Abstract
Methylglyoxal (MG) is a reactive dicarbonyl presenting both endogenous (e.g. glycolysis) and exogenous (e.g. food cooking) sources. MG induces neurotoxicity, at least in part, by affecting mitochondrial function, including a decline in the oxidative phosphorylation (OXPHOS) system activity, bioenergetics failure, and redox disturbances. Sulforaphane (SFN) is an isothiocyanate found mainly in cruciferous vegetables and exerts antioxidant and anti-inflammatory effects in mammalian cells. SFN also decreases mitochondrial vulnerability to several chemical stressors. SFN is a potent activator of the transcription factor nuclear factor erythroid 2-related factor 2 (Nrf2), which is a master regulator of the mammalian redox biology. Here, we have investigated whether and how SFN would be able to prevent the MG-induced mitochondrial collapse in the human neuroblastoma SH-SY5Y cells. The cells were exposed to SFN at 5 µM for 24 h prior to the administration of MG at 500 µM for additional 24 h. We found that SFN prevented the MG-induced OXPHOS dysfunction and mitochondrial redox impairment. SFN stimulated the activity of the enzyme γ-glutamylcysteine ligase (γ-GCL), leading to increased synthesis of glutathione (GSH). Inhibition of γ-GCL with buthionine sulfoximine (BSO) or silencing of Nrf2 using small interfering RNA (siRNA) against this transcription factor reduced the levels of GSH and abolished the mitochondrial protection promoted by SFN in the MG-treated cells. Thus, SFN protected mitochondria of the MG-challenged cells by a mechanism involving the Nrf2/γ-GCL/GSH axis.












Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article (and its supplementary information files).
References
Kalapos MP (1999) Methylglyoxal in living organisms: chemistry, biochemistry, toxicology and biological implications. Toxicol Lett 110:145–175. doi:https://doi.org/10.1016/s0378-4274(99)00160-5
Chakraborty S, Karmakar K, Chakravortty D (2014) Cells producing their own nemesis: understanding methylglyoxal metabolism. IUBMB Life 66:667–678. doi:https://doi.org/10.1002/iub.1324
Allaman I, Bélanger M, Magistretti PJ (2015) Methylglyoxal, the dark side of glycolysis. Front Neurosci 9:23. doi:https://doi.org/10.3389/fnins.2015.00023
Beisswenger PJ (2014) Methylglyoxal in diabetes: link to treatment, glycaemic control and biomarkers of complications. Biochem Soc Trans 42:450–456. doi:https://doi.org/10.1042/BST20130275
Angeloni C, Zambonin L, Hrelia S (2014) Role of methylglyoxal in Alzheimer’s disease. Biomed Res Int 2014:238485. doi:https://doi.org/10.1155/2014/238485
Hipkiss AR (2017) On the relationship between energy metabolism, proteostasis, aging and Parkinson’s disease: possible causative role of methylglyoxal and alleviative potential of carnosine. Aging Dis 8:334–345. https://doi.org/10.14336/AD.2016.1030
Kuntz S, Rudloff S, Ehl J, Bretzel RG, Kunz C (2009) Food derived carbonyl compounds affect basal and stimulated secretion of interleukin-6 and -8 in Caco-2 cells. Eur J Nutr 48:499–503. https://doi.org/10.1007/s00394-009-0035-9
Pfeifer YV, Haase PT, Kroh LW (2013) Reactivity of thermally treated α-dicarbonyl compounds. J Agric Food Chem 61:3090–3096. doi:https://doi.org/10.1021/jf302959k
Camel V, Bermond A (1998) The use of ozone and associated oxidation processes in drinking water treatment. Water Res 32:3208–3222
Fujioka K, Shibamoto T (2006) Determination of toxic carbonyl compounds in cigarette smoke. Environ Toxicol 21:47–54. doi:https://doi.org/10.1002/tox.20153
Fu T-M, Jacob DJ, Wittrock F, Burrows JP, Vrekoussis M, Henze DK (2008) Global budgets of atmospheric glyoxal and methylglyoxal, and implications for formation of secondary organic aerosols. J Geophys Res 113:D15303
Yamaguchi T, Nakagawa K (1983) Mutagenicity of and formation of oxygen radicals by trioses and glyoxal derivatives. Agric Biol Chem 47:2461–2465
Thornalley PJ, Wolff SP, Crabbe MJ, Stern A (1984) The oxidation of oxyhaemoglobin by glyceraldehyde and other simple monosaccharides. Biochem J 217:615–622. doi:https://doi.org/10.1042/bj2170615
