.plaphy.2013.01.006
Mahajan S, Sopory SK, Tuteja N (2006) Cloning and characterization of CBL-CIPK signalling components from a legume (Pisum sativum). FEBS J 273:907–925. doi:10.1111/j.1742-4658.2006.05111.x
Mazars C, Thuleau P, Lamotte O, Bourque S (2010) Cross-talk between ROS and calcium in regulation of nuclear activities. Mol Plant 3:706–718. doi:10.1093/mp/ssq024
Nomura H, Shiina T (2014) Calcium signaling in plant endosymbiotic organelles: mechanism and role in physiology. Mol Plant 7:1094–1104
Nomura H, Komori T, Kobori M, Nakahira Y, Shiina T (2008) Evidence for chloroplast control of external Ca2+-induced cytosolic Ca2+ transients and stomatal closure. Plant J 53:988–998. doi:10.1111/j.1365-313X.2007.03390.x
Nomura H et al (2012) Chloroplast-mediated activation of plant immune signalling in Arabidopsis. Nat Commun 3:926. doi:10.1038/ncomms1926
Nott A, Jung H-S, Koussevitzky S, Chory J (2006) Plastid-to-nucleus retrograde signaling. Annu Rev Plant Biol 57:739–759. doi:10.1146/annurev.arplant.57.032905.105310
Papenbrock J, Pfündel E, Mock H-P, Grimm B (2000) Decreased and increased expression of the subunit CHL I diminishes Mg chelatase activity and reduces chlorophyll synthesis in transgenic tobacco plants. Plant J 22:155–164. doi:10.1046/j.1365-313x.2000.00724.x
Peter E, Rothbart M, Oelze M-L, Shalygo N, Dietz K-J, Grimm B (2010) Mg protoporphyrin monomethylester cyclase deficiency and effects on tetrapyrrole metabolism in different light conditions. Plant Cell Physiol 51:1229–1241. doi:10.1093/pcp/pcq071
Pfannschmidt T, Allen JF, Oelmüller R (2001) Principles of redox control in photosynthesis gene expression. Physiol Plant 112:1–9. doi:10.1034/j.1399-3054.2001.1120101.x
Pospisil P (2012) Molecular mechanisms of production and scavenging of reactive oxygen species by photosystem II. Biochim Biophys Acta (BBA)-Bioenerg 1817:218–231. doi:10.1016/j.bbabio.2011.05.017
Rocha AG, Vothknecht UC (2012) The role of calcium in chloroplasts—an intriguing and unresolved puzzle. Protoplasma 249:957–966. doi:10.1007/s00709-011-0373-3
Smith CM, Liu J, Davies E (1995) Sequencing and Analysis of a Calmodulin cDNA from Pea (Pisum sativum L. var Alaska). Plant Physiol 108:435–436. doi:10.1104/pp.108.1.435
Stael S, Wurzinger B, Mair A, Mehlmer N, Vothknecht UC, Teige M (2012) Plant organellar calcium signalling: an emerging field. J Exp Bot 63:1525–1542. doi:10.1093/jxb/err394
Steinhorst L, Kudla J (2013) Calcium and reactive oxygen species rule the waves of signaling. Plant Physiol 163:471–485. doi:10.1104/pp.113.222950
Stenbaek A, Jensen PE (2010) Redox regulation of chlorophyll biosynthesis. Phytochemistry 71:853–859. doi:10.1016/j.phytochem.2010.03.022
Sundberg E, Slagter JG, Fridborg I, Cleary SP, Robinson C, Coupland G (1997) ALBINO3, an Arabidopsis nuclear gene essential for chloroplast differentiation, encodes a chloroplast protein that shows homology to proteins present in bacterial membranes and yeast mitochondria. Plant Cell 9:717–730
Tanaka A, Tsuji H (1980) Effects of calcium on chlorophyll synthesis and stability in the early phase of greening in cucumber cotyledons. Plant Physiol 65:1211–1215
Weinl S, Held K, Schlucking K, Steinhorst L, Kuhlgert S, Hippler M, Kudla J (2008) A plastid protein crucial for Ca2+-regulated stomatal responses. New Phytol 179:675–686. doi:10.1111/j.1469-8137.2008.02492.x
Zakharov SD, Ewy RG, Dilley RA (1993) Subunit III of the chloroplast ATP-synthase can form a Ca2+ -binding site on the lumenal side of the thylakoid membrane. FEBS Lett 336:95–99. doi:10.1016/0014-5793(93)81617-9
Acknowledgments
This work was supported by National Natural Science Foundation of China (Grant No. 31360050) and the Natural Science Foundation of Jiangxi, China (Grant No. 20151BAB214004).
Author contribution
SL and TL designed the experiments. TL, PP, YPL and XGL performed the experiments. SL and TL analyzed the raw data. SL and TL wrote the manuscript. All authors read and approved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, S., Luo, T., Peng, P. et al. Disturbance of chlorophyll biosynthesis at Mg branch affects the chloroplast ROS homeostasis and Ca2+ signaling in Pisum sativum . Plant Cell Tiss Organ Cult 127, 729–737 (2016). https://doi.org/10.1007/s11240-016-1008-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-1008-3