Abstract
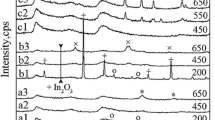
In this paper, chromium borophosphates have been prepared by the solution combustion method using different types of fuel and enhanced as a catalyst for liquid-phase oxidation of benzyl alcohol to benzaldehyde for the first time. Carbohydrazide, citric acid, hexamethylenetetramine, oxalyldihydrazide and urea used as an organic fuel and the obtained chromium borophosphates were systematically characterized by infrared spectroscopy, X-ray powder diffraction, thermogravimetry, scanning electron microscopy, and surface analysis techniques. They are structurally identical and thermally stable up to 1000 °C. According to fuel, their surface properties differ from each other and the long and short rod-like particles, which have a surface area between 7 and 19 m2 g−1, are formed. The effects of solvent type, reaction temperature, reaction time, peroxide amount and catalyst amount were also examined for catalytic studies, and the reaction conditions were optimized. As compared with the other fuels, hexamethylenetetramine-assisted synthesized chromium borophosphate catalyst provides high benzaldehyde conversion (52.09%) and selectivity (100%) under the optimized condition and has potential as a heterogeneous catalyst for oxidation reactions.




Similar content being viewed by others
References
M. Li, A. Verena-Mudring, Cryst. Growth Des. 16, 2441 (2016)
B. Han, P. Li, J. Zhang, H. Shi, J. Lumin. 155, 15 (2014)
S. Panda, P. Vinodkumar, U. Madhusoodanan, B. Panigrahi, Luminescence 34, 887 (2019)
N. Yamnova, S. Aksenov, E.Y. Borovikova, A. Volkov, O. Gurbanova, O. Dimitrova, P. Burns, Crystallogr. Rep. 64, 228 (2019)
M. Hassan, F. Ahmad, Z.A. El-Fattah, J. Alloys Compd. 750, 320 (2018)
Y.-Q. Feng, Z. Li, Z. Zhong, H. Wang, Dalton Trans. 49, 1388 (2020).
E. Belokoneva, T. Eremina, D. Koshchug, O. Dimitrova, A. Volkov, Solid State Sci. 98, 106013 (2019)
R.K. Guntu, J. Lumin. 209, 258 (2019)
L.C. Belusso, G.F. Lenz, E.E. Fiorini, A.J. Pereira, R. Sequinel, R.A. Bini, J.F. Felix, R. Schneider, Appl. Surf. Sci. 473, 303 (2019)
M. Seshadri, M. Radha, M. Bell, V. Anjos, Ceram. Int. 44, 20790 (2018)
H. ElBatal, A. El-Kheshen, N. Ghoneim, M. Marzouk, F. ElBatal, A. Fayad, A. Abdelghany, A. El-Beih, Silicon 11, 197 (2019)
Y.H. Matzkeit, B.L. Tornquist, F. Manarin, G.V. Botteselle, J. Rafique, S. Saba, A.L. Braga, J.F. Felix, R. Schneider, J. Non Cryst. Solids 498, 153 (2018)
J. Zhu, J. Xiang, D. Yang, Z. Fang, Y. Zheng, Y. Mao, H. Zhao, Ceram. Int. 46, 844 (2020)
J. Zhu, M. Yang, Y. Che, Z. Fang, Y. Zheng, D. Yang, Y. Mao, K. Xiong, J. Liao, J. Alloys Compd. 822, 153606 (2020)
K. Sakasai, M. Katagiri, M. Matsubayashi, T. Nakamura, Y. Kondo, Nuclear Instrum. Methods Phys. Res Sect. A Accel. Spectrom. Detect. Assoc. Equip. 529, 378 (2004)
N. Chanthima, Y. Tariwong, N. Kiwsakunkran, J. Kaewkhao, Effects of BaO and Bi2O3 on the optical and luminescence properties of Dy3+ doped borophosphate glasses, in Journal of Physics: Conference Series, IOP Publishing, 2019, p. 012003.
