Abstract
Background and aims
The endophytic fungus Epichloë bromicola forms mutualistic symbiotic associations with wild barley (Hordeum brevisubulatum) in the saline-alkali areas of northwestern China. E. bromicola enhances the tolerance of H. brevisubulatum to salt stress. Because plant polyamine metabolism is closely related to microbial infection and tolerance to diverse abiotic stresses, we hypothesized that in symbiotic plants polyamine modification may result from E. bromicola infection, and that improved tolerance to abiotic stress by the presence of this endophyte might be related to polyamine modification. Our focus in this study was to investigate whether E. bromicola affects polyamine metabolism in host plants under salt stress.
Methods
E. bromicola infected (E+) and endophyte free (E-) wild barley plants were subjected to NaCl treatments (0, 100, 200 and 300 mM). Dry weight, diamine putrescine (Put), triamine spermidine (Spd) and tetramine spermine (Spm) content and the content of their free, soluble conjugated and insoluble bound forms were measured after 21 d exposure to stress.
Results
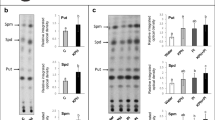
E. bromicola infection led to significant amelioration of salt stress in H. brevisubulatum. The presence of the endophyte significantly increased dry weight, spermidine and spermine content, but decreased putrescine content and the putrescine: (spermidine + spermine) ratio. E. bromicola infection also lowered the proportion of putrescine in total polyamines, but increased the proportion of spermidine and spermine in total polyamines. Furthermore, E. bromicola infection significantly increased the proportion of insoluble bound forms of polyamines, and decreased the proportion of free forms of polyamines and soluble conjugated forms of polyamines.
Conclusions
H. brevisubulatum salinity stress tolerance induced by E. bromicola infection correlated with enhanced conversion of putrescine to spermidine and spermine, as well as improved shift ability from free forms and soluble conjugated forms of polyamines to insoluble bound forms of polyamines.









Similar content being viewed by others
References
Alcázar R, Marco F, Cuevas JC, Patron M, Ferrando A, Carrasco P, Tiburcio AF, Altabella T (2006) Involvement of polyamines in plant response to abiotic stress. Biotechnol Lett 28:1867–1876. https://doi.org/10.1007/s10529-006-9179-3
Asthir B, Spoor W, Duffus CM (2004) Involvement of polyamines, diamine oxidase and polyamine oxidase in resistance of barley to Blumeria graminis f. sp. Hordei. Euphytica 136:307–312. https://doi.org/10.1023/B:EUPH.0000032730.48474.b1
Aziz A, Martin-Tanguy J, Larher F (1998) Stress-induced changes in polyamine and tyramine levels can regulate proline accumulation in tomato leaf discs treated with sodium chloride. Physiol Plant 104:195–202. https://doi.org/10.1034/j.1399-3054.1998.1040207.x
Besford RT, Richardson CM, Campos JL, Tiburcio AF (1993) Effect of polyamines on stabilization of molecular complexes in thylakoid membranes of osmotically stressed oat leaves. Planta 189:201–206. https://doi.org/10.1007/BF00195077
Bose J, Rodrigomoreno A, Shabala S (2014) ROS homeostasis in halophytes in the context of salinity stress tolerance. J Exp Bot 65:1241–1257. https://doi.org/10.1093/jxb/ert430
