ABSTRACT
Purpose
The fate of inhaled salmon calcitonin (sCT) at the respiratory epithelial barrier was studied with particular emphasis on enzymatic degradation by trypsin, chymotrypsin, and neutrophil elastase.
Methods
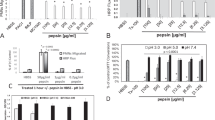
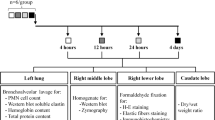
Degradation of sCT was assessed by HPLC in cell homogenate, supernatant and intact monolayers of human respiratory epithelial cells (hBEpC, Calu-3, 16HBE14o-, A549) and Caco-2 as comparison at 37°C for 2 h. Breakdown of sCT by trypsin, chymotrypsin and neutrophil elastase was investigated. The presence of enzymes in cell supernatant and homogenate was studied by immunoblot and enzyme activity by model substrate assay. Transport studies across Calu-3 monolayers were performed.
Results
sCT concentration remained unchanged over 2 h, when incubated in supernatant or with cell monolayers, independent of cell type studied. When cell homogenates were used, sCT concentrations were reduced to varying extents. sCT was degraded when incubated with enzymes alone. Western blot revealed abundance of all proteinases in cell homogenates and weaker expression in supernatants. Transport studies indicated net-absorptive sCT translocation; presence of bacitracin resulted in increased amount of sCT in receiver compartments.
Conclusions
Epithelial proteases play a role in the disposition of sCT after pulmonary delivery.









Similar content being viewed by others
REFERENCES
Stevenson CL. Advances in peptide pharmaceuticals. Curr Pharm Biotechnol. 2009;10(1):122–37.
Patton JS, Brain JD, Davies LA, Fiegel J, Gumbleton M, Kim KJ, et al. The particle has landed–characterizing the fate of inhaled pharmaceuticals. J Aerosol Med Pulm Drug Deliv. 2010;23 Suppl 2:S71–87.
Patton JS, Platz RM. (D) Routes of delivery: Case studies: (2) Pulmonary delivery of peptides and proteins for systemic action. Adv Drug Deliv Rev. 1992;8(2–3):179–96.
Woodley J. Enzymatic barriers for GI peptide and protein delivery. Crit Rev Ther Drug Carrier Syst. 1994;11(2–3):61–95.
Baginski L, Tachon G, Falson G, Patton J, Bakowsky U, Ehrhardt C. Reverse transcription polymerase chain reaction (RT-PCR) analysis of proteolytic enzymes in cultures of human respiratory epithelial cells. J Aerosol Med Pulm Drug Deliv. 2011;24(2):89–101.
Patton JS. Mechanisms of macromolecule absorption by the lungs. Adv Drug Deliv Rev. 1996;19(1):3–36.
Dershwitz M, Walsh J, Morishige R, Connors P, Rubsamen R, Shafer S, et al. Pharmacokinetics and pharmacodynamics of inhaled versus intravenous morphine in healthy volunteers. Anesthesiology. 2000;93(3):619–28.
Patton J, Byron P. Inhaling medicines: delivering drugs to the body through the lungs. Nat Rev Drug Discov. 2007;6(1):67–74.
Bernkop-Schnürch A, Kast C, Guggi D. Permeation enhancing polymers in oral delivery of hydrophilic macromolecules: thiomer/GSH systems. J Control Release. 2003;93(2):95–103.
Lang S, Staudenmann W, James P, Manz H, Kessler R, Galli B, et al. Proteolysis of human calcitonin in excised bovine nasal mucosa: elucidation of the metabolic pathway by liquid secondary ionization mass spectrometry (LSIMS) and matrix assisted laser desorption ionization mass spectrometry (MALDI). Pharm Res. 1996;13(11):1679–85.
Dohi M, Nishibe Y, Makino Y, Suzuki Y, editors. Enzymatic barrier to nasal delivery of salmon calcitonin in rabbits. Proceedings of the International Symposium Control on Relative Society. Kyoto, Japan: 1993.
Guggi D, Bernkop-Schnürch A. In vitro evaluation of polymeric excipients protecting calcitonin against degradation by intestinal serine proteases. Int J Pharm. 2003;252(1–2):187–96.
Reid P, Marsden M, Cunningham G, Haslett C, Sallenave J. Human neutrophil elastase regulates the expression and secretion of elafin (elastase-specific inhibitor) in type II alveolar epithelial cells. FEBS Lett. 1999;457(1):33–7.
Fogh J, Trempe G. New human tumor cell lines. In: Fogh J, editor. Human tumor cells in vitro. New York: Plenum; 1975. p. 115–59.
Giard D, Aaronson S, Todaro G, Arnstein P, Kersey J, Dosik H, et al. In vitro cultivation of human tumors: establishment of cell lines derived from a series of solid tumors. J Natl Canc Inst. 1973;51(5):1417–23.
Sporty J, Horálková L, Ehrhardt C. In vitro cell culture models for the assessment of pulmonary drug disposition. Expert Opin Drug Metab Toxicol. 2008;4(4):333–45.
Schwert G, Takenaka Y. A spectrophotometric determination of trypsin and chymotrypsin. Biochim Biophys Acta. 1955;16(4):570–5.
Cotter T, Robinson G. Purification and characterisation of an ‘elastase-like’ enzyme from rabbit polymorphonuclear leucocytes. Biochim Biophys Acta. 1980;615(2):414–25.
