Abstract
Purpose
Bioceramic(Hydroxyapatite) based Poly(D,L-lactide-co-glycolide) (PLGA) and polyethylene glycol (PEG) nanoparticles of Risedronate was prepared by dialysis method for bone-targeting.
Methods
Risedronate, a targeting moiety that has a strong affinity for bone, was conjugated to PLGA via carbodiimide chemistry. Mono-methoxy PEG(mPEG)-PLGA block polymers were synthesized and used to impart surface hydrophilicity to nanoparticles to avoid its uptake by reticuloendothelial system (RES). The structure of prepared di block copolymers were characterized by FT-IR and NMR spectrometry. Risedronate was adsorbed on the surface of hydroxyapatite (RIS-HA) and it was conjugated with different ratios of mPEG-PLGA. The formation of surface-modified PLGA nanoparticle prepared with various ratios of risedronate as well as hydroxyapatite and mPEG was confirmed by 1H NMR and FT-IR spectrometry.
Results
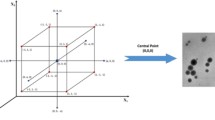
Size and % entrapment of the prepared nanoparticle was found to be 79.3 ± 2.3 nm and 93 ± 3.1%. Transmission electron microscopy (TEM) revealed that mPEG-PLGA-RIS-HA nanoparticles possess smooth and uniform surface. Pharmacodynamic study was performed on Dexamethasone (DEX) induced osteoporotic model. The effect of various formulations (mPEG-PLGA-RIS, mPEG-PLGA-RIS-HA and RISOFOS tablet) on bone was studied by Volume bone density (VBD) and by histopathological evaluation. Interestingly mPEG-PLGA-RIS-HA, showed a significant enhancement in bone micro-architecture when compared with other formulations.
Conclusions
The results strongly implicated that mPEG-PLGA-RIS-HA has a therapeutic benefits over risedronate sodium monotherapy for the treatment of osteoporosis in a rat model.







Similar content being viewed by others
Abbreviations
- CaP:
-
Calcium phosphate
- DCC:
-
Dicyclohexyl carbodiimide
- DEX:
-
Dexamethasone
- DLS:
-
Dynamic light scattering
- DMSO:
-
Dimethyl sulfoxide
- HA:
-
Hydroxyapatite
- mPEG:
-
Methoxy polyethylene glycol
- NHS:
-
N-hydroxyl succinimide
- PLGA:
-
Poly(D,L-lactide-co-glycolide)
- RIS:
-
Risedronate
- VBD:
-
Volumetric bone density analysis
References
Kristina A. Bull World Health Organ. 2003;81:657–63.
Cooper C, Melton LJ. Epidemiology of osteoporosis. Trends Endocrinol Metab. 1992;3:224–9.
Nordin BEC. International patterns of osteoporosis. Clin Orthop. 1966;45:17–30.
Currey JD. Biomechanics of mineralized skeletons. In: Carter JG, editor. Skeletal biomineralization: patterns, processes and evolutionary trends, I. New York: Van Nostrand Reinhold; 1990.
Lin L, Chow KL, Leng Y. Study of hydroxyapatite osteo inductivity with an osteogenic 257 differentiation of mesenchymal stem cells. J Biomed Mater Res A. 2009;89:326–35.
Negi P, Negi LM, Vohora D, Ahmad FJ, Talegaonkar S. Enhancing safety and efficacy of bisphosphonate therapy by association with hydroxyapatite as adjuvant drug carriers. OA Med Hypothesis. 2014;2:6.
Bonnick S, Saag KG, Kiel DE. Comparison of weekly treatment of postmenopausal osteoporosis with alendronate vs. risedronate over two years. J Clin Endocrinol Metab. 2006;91:2631–7.
Kathryn L, Kavanagh KG, James ED, Xiaoqiu W, Stefan K, Ebetin FH, et al. The molecular mechanism of nitrogen-containing bisphosphonates as anti-osteoporosis drugs. Proc Natl Acad Sci U S A. 2006;103:7829–34.
Vaculikova E, Placha D, Pisarcik M, Peikertova P, Dedkova K, Devinsky F, et al. Preparation of Risedronate nanoparticles by solvent evaporation technique. Molecules. 2014;19:17848–61.
Nasr M, Awad GA, Mansour S, Taha I, Al Shamy A, Mortada ND. Different modalities of NaCl osmogen in biodegradable microspheres for bone deposition of risedronate sodium by alveolar targeting. Eur J Pharm Biopharm. 2011;79:601–11.
Cenni E, Granchi D, Avnet S, Fotia C, Salerno M, Micieli D, et al. Biocompatibility of poly(D, L-lactide-co-glycolide) nanoparticles conjugated with alendronate. Biomaterials. 2008;29:1400–11.