Bouchereau A, Aziz A, Larher F, Martin-Tanguy J (1999) Polyamines and environmental challenges: recent development. Plant Sci 140:103–125. https://doi.org/10.1016/S0168-9452(98)00218-0
Bratton DL (1994) Polyamine inhibition of transbilayer movement of plasma membrane phospholipids in the erythrocyte ghost. J Biol Chem 269:22517–22523
Chattopadhayay MK, Tiwari BS, Chattopadhyay G, Bose A, Sengupta DN, Ghosh B (2002) Protective role of exogenous polyamines on salinity-stressed rice ( Oryza sativa ) plants. Physiol Plant 116:192–199. https://doi.org/10.1034/j.1399-3054.2002.1160208.x
Chen TX, Simpson WR, Song QY, Chen SH, Li CJ, Ahmad RZ (2018) Identification of Epichloë endophytes associated with wild barley (Hordeum brevisubulatum) and characterisation of their alkaloid biosynthesis. New Zeal J Agr Res. https://doi.org/10.1080/00288233.2018.1461658
Christensen MJ, Bennett RJ, Ansari HA, Koga H, Johnson RD, Bryan GT, Simpson WR, Koolaard JP, Nickless EM, Voisey CR (2008) Epichloë endophytes grow by intercalary hyphal extension in elongating grass leaves. Fungal Genet Biol 45(2):84–93. https://doi.org/10.1016/j.fgb.2007.07.013
Duan J, Li J, Guo S, Kang Y (2008) Exogenous spermidine affects polyamine metabolism in salinity-stressed Cucumis sativus roots and enhances short-term salinity tolerance. J Plant Physiol 165:1620–1635. https://doi.org/10.1016/j.jplph.2007.11.006
Edreva A (1997) Tobacco polyamines as affected by stresses induced by different pathogens. Biol Plant 40:317–320. https://doi.org/10.1023/a:1001093209229
Ekanayake PN, Hand ML, Spangenberg GC, Forster JW, Guthridge KM (2012) Genetic diversity and host specificity of fungal endophyte taxa in fescue pasture grasses. Crop Sci 52:2243–2252. https://doi.org/10.2135/cropsci2011.12.0664
Erdei L, Szegletes Z, Barabás K, Pestenácz A (1996) Responses in polyamine titer under osmotic and salt stress in sorghum and maize seedlings. J Plant Physiol 147:599–603. https://doi.org/10.1016/S0176-1617(96)80052-6
Fiscaletti D, Angeli D, Tarozzi L, Barozzi GS (2013) Plant polyamines in abiotic stress responses. Acta Physiol Plant 35:2015–2036. https://doi.org/10.1016/j.ijheatmasstransfer.2012.11.039
Flowers TJ (2004) Improving crop salt tolerance. J Exp Bot 55:307–319. https://doi.org/10.1093/jxb/erh003
Fu XZ, Huang Y, Xing F, Chun CP, Ling LL, Cao L, Peng LZ (2016) Changes in free polyamines and expression of polyamine metabolic genes under drought and high-temperature in Citrus sinensis. Biol Plant 60:1–6. https://doi.org/10.1007/s10535-016-0636-0
Ghachtouli NE, Martin-Tanguy J, Paynot M, Gianinazzi S (1996) First-report of the inhibition of arbuscular mycorrhizal infection of Pisum sativum by specific and irreversible inhibition of polyamine biosynthesis or by gibberellic acid treatment. FEBS Lett 385:189–192. https://doi.org/10.1016/0014-5793(96)00379-1
Greenland AJ, Lewis DH (1984) Amines in barley leaves infected by brown rust and their possible relevance to formation of 'green islands'. New Phytol 96:283–291. https://doi.org/10.1111/j.1469-8137.1984.tb03565.x
Groppa MD, Benavides MP (2008) Polyamines and abiotic stress: recent advances. Amino Acids 34:35–45. https://doi.org/10.1007/s00726-007-0501-8
