Abstract
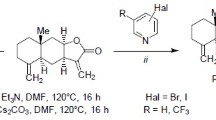
The Heck reaction of the eudesman-type methylidenelactones (alantolactone, alloalantolactone, and 4,15-epoxyisoalantolactone) with haloarenes afforded the corresponding (E)-13-aryleudesma-4(15),11(13)-dien-8β,12-olides and 11-arylmethyl-13-noreudesma-4(15),7(11)-dien-8α,12-olides. The yields and the ratios of the arylation products depended on the reaction conditions and the structure of lactone. Certain side processes were found to take place.
Similar content being viewed by others
References
A. V. Belovodskii, E. E. Shul’ts, M. M. Shakirov, I. Yu. Bagryanskaya, Yu. V. Gatilov, G. A. Tolstikov, Zh. Org. Khim., 2010, 46, 1710 [Russ. J. Org. Chem. (Engl. Transl.), 2010, 46, 1719].
A. V. Belovodskii, E. E. Shul’ts, M. M. Shakirov, G. A. Tolstikov, Dokl. Akad. Nauk, 2009, 426, 762 [Dokl. Chem. (Engl. Transl.), 2009].
A. V. Belovodskii, E. E. Shul’ts, M. M. Shakirov, V. E. Romanov, B. Zh. Elmuradov, Kh. M. Shakhidoyatov, G. A. Tolstikov, Khim. Prirod. Soedin., 2010, 747 [Chem. Nat. Compd. (Engl. Transl.), 2010, 46, 880].
S. Zhang, Y. K. Wong, C. N. Ong, H. M. Shen, Curr. Med. Chem. Anticancer Agents, 2005, 5, 239.
Pat. No. 2413724 RF, E. E. Shul’ts, A. V. Belovodskii, T. G. Tolstikova, M. P. Dolgikh, E. A. Morozova, G. A. Tolstikov, Byul. isobret. [Invention Bull.], 2011, No. 7, 10 (in Russian).
G. Schlewer, J. L. Stampf, C. Benezra, J. Med. Chem., 1980, 23, 1031.
S. G. Klochkov, S. V. Afanas’eva, A. N. Pushchin, Khim. Prirod. Soedin., 2006, 325 [Chem. Nat. Compd. (Engl. Transl.), 2006, 42, 400].
M. Larhed, A. Hallberg, The Heck Reaction, in Handbook of Organopalladium Chemistry for Organic Synthesis, Wiley, New York, 2002, p. 1133; I. P. Beletskaya, A. V. Cheprakov, Chem. Rev., 2000, 100, 3009.
C. Han, F. J. Barrios, M. V. Riofski, D. A. Colby, J. Org. Chem., 2009, 74, 7176.
A. Arcadi, M. Chiarini, F. Marinelli, Z. Berente, L. Kollar, Org. Lett., 2000, 2, 69; A. Arcadi, M. Chiarini, F. Marinelli, Z. Berente, L. Kollar, Eur. J. Org. Chem., 2001, 16, 3165.
R. Matusch, H. Häberlein, Liebigs Ann. Chem., 1987, 455.
H. Erdman, L. Westfeld, Acta Chem. Scand., 1963, 17, 1826.
S. C. Srivastava, M. M. Mehra, G. K. Trivedi, S. C. Bhattacharyy, Ind. J. Chem., 1971, 9, 512.
S. T. Caldwell, H. M. Petersson, L. J. Farrugia, W. Mullen, A. Crozier, R. C. Hartley, Tetrahedron, 2006, 62, 7257.
V. K. Chaikovskii, A. N. Novikov, Zh. Prikl. Khim., 1984, 57, 134 [Russ. J. Appl. Chem. (Engl. Transl.), 1984, 57].
J. Pavlinac, M. Zupan, S. Stavber, J. Org. Chem., 2006, 71, 1027.
G. B. Shul’pin, Organicheskie reaktsii, kataliziruemye kompleksami metallov [Organic Reactions Catalyzed by Metal Complexes], Nauka, Moscow, 1988, 275 pp. (in Russian).
Author information
Authors and Affiliations
Corresponding author
Additional information
For Part 5, see Ref. 1.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 1959–1968, October, 2012.
Rights and permissions
About this article
Cite this article
Shul’ts, E.E., Belovodskii, A.V., Shakirov, M.M. et al. Synthetic transformations of sesquiterpene lactones 6. Alantolactone and isoalantolactone derivatives in the Heck reaction. Russ Chem Bull 61, 1975–1985 (2012). https://doi.org/10.1007/s11172-012-0274-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-012-0274-4