Abstract
Purpose
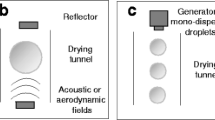
The purpose of this study was to investigate the possibility of producing identical powders in pilot and production scale spray drying equipment by matching the droplet size distributions produced by two differently sized atomizers.
Methods
Particles were prepared by spray drying solutions of acetaminophen and polyvinylpyrrolidone K-30. The success of the up-scaling was evaluated by comparing the powders in terms of particle size distribution (laser diffraction), crystallinity (XPRD) and morphology (SEM). Furthermore, the influence of process parameters on other product characteristics such as stability and residual volatile content was also evaluated.
Results
The spray drying experiments resulted in spherical, amorphous particles with volumetric median diameters of typically 4–10 μm for pilot scale and 4–30 μm for production scale. The results showed that particles with similar morphology and crystallinity could be produced in the two applied spray dryers. However, scale-up based purely on matching droplet size distributions was not feasible.
Conclusions
The scale-up criterion did not account for the differences between the droplet-drying gas mixing and residence time distribution within the two spray dryers. Therefore, production scale experiments are required in order to obtain similar product characteristics as in pilot scale.






Similar content being viewed by others
References
K. Masters. Spray Drying Handbook. Longman Scientific & Technical, Essex, England, 1991.
P. Giunchedi, and U. Conte. Spray-drying as a preparation method of microparticulate drug-delivery systems: an overview. S. T. P. Pharm. Sci. 5:276–290 (1995).
S. Wendel, and M. Celik. An overview of spray-drying applications. Pharm. Technol. 21:124–144 (1997).
K. Masters. Spray Drying in Practice. SprayDryConsult International Aps, Charlottenlund, 2002.
M. Yoshioka, B. C. Hancock, and G. Zografi. Inhibition of indomethacin crystallization in poly(vinylpyrrolidone) coprecipitates. J. Pharm. Sci. 84:983–986 (1995).
A. A. Ambike, K. R. Mahadik, and A. Paradkar. Stability study of amorphous valdecoxib. Int. J. Pharm. 282:151–162 (2004).
R. Chen, M. Tagawa, N. Hoshi, T. Ogura, H. Okamoto, and K. Danjo. Improved dissolution of an insoluble drug using a 4-fluid nozzle spray-drying technique. Chem. Pharm. Bull. 52:1066–1070 (2004).
K. Khougaz, and S. D. Clas. Crystallization inhibition in solid dispersions of MK-0591 and poly(vinylpyrrolidone) polymers. J. Pharm. Sci. 89:1325–1334 (2000).
T. Matsumoto, and G. Zografi. Physical properties of solid molecular dispersions of indomethacin with poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-vinylacetate) in relation to indomethacin crystallization. Pharm. Res. 16:1722–1728 (1999).
G. Van den Mooter, M. Wuyts, N. Blaton, R. Busson, P. Grobet, P. Augustijns, and R. Kinget. Physical stabilisation of amorphous ketoconazole in solid dispersions with polyvinylpyrrolidone K25. Eur. J. Pharm. Sci. 12:261–269 (2001).
E. Esposito, R. Roncarati, R. Cortesi, F. Cervellati, and C. Nastruzzi. Production of eudragit microparticles by spray-drying technique: influence of experimental parameters on morphological and dimensional characteristics. Pharm. Dev. Technol. 5:267–278 (2000).
Y. Maa, H. R. Costantino, P. Nguyen, and C. C. Hsu. The effect of operating and formulation variables on the morphology of spray-dried protein particles. Pharm. Dev. Technol. 2:213–223 (1997).
P. Tewa-Tagne, G. Degobert, S. Briancon, C. Bordes, J. Y. Gauvrit, P. Lanteri, and H. Fessi. Spray-drying nanocapsules in presence of colloidal silica as drying auxiliary agent: formulation and process variables optimization using experimental designs. Pharm. Res. 24:650–661 (2007).