Gundel PE, Martínez-Ghersa MA, Omacini M, Cuyeu R, Pagano E, Ríos R, Ghersa CM (2012) Mutualism effectiveness and vertical transmission of symbiotic fungal endophytes in response to host genetic background. Evol Appl 5:838–849. https://doi.org/10.1111/j.1752-4571.2012.00261.x
Gupta B, Huang B (2014) Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization. Int J Genomics 2014:701596. https://doi.org/10.1155/2014/701596
Gupta K, Dey A, Gupta B (2013) Plant polyamines in abiotic stress responses. Acta Physiol Plant 35:2015–2036. https://doi.org/10.1007/s11738-013-1239-4
Hettiarachchige IK, Ekanayake PN, Mann RC, Guthridge KM, Sawbridge TI, Spangenberg GC, Forster JW (2015) Phylogenomics of asexual Epichloë fungal endophytes forming associations with perennial ryegrass. BMC Evol Biol 15:72. https://doi.org/10.1186/s12862-015-0349-6
Hu X, Zhang Y, Shi Y, Zhang Z, Zou Z, Zhang H, Zhao J (2012) Effect of exogenous spermidine on polyamine content and metabolism in tomato exposed to salinity-alkalinity mixed stress. Plant Physiol Biochem 57:200–209. https://doi.org/10.1016/j.plaphy.2012.05.015
Iannone LJ, Vignale MV, Pinget AD, Re A, Cargo PDM, Novas MV (2017) Seed-transmitted Epichloë sp. endophyte alleviates the negative effects of head smut of grasses (Ustilago bullata) on Bromus auleticus. Fungal Ecol 29:45–51. https://doi.org/10.1016/j.funeco.2017.06.001
Ioannidis NE, Cruz JA, Kotzabasis K, Kramer DM (2012) Evidence that putrescine modulates the higher plant photosynthetic proton circuit. PLoS One 7:e29864. https://doi.org/10.1371/journal.pone.0029864
Kasukabe Y, He L, Nada K, Misawa S, Ihara I, Tachibana S (2004) Overexpression of spermidine synthase enhances tolerance to multiple environmental stresses and up-regulates the expression of various stress-regulated genes in transgenic Arabidopsis thaliana. Plant Cell Physiol 45:712–722. https://doi.org/10.1093/pcp/pch083
Krishnamurthy R, Bhagwat KA (1989) Polyamines as modulators of salt tolerance in rice cultivars. Plant Physiol 91:500–504. https://doi.org/10.1104/pp.91.2.500
Kubi J (2005) The effect of exogenous spermidine on superoxide dismutase activity, H2O2 and superoxide radical level in barley leaves under water deficit conditions. Acta Physiol Plant 27:289–295. https://doi.org/10.1007/s11738-005-0005-7
Kuldau G, Bacon C (2008) Clavicipitaceous endophytes: their ability to enhance resistance of grasses to multiple stresses. Biol Control 46:57–71. https://doi.org/10.1016/j.biocontrol.2008.01.023
Kusano T, Berberich T, Tateda C, Takahashi Y (2008) Polyamines: essential factors for growth and survival. Planta 228:367–381. https://doi.org/10.1007/s00425-008-0772-7
Kuznetsov VV, Shevyakova NI (2007) Polyamines and stress tolerance of plants. Plant Stress 1:50–71
Kytöviita MM, Sarjala T (1997) Effects of defoliation and symbiosis on polyamine levels in pine and birch. Mycorrhiza 7:107–111. https://doi.org/10.1007/s005720050170
Leuchtmann A, Bacon CW, Schardl CL, Jr WJ, Tadych M (2014) Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 106:202–215. https://doi.org/10.3852/106.2.202
Ma M, Christensen MJ, Nan Z (2015) Effects of the endophyte Epichloë festucae var. lolii of perennial ryegrass ( Lolium perenne ) on indicators of oxidative stress from pathogenic fungi during seed germination and seedling growth. Eur J Plant Pathol 141:571–583. https://doi.org/10.1007/s10658-014-0563-x
