Abstract
Background and aims
Rhizosphere influences the dynamics of nutrients and contaminants through increased microbial activity, release of root exudates and alteration of pH. The objective of this study was to evaluate the rhizosphere-induced reduction (i.e. rhizoreduction) and redistribution of arsenate [As(V)] and chromate [Cr(VI)] in Australian native vegetation in relation to their bioavailability.
Methods
The reduction of As(V) and Cr(VI) was examined using rhizosphere soils from a number of Australian native vegetation (Acacia pubescens, Eucalyptus camaldulensis, Enchylaena tomentosa, Templetonia retusa, Dichantheum sericeum and Austrodanthonia richardsonii). Naturally contaminated As and Cr soils were used to examine the effect of Dichantheum sericeum on the redistribution and bioavailability of these metal(loid)s.
Results
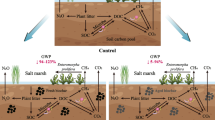
The rhizosphere soil contained higher levels of microbial activity, dissolved organic carbon and organic acid content than the non-rhizosphere soil. The rhizosphere soil caused up to 2.4 and 5.1 fold increases in the rate of reduction of As(V) and Cr(VI), respectively. There was a significant relationship between rhizosphere-induced increases in microbial activity (Δ basal respiration) and As(V) and Cr(VI) reduction (Δ rate of reduction), indicating the role of increased microbial activity in rhizosphere soil on metal(loid) reduction. In the plant growth experiment, Dichantheum sericeum enhanced the reduction of metal(loid)s in the naturally contaminated soils, thereby increasing the bioavailability of As but decreasing that of Cr.
Conclusions
Depending on the nature of metal(loid)s present in soil, the rhizosphere-induced reduction by plant species such as Dichantheum sericeum and Templetonia retusa has implications to both their bioavailability to higher plants and microorganisms, and remediation of contaminated soils. While rhizoreduction decreases Cr bioavailability it increases that of As.


Similar content being viewed by others
References
Adriano DC (2001) Trace elements in terrestrial environments: biogeochemistry, bioavailability and risks of metals, 2nd edn. Springer, New York
Alexander M (1999) Biodegradation and bioremediation, 2nd edn. Academic, San Diego
Amofah LR, Maurice C, Kumpiene J, Bhattacharya P (2011) The influence of temperature, pH/molarity and extractant on the removal of arsenic, chromium and zinc from contaminated soil. J Soil Sediment 11:1334–1344
Anderson TA, Coats JR (1995) An overview of microbial degradation in the rhizosphere and its implications to bioremediation. In: Skipper HD, Turco RF (eds) Bioremediation: science and applications. SSSA Special Publication No 43, SSSA, Madison, Wisconsin, pp 135–145
Ascar L, Ahumada I, Richter P (2008) Influence of redox potential (Eh) on the availability of arsenic species in soils and soils amended with biosolid. Chemosphere 72:1548–1552
Basta NT, Ryan JA, Chaney RL (2005) Trace element chemistry in residual-treated soil: key concepts and metal bioavailability. J Environ Qual 34:49–63
Baziramakenga R, Simard RR, Leroux GD (1995) Determination of organic acids in soil extracts by ion chromatography. Soil Biol Biochem 27:349–356
Bloem J, Hopkins DW, Benedetti A (2006) Microbiological methods for assessing soil quality. CABI Publishing, Oxfordshire
Bolan NS, Elliott J, Gregg PEH, Weil S (1996) Rhizosphere induced dissolution of phosphate rocks in soils. Biol Fertil Soils 24:169–174
Bolan NS, Adriano DC, Natesan R, Bon-jun K (2003) Reduction and phytoavailability of Cr(VI) as influenced by organic manure compost. J Environ Qual 32:120–128