S. Karki, C. Kesavulu, H. Kim, J. Kaewkhao, N. Chanthima, S. Kothan, S. Kaewjaeng, J. Non Cryst. Solids 521, 119483 (2019)
S.M. Aziz, R. Umar, N.M. Yusoff, S.J.M. Rosid, S.N.S.M. Amin, Int. J. Sci. Technol. Res. 8, 1976 (2019)
P.W. Menezes, A. Indra, I. Zaharieva, C. Walter, S. Loos, S. Hoffmann, R. Schlögl, H. Dau, M. Driess, Energy Environ. Sci. 12, 988 (2019)
W. Liu, L. Zhang, G. Su, L.-X. Cao, Y.-G. Wang, Dalton Trans. 39, 7262 (2010)
G. Elmaci, D. Ozer, B. Zumreoglu-Karan, Catal. Commun. 89, 56 (2017)
N. Dimitratos, J.A. Lopez-Sanchez, D. Morgan, A. Carley, L. Prati, G.J. Hutchings, Catal. Today 122, 317 (2007)
J. Luo, F. Peng, H. Yu, H. Wang, Chem. Eng. J. 204, 98 (2012)
S. Akbari, J. Mokhtari, Z. Mirjafari, RSC Adv. 7, 40881 (2017)
M. Salavati-Niasari, J. Mol. Catal. A Chem. 245, 192 (2006)
G. Zhan, Y. Hong, F. Lu, A.-R. Ibrahim, M. Du, D. Sun, J. Huang, Q. Li, J. Li, J. Mol. Catal. A Chem. 366, 215 (2013)
V. Choudhary, D. Dumbre, B. Uphade, V. Narkhede, J. Mol. Catal. A Chem. 215, 129 (2004)
C.-C. Guo, Q. Liu, X.-T. Wang, H.-Y. Hu, Appl. Catal. A 282, 55 (2005)
V.R. Choudhary, P.A. Chaudhari, V.S. Narkhede, Catal. Commun. 4, 171 (2003)
Z. Weng, G. Liao, J. Wang, X. Jian, Catal. Commun. 8, 1493 (2007)
D. Ozer, T. Simsek, S. Ozcan, N.A. Oztas, Ceram. Int. 46, Part B, 12238 (2020)
S. Jain, K. Adiga, V.P. Verneker, Combust. Flame 40, 71 (1981)
F.-R. Xiu, Y. Lu, Y. Qi, Chemosphere 249, 126138 (2020)
F.-R. Xiu, Y. Wang, X. Yu, Y. Li, Y. Lu, K. Zhou, J. He, Z. Song, X. Gao, Sci. Total Environ. 708, 134532 (2020)
A. Baykal, G. Gözel, M. Kizilyalli, M. Toprak, R. Kniep, Turk J Chem 24, 381 (2000)
J.-X. Mi, J.-T. Zhao, S.-Y. Mao, Y.-X. Huang, H. Engelhardt, R. Kniep, Zeitschrift für Kristallographie New Cryst. Struct. 215, 201 (2000)
P. Erri, P. Pranda, A. Varma, Ind. Eng. Chem. Res. 43, 3092 (2004)
K. Frikha, L. Limousy, J. Bouaziz, S. Bennici, K. Chaari, M. Jeguirim, C. R. Chim. 22, 206 (2019)
M. Aghayan, M. Rodríguez, Mater. Sci. Eng. C 32, 2464 (2012)
V. Mahdavi, M. Mardani, Res. Chem. Intermed. 41, 8907 (2015)
D. Hoare, J. Protheroe, A. Walsh, Trans. Faraday Soc. 55, 548 (1959)
S.D. Kr, N. Venkatesha, J. Aust. Ceram. Soc. 56, 217 (2020)
S. Oudi, A.R. Oveisi, S. Daliran, M. Khajeh, E. Teymoori, J. Colloid Interface Sci. 561, 782 (2020)
Y. Yu, B. Lu, X. Wang, J. Zhao, X. Wang, Q. Cai, Chem. Eng. J. 162, 738 (2010)
N.T. Thao, N.T. Nhu, K.-S. Lin, J Taiwan Inst. Chem. Eng. 83, 10 (2018)
J. Muzart, Chem. Rev. 92, 113 (1992)
A.L. Cánepa, V.R. Elías, V.M. Vaschetti, E.V. Sabre, G.A. Eimer, S.G. Casuscelli, Appl. Catal. A 545, 72 (2017)
Z. Miao, J. Zhou, Z. Li, D. Liu, J. Zhao, L. Chou, S. Zhuo, J. Mater. Sci. 52, 12141 (2017)
L. Jing, J. Shi, F. Zhang, Y. Zhong, W. Zhu, Ind. Eng. Chem. Res. 52, 10095 (2013)
L. Xiong, R. Chen, F. Chen, RSC Adv. 6, 101048 (2016)
C.Y. Ma, B.J. Dou, J.J. Li, J. Cheng, Q. Hu, Z.P. Hao, S.Z. Qiao, Appl. Catal. B 92, 202 (2009)
Acknowledgements
The author is grateful to Prof. Dr. Nursen Altuntas-Oztas (Hacettepe University) for allowing the use of the laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The author declares no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ozer, D. Liquid phase oxidation of benzyl alcohol to benzaldehyde over chromium borophosphate catalyst synthesized by solution combustion method using different types of fuel. Res Chem Intermed 46, 3449–3458 (2020). https://doi.org/10.1007/s11164-020-04155-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04155-2