Malinowski DP, Belesky DP (2000) Adaptations of endophyte-infected cool-season grasses to environmental stresses: mechanisms of drought and mineral stress tolerance. Crop Sci 40:923–940. https://doi.org/10.2135/cropsci2000.404923x
Margosiak SA, Dharma A, Brucecarver MR, Gonzales AP, Louie D, Kuehn GD (1990) Identification of the large subunit of ribulose 1,5-bisphosphate carboxylase/oxygenase as a substrate for transglutaminase in Medicago sativa L. (alfalfa). Plant Physiol 92:88–96. https://doi.org/10.1104/pp.92.1.88
Martin-Tanguy J (1997) Conjugated polyamines and reproductive development: biochemical, molecular and physiological approaches. Physiol Plant 100:675–688. https://doi.org/10.1111/j.1399-3054.1997.tb03074.x
Miller G, Shulaev V, Mittler R (2008) Reactive oxygen signaling and abiotic stress. Physiol Plant 133:481–489. https://doi.org/10.1111/j.1399-3054.2008.01090.x
Monnet F, Vaillant N, Hitmi A, Coudret A, Sallanon H (2001) Endophytic Neotyphodium lolii induced tolerance to Zn stress in Lolium perenne. Physiol Plant 113:557–563. https://doi.org/10.1034/j.1399-3054.2001.1130415.x
Moon CD, Miles CO, Järlfors U, Schardl CL (2002) The evolutionary origins of three new Neotyphodium endophyte species from grasses indigenous to the southern hemisphere. Mycologia 94:694–711. https://doi.org/10.2307/3761720
Mutlu F, Bozcuk S (2005) Effects of salinity on the contents of polyamines and some other compounds in sunflower plants differing in salt tolerance. Russ J Plant Physiol 52:29–34. https://doi.org/10.1007/s11183-005-0005-x
Negrel J, Lherminier J (1987) Peroxidase-mediated integration of tyramine into xylem cell walls of tobacco leaves. Planta 172:494–501. https://doi.org/10.1007/BF00393865
Oberhofer M, Güsewell S, Leuchtmann A (2014) Effects of natural hybrid and non-hybrid Epichloë endophytes on the response of Hordelymus europaeus to drought stress. New Phytol 201:242–253. https://doi.org/10.1111/nph.12496
Omar MNA, Osman MEH, Kasim WA, El-Daim IAA (2009) Improvement of salt tolerance mechanisms of barley cultivated under salt stress using Azospirillum brasilense. In: Ashraf M, Ozturk M, Athar HR (eds) Salinity and water stress: improving crop efficiency. Springer, Dordrecht, pp 133–147
Pál M, Kovács V, Vida G, Szalai G, Janda T (2011) Changes in salicylic acid and polyamine contents following powdery mildew infection of near-isogenic thatcher-based wheat lines carrying different Lr genes. Acta Biol Szeged 55:139–141
Pál M, Szalai G, Janda T (2015) Speculation: polyamines are important in abiotic stress signaling. Plant Sci 237:16–23. https://doi.org/10.1016/j.plantsci.2015.05.003
Parádi I, Bratek Z, Láng F (2003) Influence of arbuscular mycorrhiza and phosphorus supply on polyamine content, growth and photosynthesis of Plantago lanceolata. Biol Plant 46:563–569. https://doi.org/10.1023/A:1024819729317
Pottosin I, Shabala S (2014) Polyamines control of cation transport across plant membranes: implications for ion homeostasis and abiotic stress signaling. Front Plant Sci 5:154. https://doi.org/10.3389/fpls.2014.00154
Prakash L, Prathapasenan G (1988) Effect of NaCl salinity and putrescine on shoot growth, tissue ion concentration and yield of rice (Oryza sativa L. var. GR-3). J Agron Crop Sci 160:325–334. https://doi.org/10.1111/j.1439-037X.1988.tb00630.x