P. Johansen, H. P. Merkle, and B. Gander. Technological considerations related to the up-scaling of protein microencapsulation by spray-drying. Eur. J. Pharm. Biopharm. 50:413–417 (2000).
R. P. Raffin, S. S. Guterres, A. R. Pohlmann, and M. I. Re. Powder characteristics of pantoprazole delivery systems produced in different spray-dryer scales. Drying Technol. 24:339–348 (2006).
T. P. Foster, and M. W. Leatherman. Powder characteristics of proteins spray-dried from different spray-Dryers. Drug Dev. Ind. Pharm. 21:1705–1723 (1995).
D. E. Walton. The morphology of spray-dried particles a qualitative view. Drying Technol. 18:1943–1986 (2000).
M. Zlokarnik. Scale-up in Chemical Engineering. Wiley-VCH, Weinheim, 2002.
Thybo, P., Andersen, S. K., Lindeløv, J. S., and Hovgaard, L. Droplet size measurements for spray dryer scale-up. Pharm. Dev. Technol. In Press.
A. Billon, B. Bataille, G. Cassanas, and M. Jacob. Development of spray-dried acetaminophen microparticles using experimental designs. Int. J. Pharm. 203:159–168 (2000).
M. M. de Villiers, D. E. Wurster, J. G. Van der Watt, and A. Ketkar. X-ray powder diffraction determination of the relative amount of crystalline acetaminophen in solid dispersions with polyvinylpyrrolidone. Int. J. Pharm. 163:219–224 (1998).
T. Miyazaki, S. Yoshioka, Y. Aso, and S. Kojima. Ability of polyvinylpyrrolidone and polyacrylic acid to inhibit the crystallization of amorphous acetaminophen. J. Pharm. Sci. 93:2710–2717 (2004).
M. Angberg, C. Nystrom, and S. Castensson. Evaluation of heat-conduction microcalorimetry in pharmaceutical stability studies.5. A new approach for continuous measurements in abundant water-vapor. Int. J. Pharm. 81:153–167 (1992).
M. Angberg, C. Nystrom, and S. Castensson. Evaluation of heat-conduction microcalorimetry in pharmaceutical stability studies.6. Continuous monitoring of the interaction of water-vapor with powders and powder mixtures at various relative humidities. Int. J. Pharm. 83:11–23 (1992).
K. Mosen, K. Backstrom, K. Thalberg, T. Schaefer, H. G. Kristensen, and A. Axelsson. Particle formation and capture during spray drying of inhalable particles. Pharm. Dev. Technol. 9:409–417 (2004).
M. Haisa, S. Kashino, R. Kawai, and H. Maeda. The monoclinic form of p-hydroxyacetanilide. Acta Crystallogr. B32:1283–1285 (1976).
P. Thybo, J. Kristensen, and L. Hovgaard. Characterization and physical stability of tolfenamic Acid-PVP K30 solid dispersions. Pharm. Dev. Technol. 12:43–53 (2007).
P. Di Martino, G. F. Palmieri, and S. Martelli. Molecular mobility of the paracetamol amorphous form. Chem. Pharm. Bull. 48:1105–1108 (2000).
Acknowledgments
The Drug Research Academy, GEA Pharmaceutical A/S, and Niro A/S are acknowledged for financial support. The authors would like to thank Matti Murtomaa and University of Turku for use of X-ray diffractometer and microcalorimeter. Mikko Tenho and Olli-Pekka Hämäläinen are greatly appreciated for their assistance in XRPD and isothermal microcalorimetry measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thybo, P., Hovgaard, L., Lindeløv, J.S. et al. Scaling Up the Spray Drying Process from Pilot to Production Scale Using an Atomized Droplet Size Criterion. Pharm Res 25, 1610–1620 (2008). https://doi.org/10.1007/s11095-008-9565-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-008-9565-8