Bowen GD, Rovira AD (1992) The rhizosphere: the hidden half of the hidden half. In: Waisel Y, Eshel A, Kafkafi U (eds) Plant roots: the hidden half. Marcel Decker, New York, pp 641–669
Campos V (2009) Influence of redox environments on the solubility of arsenic in soils. Commun Soil Sci Plant Anal 40:1590–1603
Cheng L, Zhu J, Chen G, Zheng X, Oh NH, Rufty TW, Richter DD, Hu S (2010) Atmospheric CO2 enrichment facilitates cation release from soil. Ecol Lett 13:284–291
Choppala G, Bolan NS, Megharaj M, Chen Z, Naidu R (2011) The influence of biochar and black carbon on reduction and bioavailability of chromate in soils. J Environ Qual 41. doi:10.2134/jeq2011.0145
Cofield N, Banks MK, Schwab AP (2008) Lability of polycyclic aromatic hydrocarbons in the rhizosphere. Chemosphere 70:1644–1652
Fendorf SE, Zasoski RJ (1992) Chromium (III) oxidation by δ-MnO2. 1. Characterization. Environ Sci Technol 26:79–85
Fitz WJ, Wenzel WW, Zhang H, Nurmi J, Stipek K, Fischerova Z, Schweiger P, Kollemsperger G, Ma L, Stingeder G (2003) Rhizosphere characteristics of the arsenic hyperaccumulator Pteris vittata L. and monitoring of phytoremoval efficiency. Environ Sci Technol 37:5008–5014
Flessa H, Beese F (1995) Effects of sugar-beet residues on soil redox potential and nitrous-oxide emission. Soil Sci Soc Am J 59:1044–1051
Gadd GM (2010) Metals, minerals and microbes: geomicrobiology and bioremediation. Microbiology 156:609–643
Gaskin SE, Bentham RH (2010) Rhizoremediation of hydrocarbon contaminated soil using Australian native grasses. Sci Total Environ 408:3683–3688
Georgiadis M, Cai Y, Solo-Gabriele HM (2006) Extraction of arsenate and arsenite species from soils and sediments. Environ Pollut 141:22–29
Gutierrez AM, Cabriales JJP, Vega MM (2010) Isolation and characterization of hexavalent chromium-reducing rhizospheric bacteria from a wetland. Int J Phytorem 12:317–334
Goetz H, Erika K (2007) Microbes and metals: Interactions in the environment. J Basic Microbiol 47:453–467
Gonzaga MIS, Santos JAG, Ma LQ (2006) Arsenic chemistry in the rhizosphere of Pteris vittata L. and Nephrolepis exaltata L. Environ Pollut 143:254–260
Harris RF, Arnold SM (1995) Redox and energy aspects of soil bioremediation. In: Bioremediation: science and applications. Soil Science Society of America, Madison, WI, pp. 55–85
Hinsinger P (1998) How do plant roots acquire mineral nutrients? Chemical processes involved in the rhizosphere. Adv Agron 24:225–265
Hinsinger P, Bengough G, Vetterlein D, Young IM (2009) Rhizosphere: biophysics, biochemistry and ecological relevance. Plant Soil 321:117–152
James BR, Petura JC, Vitale RJ, Mussoline GR (1995) Hexavalent chromium extraction from soils: a comparison of five methods. Environ Sci Technol 29:2377–2380
Kantar C, Cetin Z, Demiray H (2008) In situ stabilization of chromium (VI) in polluted soils using organic ligands: the role of galacturonic, glucuronic and alginic acids. J Hazard Mater 159:287–293
Kuppardt A, Vetterlein D, Harms H, Chatzinotas A (2010) Visualisation of gradients in arsenic concentration around individual roots of Zea mays L. using agar-immobilized bioreporter bacteria. Plant Soil 329:295–306
Ma LQ, Komar KM, Tu C, Zhang W, Cai Y, Kennelley ED (2001) A fern that hyperaccumulates arsenic. Nature 409:579
Mahimairaja S, Bolan NS, Adriano DC, Robinson B (2005) Arsenic contamination and its risk management in complex environmental settings. Adv Agron 86:1–82
Marschner H, Römheld V, Horst WJ, Martin P (1986) Root induced changes in the rhizosphere: importance for the mineral nutrition of plants. Z Pflanzernähr Bodenk 149:441–456