Reza SM, Mirlohi A (2010) Neotyphodium endophytes trigger salt resistance in tall and meadow fescues. J Plant Nutr Soil Sci 173:952–957. https://doi.org/10.1002/jpln.200900345
Rodriguez RJ, Henson J, Volkenburgh EV, Hoy M, Wright L, Beckwith F, Kim YO, Redman RS (2008) Stress tolerance in plants via habitat-adapted symbiosis. ISME J 2:404–416. https://doi.org/10.1038/ismej.2007.106
Roy SJ, Negrão S, Tester M (2014) Salt resistant crop plants. Curr Opin Biotechnol 26:115–124. https://doi.org/10.1016/j.copbio.2013.12.004
Roychoudhury A, Basu S, Sengupta DN (2011) Amelioration of salinity stress by exogenously applied spermidine or spermine in three varieties of indica rice differing in their level of salt tolerance. J Plant Physiol 168:317–328. https://doi.org/10.1016/j.jplph.2010.07.009
Rozpądek P, Rozpadek P, Wezowicz K, Nosek M, Wazny R, Tokarz K, Lembicz M, Miszalski Z, Turnau K (2015) The fungal endophyte Epichloë typhina improves photosynthesis efficiency of its host orchard grass (Dactylis glomerata). Planta 242:1025–1035. https://doi.org/10.1007/s00425-015-2337-x
Saha J, Brauer EK, Sengupta A, Popescu SC, Gupta K, Gupta B (2015) Polyamines as redox homeostasis regulators during salt stress in plants. Front Environ Sci 3:21. https://doi.org/10.3389/fenvs.2015.00021
Saikkonen K, Wäli P, Helander M, Faeth SH (2004) Evolution of endophyte-plant symbioses. Trends Plant Sci 9:275–280. https://doi.org/10.1016/j.tplants.2004.04.005
Saikkonen K, Lehtonen P, Helander M, Koricheva J, Faeth SH (2006) Model systems in ecology: dissecting the endophyte-grass literature. Trends Plant Sci 11:428–433. https://doi.org/10.1016/j.tplants.2006.07.001
Saikkonen K, Gundel PE, Helander M (2013) Chemical ecology mediated by fungal endophytes in grasses. J Chem Ecol 39:962–968. https://doi.org/10.1007/s10886-013-0310-3
Sannazzaro AI, Echeverría M, Albertó EO, Ruiz OA, Menéndez AB (2007) Modulation of polyamine balance in Lotus glaber by salinity and arbuscular mycorrhiza. Plant Physiol Biochem 45:39–46. https://doi.org/10.1016/j.plaphy.2006.12.008
Santa-Cruz A, Acosta M, Pérez-Alfocea F, Bolarin MC (1997) Changes in free polyamine levels induced by salt stress in leaves of cultivated and wild tomato species. Physiol Plant 101:341–346. https://doi.org/10.1111/j.1399-3054.1997.tb01006.x
Schardl CL, Young CA, Hesse U, Amyotte SG, Andreeva K, Calie PJ, Fleetwood DJ, Haws DC, Moore N, Oeser B, Panaccione DG, Schweri KK, Voisey CR, Farman ML, Jaromczyk JW, Roe BA, O'Sullivan DM, Scott B, Tudzynski P, An Z, Arnaoudova EG, Bullock CT, Charlton ND, Chen L, Cox M, Dinkins RD, Florea S, Glenn AE, Gordon A, Güldener U, Harris DR, Hollin W, Jaromczyk J, Johnson RD, Khan AK, Leistner E, Leuchtmann A, Li C, Liu JG, Liu J, Liu M, Mace W, Machado C, Nagabhyru P, Pan J, Schmid J, Sugawara K, Steiner U, Takach JE, Tanaka E, Webb JS, Wilson EV, Wiseman JL, Yoshida R, Zeng Z (2013) Plant-symbiotic fungi as chemical engineers: multi-genome analysis of the Clavicipitaceae reveals dynamics of alkaloid loci. PLoS Genet 9:e1003323. https://doi.org/10.1371/journal.pgen.1003323
Shabala S, Demidchik V, Shabala L, Cuin TA, Smith SJ, Miller AJ, Davies JM, Newman IA (2006) Extracellular Ca2+ ameliorates NaCl-induced K+ loss from arabidopsis root and leaf cells by controlling plasma membrane K+-permeable channels. Plant Physiol 141:1653–1665. https://doi.org/10.1104/pp.106.082388