Angeloni C, Malaguti M, Rizzo B, Barbalace MC, Fabbri D, Hrelia S (2015) Neuroprotective effect of sulforaphane against methylglyoxal cytotoxicity. Chem Res Toxicol 28:1234–1245. doi:https://doi.org/10.1021/acs.chemrestox.5b00067
Fürstenau CR, de Souza ICC, de Oliveira MR (2019) Tanshinone I induces mitochondrial protection by a mechanism involving the Nrf2/GSH axis in the human neuroblastoma SH-SY5Y cells exposed to methylglyoxal. Neurotoxicol Res 36:491–502. https://doi.org/10.1007/s12640-019-00091-1
Pun PB, Murphy MP (2012) Pathological significance of mitochondrial glycation. Int J Cell Biol 2012:843505. doi:https://doi.org/10.1155/2012/843505
Bo J, Xie S, Guo Y, Zhang C, Guan Y, Li C, Lu J, Meng QH (2016) Methylglyoxal impairs insulin secretion of pancreatic β-cells through increased production of ros and mitochondrial dysfunction mediated by upregulation of UCP2 and MAPKs. J Diabetes Res 2016:2029854. https://doi.org/10.1155/2016/2029854
Cobley JN, Fiorello ML, Bailey DM (2018) 13 reasons why the brain is susceptible to oxidative stress. Redox Biol 15:490–503. doi:https://doi.org/10.1016/j.redox.2018.01.008
Smith MA, Richey PL, Taneda S, Kutty RK, Sayre LM, Monnier VM, Perry G (1994) Advanced Maillard reaction end products, free radicals, and protein oxidation in Alzheimer’s disease. Ann N Y Acad Sci 738:447–454. doi:https://doi.org/10.1111/j.1749-6632.1994.tb21836.x
Smith MA, Taneda S, Richey PL, Miyata S, Yan SD, Stern D, Sayre LM, Monnier VM, Perry G (1994) Advanced Maillard reaction end products are associated with Alzheimer disease pathology. Proc Natl Acad Sci U S A 91:5710–5714. doi:https://doi.org/10.1073/pnas.91.12.5710
Dhar A, Dhar I, Desai KM, Wu L (2010) Methylglyoxal scavengers attenuate endothelial dysfunction induced by methylglyoxal and high concentrations of glucose. Br J Pharmacol 161:1843–1856. doi:https://doi.org/10.1111/j.1476-5381.2010.01017.x
Tate M, Higgins GC, De Blasio MJ, Lindblom R, Prakoso D, Deo M, Kiriazis H, Park M, Baeza-Garza CD, Caldwell ST, Hartley RC, Krieg T, Murphy MP, Coughlan MT, Ritchie RH (2019) The mitochondria-targeted methylglyoxal sequestering compound, mitogamide, is cardioprotective in the diabetic heart. Cardiovasc Drugs Ther 33:669–674. https://doi.org/10.1007/s10557-019-06914-9
Thornalley PJ (1990) The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochem J 269:1–11. doi:https://doi.org/10.1042/bj2690001
Thornalley PJ (2003) Glyoxalase I—structure, function and a critical role in the enzymatic defence against glycation. Biochem Soc Trans 31:1343–1348. https://doi.org/10.1042/bst0311343
Lu SC (2013) Glutathione synthesis. Biochim Biophys Acta 1830:3143–3153. doi:https://doi.org/10.1016/j.bbagen.2012.09.008
Lu SC (2009) Regulation of glutathione synthesis. Mol Aspects Med 30:42–59. doi:https://doi.org/10.1016/j.mam.2008.05.005
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295. doi:https://doi.org/10.1074/jbc.R900010200
Wakabayashi N, Slocum SL, Skoko JJ, Shin S, Kensler TW (2010) When NRF2 talks, who’s listening? Antioxid Redox Signal 13:1649–1663. doi:https://doi.org/10.1089/ars.2010.3216
Houghton CA (2019) Sulforaphane: its “coming of age” as a clinically relevant nutraceutical in the prevention and treatment of chronic disease. Oxid Med Cell Longev 2019:2716870. https://doi.org/10.1155/2019/2716870
Huang C, Wu J, Chen D, Jin J, Wu Y, Chen Z (2019) Effects of sulforaphane in the central nervous system. Eur J Pharmacol 853:153–168. doi:https://doi.org/10.1016/j.ejphar.2019.03.010
de Oliveira MR, Brasil FB, Fürstenau CR (2018) Sulforaphane attenuated the pro-inflammatory state induced by hydrogen peroxide in SH-SY5Y cells through the Nrf2/HO-1 signaling pathway. Neurotoxicol Res 34:241–249. https://doi.org/10.1007/s12640-018-9881-7
de Oliveira MR, Ferreira GC, Schuck PF, Dal Bosco SM (2015) Role for the PI3K/Akt/Nrf2 signaling pathway in the protective effects of carnosic acid against methylglyoxal-induced neurotoxicity in SH-SY5Y neuroblastoma cells. Chem Biol Interact 242:396–406. doi:https://doi.org/10.1016/j.cbi.2015.11.003