Visser L, Blout E. The use of p-nitrophenyl N-tert-butyloxycarbonyl-L-alaninate as substrate for elastase. Biochim Biophys Acta. 1972;268(1):257–60.
Shah R, Khan M. Protection of salmon calcitonin breakdown with serine proteases by various ovomucoid species for oral drug delivery. J Pharm Sci. 2004;93(2):392–406.
Kobayashi S, Kondo S, Juni K. Study on pulmonary delivery of salmon calcitonin in rats: effects of protease inhibitors and absorption enhancers. Pharm Res. 1994;11(9):1239–43.
Bernkop-Schnürch A. The use of inhibitory agents to overcome the enzymatic barrier to perorally administered therapeutic peptides and proteins. J Control Release. 1998;52(1–2):1–16.
Yamahara H, Lehr C, Lee V, Kim K. Fate of insulin during transit across rat alveolar epithelial cell monolayers. Eur J Pharm Biopharm. 1994;40:294–8.
Forbes B, Wilson CG, Gumbleton M. Temporal dependence of ectopeptidase expression in alveolar epithelial cell culture: implications for study of peptide absorption. Int J Pharm. 1999;180(2):225–34.
Kobayashi S, Kondo S, Juni K. Pulmonary delivery of salmon calcitonin dry powders containing absorption enhancers in rats. Pharm Res. 1996;13(1):80–3.
Kawabata K, Hagio T, Matsuoka S. The role of neutrophil elastase in acute lung injury. Eur J Pharmacol. 2002;451(1):1–10.
Sun Z, Yang P. Role of imbalance between neutrophil elastase and alpha 1-antitrypsin in cancer development and progression. Lancet Oncol. 2004;5(3):182–90.
Amelinckx A, Whitney P, Santos N, Lascano J, Salathe M, Conner G, et al. Regulation and anti-elastase activity of locally produced alpha-1-antitrypsin by well differentiated normal airway epithelial cells: a comparison with SLPI. Am J Respir Crit Care Med. 2010;181:A4158.
Wall DA, Lanutti AT. High levels of exopeptidase activity are present in rat and canine bronchoalveolar lavage fluid. Int J Pharm. 1993;97:171–81.
Lang S, Rothen-Rutishauser B, Perriard JC, Schmidt MC, Merkle HP. Permeation and pathways of human calcitonin (hCT) across excised bovine nasal mucosa. Peptides. 1998;19(3):599–607.
Gaudiano MC, Colone M, Bombelli C, Chistolini P, Valvo L, Diociaiuti M. Early stages of salmon calcitonin aggregation: effect induced by ageing and oxidation processes in water and in the presence of model membranes. Biochim Biophys Acta. 2005;1750(2):134–45.
Youn Y, Jung J, Oh S, Yoo S, Lee K. Improved intestinal delivery of salmon calcitonin by Lys18-amine specific PEGylation: stability, permeability, pharmacokinetic behavior and in vivo hypocalcemic efficacy. J Control Release. 2006;114(3):334–42.
Song K, Chung S, Shim C. Enhanced intestinal absorption of salmon calcitonin (sCT) from proliposomes containing bile salts. J Control Release. 2005;106(3):298–308.
Torres-Lugo M, García M, Record R, Peppas NA. pH-Sensitive hydrogels as gastrointestinal tract absorption enhancers: transport mechanisms of salmon calcitonin and other model molecules using the Caco-2 cell model. Biotechnol Prog. 2002;18(3):612–6.
Tréhin R, Krauss U, Beck-Sickinger A, Merkle H, Nielsen H. Cellular uptake but low permeation of human calcitonin-derived cell penetrating peptides and Tat(47–57) through well-differentiated epithelial models. Pharm Res. 2004;21(7):1248–56.
Patton JS, Trinchero P, Platz RM. Bioavailability of pulmonary delivered peptides and proteins: [alpha]-interferon, calcitonins and parathyroid hormones. J Control Release. 1994;28(1–3):79–85.
Clark A, Kuo MC, Newman S, Hirst P, Pitcairn G, Pickford M. A comparison of the pulmonary bioavailability of powder and liquid aerosol formulations of salmon calcitonin. Pharm Res. 2008;25(7):1583–90.
Lewiecki EM. Emerging drugs for postmenopausal osteoporosis. Expert Opin Emerg Drugs. 2009;14(1):129–44.
Youn YS, Kwon MJ, Na DH, Chae SY, Lee S, Lee KC. Improved intrapulmonary delivery of site-specific PEGylated salmon calcitonin: optimization by PEG size selection. J Control Release. 2008;125(1):68–75.
Lombry C, Edwards DA, Préat V, Vanbever R. Alveolar macrophages are a primary barrier to pulmonary absorption of macromolecules. Am J Physiol Lung Cell Mol Physiol. 2004;286(5):L1002–8.
ACKNOWLEDGEMENTS & DISCLOSURES
This work was funded by a Strategic Research Cluster grant (07/SRC/B1154) under the National Development Plan co-funded by EU Structural Funds and Science Foundation Ireland. S.T.B. is funded by an IRCSET Government of Ireland Postgraduate Scholarship in Science, Engineering and Technology. U.B. is funded by the DFG Forschergruppe Nanohale 627.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baginski, L., Tewes, F., Buckley, S.T. et al. Investigations into the Fate of Inhaled Salmon Calcitonin at the Respiratory Epithelial Barrier. Pharm Res 29, 332–341 (2012). https://doi.org/10.1007/s11095-011-0553-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-011-0553-z