Shabala S, Cuin TA, Pottosin I (2007) Polyamines prevent NaCl-induced K+ efflux from pea mesophyll by blocking non-selective cation channels. FEBS Lett 581:1993–1999. https://doi.org/10.1016/j.febslet.2007.04.032
Smith G, Cloutt BA, Lynch DE, Byriel KA, Kennard CHL (1998) Influence of arbuscular mycorrhizae and rhizobium on free polyamines and proline levels in water-stressed alfalfa. J Plant Physiol 153:706–711. https://doi.org/10.1016/S0176-1617(98)80224-1
Song ML (2015) Mechanisms of salt tolerance improved by Epichloë endophyte in wild barley. Dissertation: Lanzhou University
Song ML, Chai Q, Li XZ, Yao X, Li CJ, Christensen MJ, Nan ZB (2015a) An asexual Epichloë endophyte modifies the nutrient stoichiometry of wild barley (Hordeum brevisubulatum) under salt stress. Plant Soil 387:153–165. https://doi.org/10.1007/s11104-014-2289-0
Song ML, Li XZ, Saikkonen K, Li CJ, Nan ZB (2015b) An asexual Epichloë endophyte enhances waterlogging tolerance of Hordeum brevisubulatum. Fungal Ecol 13:44–52. https://doi.org/10.1016/j.funeco.2014.07.004
Soto-Barajas MC, Zabalgogeazcoa I, Gómez-Fuertes J, González-Blanco V, Vázquez-De-Aldana BR (2016) Epichloë endophytes affect the nutrient and fiber content of Lolium perenne regardless of plant genotype. Plant Soil 405:265–277. https://doi.org/10.1007/s11104-015-2617-z
Tanaka A, Christensen M, Takemoto D, Park P, Scott B (2006) Reactive oxygen species play a role in regulating a fungus-perennial ryegrass mutualistic interaction. Plant Cell 18:1052–1066. https://doi.org/10.1105/tpc.105.039263
Tiburcio AF, Altabella T, Bitrián M, Alcázar R (2014) The roles of polyamines during the lifespan of plants: from development to stress. Planta 240:1–18. https://doi.org/10.1007/s00425-014-2055-9
Tytti S, Seppo K (2002) Potassium nutrition and free polyamines of Betula pendula Roth and Betula pubescens Ehrh. Plant Soil 238:141–149. https://doi.org/10.1023/A:1014262213688
Waie B, Rajam MV (2003) Effect of increased polyamine biosynthesis on stress responses in transgenic tobacco by introduction of human S-adenosylmethionine gene. Plant Sci 164:727–734. https://doi.org/10.1016/S0168-9452(03)00030-X
Walters DR (2000) Polyamines in plant-microbe interactions. Physiol Mol Plant Pathol 57:137–146. https://doi.org/10.1006/pmpp.2000.0286
Walters DR (2003) Polyamines and plant disease. Phytochemistry 64:97–107. https://doi.org/10.1016/S0031-9422(03)00329-7
Walters DR, Wylie MA (1986) Polyamines in discrete regions of barley leaves infected with the powdery mildew fungus, Erysiphe graminis. Physiol Plant 67:630–633. https://doi.org/10.1111/j.1399-3054.1986.tb05068.x
Wang CM, Xia ZR, Wu GQ, Yuan HJ, Wang XR, Li JH, Tian FP, Zhang Q, Zhu XQ, He JJ, Kumar T, Wang XL, Zhang JL (2016) The coordinated regulation of Na+ and K+ in Hordeum brevisubulatum responding to time of salt stress. Plant Sci 252:358–366. https://doi.org/10.1016/j.plantsci.2016.08.009
White JF Jr, Torres MS (2010) Is plant endophyte-mediated defensive mutualism the result of oxidative stress protection? Physiol Plant 138:440–446. https://doi.org/10.1111/j.1399-3054.2009.01332.x
Wimalasekera R, Tebartz F, Scherer GF (2011) Polyamines, polyamine oxidases and nitric oxide in development, abiotic and biotic stresses. Plant Sci 181:593–603. https://doi.org/10.1016/j.plantsci.2011.04.002