Masscheleyn PH, Delaune RD, Partick WH Jr (1991) Effect of redox potential and pH on arsenic speciation and solubility in a contaminated soil. Environ Sci Technol 25:1414–1429
Megharaj M, Avudainayagam S, Naidu R (2003) Toxicity of hexavalent chromium and its reduction by bacteria isolated from soil contaminated with tannery waste. Curr Microbiol 47:51–54
Nickson RT, McArthur JM, Ravenscroft P, Burgess WG, Ahmed KM (2000) Mechanisms of arsenic release to groundwater, Bangladesh and West Bengal. Appl Geochem 15:403–413
Park JH, Lamb D, Panneerselvam P, Choppala G, Bolan NS (2011) Role of organic amendments on enhanced bioremediation of heavy metal(loid) contaminated soils. J Hazard Mater 185:549–574
Rovira AD, Davey CB (1974) Biology of the rhizosphere. In: Carsen EW (ed) The plant root and its environment. Univ Virginia Press, Charlottesville, pp 153–204
Shi SJ, Richardson AE, O’Callaghan M, DeAngelis KM, Jones EE, Stewart A, Firestone MK, Condron LM (2011) Effects of selected root exudate components on soil bacterial communities. FEMS Microbiol Ecol 77:600–610
Smith E, Smith J, Naidu R (2006) Distribution and nature of arsenic along former railway corridors of South Australia. Sci Total Environ 363:175–182
Sun HW, Xu J, Yang SH, Liu GL, Dai SG (2004) Plant uptake of aldicarb from contaminated soil and its enhanced degradation in the rhizosphere. Chemosphere 54:569–574
Stanford G, Dement JD (1957) A method for measuring short term nutrient absorption by plants: I. Phosphorus. Soil Sci Soc Am J 21:612–617
Tang C, Rengel Z (2003) Role of plant cation/anion uptake ratio in soil acidification. In: Rengel Z (ed) Handbook of soil acidity. Marcel Dekker, New York, pp 57–81
Trolove SN, Hedley MJ, Kirk GJD, Bolan NS, Loganathan P (2003) Progress in selected areas of rhizosphere research on P acquisition. Aust J Soil Res 41:471–499
Tu S, Ma LQ, Luongo T (2004) Root exudates and arsenic accumulation in arsenic hyperaccumulating Pteris vittata and non-hyperaccumulating Nephrolepis exaltata. Plant Soil 258:9–19
US EPA (1995) Chromium, Hexavalent (colorimetric). Test methods for evaluating solid waste, physical/chemical methods: SW-846. U.S. Environmental Protection Agency, U.S. Government Printing Office: Washington, DC
US EPA (1997) Method 3051a: Microwave assisted acid dissolution of sediments, sludges, soils, and oils. 2nd Edn, U.S. Environmental Protection Agency, U.S. Government Printing Office: Washington, DC
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Xu XY, McGrath SP, Zhao FJ (2007) Rapid reduction of arsenate in the medium mediated by plant roots. New Phytol 176:590–599
Zazo JA, Paull JS, Jaffe PR (2008) Influence of plants on the reduction of hexavalent chromium in wetland sediments. Environ Pollut 156:29–35
Zhang SZ, Li W, Shan XQ, Lu AX, Zhou PJ (2005) Effects of low molecular weight organic anions on the release of arsenite and arsenate from a contaminated soil. Water Air Soil Pollut 167:111–122
Zhong L, Yang J (2012) Reduction of Cr(VI) by malic acid in aqueous Fe-rich soil suspension. Chemosphere 86:973–978
Acknowledgments
The 2012 Postdoctoral fellowship program (PJ007448) at National Academy of Agricultural Science, Rural Development Administration, Republic of Korea, supported Dr Kunhikrishnan’s contribution.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: H. Schat.
Rights and permissions
About this article
Cite this article
Bolan, N., Kunhikrishnan, A. & Gibbs, J. Rhizoreduction of arsenate and chromate in Australian native grass, shrub and tree vegetation. Plant Soil 367, 615–625 (2013). https://doi.org/10.1007/s11104-012-1506-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1506-y