Bigi A, Boanini E, Capuccini C, Fini M, Mihailescu IN, Ristoscu C, et al. Biofunctional alendronate–Hydroxyapatite thin films deposited by matrix assisted pulsed laser evaporation. Biomaterials. 2009;30:6168–77.
Hruby M, Konak C, Ulbrich K. Polymeric micellar pH sensitive drug delivery system for doxorubicin. J Control Release. 2005;103:137–48.
Shi X, Wang Y, Varshney RR, Ren L, Zhang F, Wang DA. In-vitro osteogenesis of synovium stem cells induced by controlled release of bisphosphonate additives from microspherical mesoporous silica composite. Biomaterials. 2009;30:3996–4005.
Niemi R, Vepsalainen J, Taipale H, Jairvinen T. Bisphosphonate prodrugs: synthesis and in vitro evaluation of novel acyloxyalkyl esters of clodronic acid. J Med Chem. 1999;2:5053–8.
Ezra A, Hoffman A, Breuer E, Alferiev IS, Monkkonen J, El Hanany-Rozen N, et al. A peptide prodrug approach for improving bisphosphonate oral absorption. J Med Chem. 2000;43:3641–52.
Ogawa K, Mukai T, Inoue Y, Ono M, Saji H. Development of a novel 99mTc-chelate conjugated bisphosphonate with high affinity for bone as a bone scintigraphic agent. J Nucl Med. 2006;47:2042–7.
Choi SW, Kim JH. Design of surface-modified poly(D, L-lactide-co-glycolide) nanoparticles for targeted drug delivery to bone. J Control Release. 2007;122:24–30.
Sahana H, Khajuria DK, Razdan R, Mahapatra DR, Bhat MR, Suresh S, et al. Improvement in bone properties by using risedronate adsorbed hydroxyapatite novel nanoparticle based formulation in a rat model of osteoporosis. J Biomed Nanotechnol. 2013;9:193–201.
Fu YC, Fu TF, Wang HJ, Lin CW, Lee GH, Wu SC, et al. Aspartic acid-based modified PLGA-PEG nanoparticles for bone targeting: in vitro and in vivo evaluation. Acta Biomater. 2014;10:4583–96.
Wu CC, Wang CC, Lu DH, Hsu LH, Yang KC, Lin FH. Calcium phosphate cement delivering zoledronate decreases bone turnover rate and restores bone architecture in ovariectomized rats. Biomed Mater. 2012;7:035009.
Kyllonen L, Este MD, Alini M, Eglin D. Local drug delivery for enhancing fracture healing in osteoporotic bone. Acta Biomater. 2015;11:412–34.
Bozzo Rde O, Rocha RG, Haiter Neto F, Paganini GA, Cavalcanti MG. Linear density analysis of bone repair in rats using digital direct radiograph. J Appl Oral Sci. 2004;12:317–21.
Bancroft JD, Cook HC. Manual of histological techniques and their diagnostic application. Edinburgh: Churchill Livingstone; 1994.
Saikia KC, Bhattacharya TD, Bhuyan SK, Talukdar DJ, Saikia SP, Jitesh P. Calcium phosphate ceramics as bone graft substitutes in filling bone tumor defects. Indian J Orthop. 2008;42:169–72.
Murakami H, Takahashi N, Sasaki T, Udagawa N, Tanaka S, Nakamura I, et al. A possible mechanism of the specific action of bisphosphonates on osteoclasts: tiludronate preferentially affects polarized osteoclasts having ruffled borders. Bone. 1995;17:137–44.
Khanna S, Pillai KK, Vohora D. Bisphosphonates in phenytoin-induced bone disorder. Bone. 2011;48:597–606.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors are thankful for the financial assistance from ICMR, New Delhi. We also acknowledge the Evonik Research for providing the gift sample of PLGA (50:50) polymer and lipoid for providing the gift sample of methoxy PEG. The authors are also thankful to Mr. Jitender singh, Department of Pharmaceutical chemistry Jamia Hamdard and Dr. Ambrish Tiwari, Incharge of animal house, Faculty of Science, Jamia Hamdard, New Delhi for their inputs regarding animal studies.
Conflicts of Interest
The authors state no conflicts of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rawat, P., Manglani, K., Gupta, S. et al. Design and Development of Bioceramic Based Functionalized PLGA Nanoparticles of Risedronate for Bone Targeting: In-vitro Characterization and Pharmacodynamic Evaluation. Pharm Res 32, 3149–3158 (2015). https://doi.org/10.1007/s11095-015-1692-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-015-1692-4