Xia C, Zhang XX, Christensen MJ, Nan ZB, Li CJ (2015) Epichloë endophyte affects the ability of powdery mildew (Blumeria graminis) to colonise drunken horse grass (Achnatherum inebrians). Fungal Ecol 16:26–33. https://doi.org/10.1016/j.funeco.2015.02.003
Xia C, Li NN, Zhang XX, Feng Y, Christensen MJ, Nan ZB (2016) An Epichloë endophyte improves photosynthetic ability and dry matter production of its host Achnatherum inebrians infected by Blumeria graminis under various soil water conditions. Fungal Ecol 22:26–34. https://doi.org/10.1016/j.funeco.2016.04.002
Yamaguchi Y, Sano H (2003) Induction of hypersensitive cell death by hydrogen peroxide produced through polyamine degradation in tobacco plants. Plant Physiol 132:1973–1981. https://doi.org/10.1104/pp.103.024737
Yamasaki H, Cohen MF (2006) NO signal at the crossroads: polyamine-induced nitric oxide synthesis in plants? Trends Plant Sci 11:522–524. https://doi.org/10.1016/j.tplants.2006.09.009
Zapata PJ, Serrano M, Pretel MT, Amoros A, Botella MA (2004) Polyamines and ethylene changes during germination of different plant species under salinity. Plant Sci 167:781–788. https://doi.org/10.1016/j.plantsci.2004.05.014
Zepeda-Jazo I, Shabala S, Chen Z, Pottosin II (2008) Na+-K+ transport in roots under salt stress. Plant Signal Behav 3:401–403. https://doi.org/10.4161/psb.3.6.5429
Zepeda-Jazo I, Velarde-Buendia AM, Enriquez-Figueroa R, Bose J, Shabala S, Muniz-Murguia J, Pottosin II (2011) Polyamines interact with hydroxyl radicals in activating Ca2+ and K+ transport across the root epidermal plasma membranes. Plant Physiol 157:2167–2180. https://doi.org/10.1104/pp.111.179671
Zhang XX, Li CJ, Nan ZB (2010) Effects of cadmium stress on growth and anti-oxidative systems in Achnatherum inebrians symbiotic with Neotyphodium gansuense. J Hazard Mater 175:703–709. https://doi.org/10.1016/j.jhazmat.2009.10.066
Zhao F, Song CP, He J, Zhu H (2007) Polyamines improve K+/Na+ homeostasis in barley seedlings by regulating root ion channel activities. Plant Physiol 145:1061–1072. https://doi.org/10.1104/pp.107.105882
Funding
The study was supported by National Basic Research Program of China (2014CB138702), the Natural Science Foundation of China (31372366), the Strategic Priority Research Program of Chinese Academy of Sciences (XDAXDA20100102), Program for Changjiang Scholars and Innovative Research Team in University of China (IRT17R50), Fundamental Research Funds for the Central Universities (LZUJBKY-2018-kb10), 111Project (B12002), USDA-NIFA Multistate Project W3147 and the New Jersey Agricultural Experiment Station.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Disclosures
The article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
This study did not involve human participants, and so informed consent was not required.
Additional information
Responsible Editor: Birgit Mitter.
Rights and permissions
About this article
Cite this article
Chen, T., Li, C., White, J.F. et al. Effect of the fungal endophyte Epichloë bromicola on polyamines in wild barley (Hordeum brevisubulatum) under salt stress. Plant Soil 436, 29–48 (2019). https://doi.org/10.1007/s11104-018-03913-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-018-03913-x