de Arriba SG, Krügel U, Regenthal R, Vissiennon Z, Verdaguer E, Lewerenz A, García-Jordá E, Pallas M, Camins A, Münch G, Nieber K, Allgaier C (2006) Carbonyl stress and NMDA receptor activation contribute to methylglyoxal neurotoxicity. Free Radic Biol Med 40:779–790. doi:https://doi.org/10.1016/j.freeradbiomed.2005.09.038
de Arriba SG, Stuchbury G, Yarin J, Burnell J, Loske C, Münch G (2007) Methylglyoxal impairs glucose metabolism and leads to energy depletion in neuronal cells–protection by carbonyl scavengers. Neurobiol Aging 28:1044–1050. doi:https://doi.org/10.1016/j.neurobiolaging.2006.05.007
de Oliveira MR, de Souza ICC, Fürstenau CR (2020) Mitochondrial protection promoted by the coffee diterpene kahweol in methylglyoxal-treated human neuroblastoma SH-SY5Y cells. Neurotoxicol Res 37:100–110. https://doi.org/10.1007/s12640-019-00107-w
Jiang X, Wang X (2004) Cytochrome C-mediated apoptosis. Annu Rev Biochem 73:87–106. doi:https://doi.org/10.1146/annurev.biochem.73.011303.073706
Borutaite V, Morkuniene R, Brown GC (1999) Release of cytochrome c from heart mitochondria is induced by high Ca2+ and peroxynitrite and is responsible for Ca(2+)-induced inhibition of substrate oxidation. Biochim Biophys Acta 1453:41–48. https://doi.org/10.1016/s0925-4439(98)00082-9
Rieske JS (1967) The quantitative determination of mitochondrial hemoproteins. Methods Enzymol 10:488–493
Wang K, Zhu L, Zhu X, Zhang K, Huang B, Zhang J, Zhang Y, Zhu L, Zhou B, Zhou F (2014) Protective effect of paeoniflorin on Aβ25-35-induced SH-SY5Y cell injury by preventing mitochondrial dysfunction. Cell Mol Neurobiol 34:227–234. doi:https://doi.org/10.1007/s10571-013-0006-9
Poderoso JJ, Carreras MC, Lisdero C, Riobó N, Schöpfer F, Boveris A (1996) Nitric oxide inhibits electron transfer and increases superoxide radical production in rat heart mitochondria and submitochondrial particles. Arch Biochem Biophys 328:85–92. doi:https://doi.org/10.1006/abbi.1996.0146
White CC, Viernes H, Krejsa CM, Botta D, Kavanagh TJ (2003) Fluorescence-based microtiter plate assay for glutamate-cysteine ligase activity. Anal Biochem 318:175–180. doi:https://doi.org/10.1016/s0003-2697(03)00143-x
Tarozzi A, Morroni F, Merlicco A, Hrelia S, Angeloni C, Cantelli-Forti G, Hrelia P (2009) Sulforaphane as an inducer of glutathione prevents oxidative stress-induced cell death in a dopaminergic-like neuroblastoma cell line. J Neurochem 111:1161–1171. doi:https://doi.org/10.1111/j.1471-4159.2009.06394.x
Dias IH, Chapple IL, Milward M, Grant MM, Hill E, Brown J, Griffiths HR (2013) Sulforaphane restores cellular glutathione levels and reduces chronic periodontitis neutrophil hyperactivity in vitro. PLoS ONE 8:e66407. https://doi.org/10.1371/journal.pone.0066407
Steele ML, Fuller S, Patel M, Kersaitis C, Ooi L, Münch G (2013) Effect of Nrf2 activators on release of glutathione, cysteinylglycine and homocysteine by human U373 astroglial cells. Redox Biol 1:441–445. doi:https://doi.org/10.1016/j.redox.2013.08.006
Sedlak TW, Nucifora LG, Koga M, Shaffer LS, Higgs C, Tanaka T, Wang AM, Coughlin JM, Barker PB, Fahey JW, Sawa A (2018) Sulforaphane augments glutathione and influences brain metabolites in human subjects: a clinical pilot study. Mol Neuropsychiatry 3:214–222. https://doi.org/10.1159/000487639
Wang X, Yu S, Wang CY, Wang Y, Liu HX, Cui Y, Zhang LD (2015) Advanced glycation end products induce oxidative stress and mitochondrial dysfunction in SH-SY5Y cells. In Vitro Cell Dev Biol Anim 51:204–209. doi:https://doi.org/10.1007/s11626-014-9823-5
Patel SH, Yue F, Saw SK, Foguth R, Cannon JR, Shannahan JH, Kuang S, Sabbaghi A, Carroll CC (2019) Advanced glycation end-products suppress mitochondrial function and proliferative capacity of achilles tendon-derived fibroblasts. Sci Rep 9:12614. https://doi.org/10.1038/s41598-019-49062-8
Angeloni C, Malaguti M, Hrelia S (2015) Antiglycative activity of sulforaphane: a new avenue to counteract neurodegeneration? Neural Regen Res 10:1750–1751. doi:https://doi.org/10.4103/1673-5374.169613
Sies H, Berndt C, Jones DP (2017) Oxidative stress. Annu Rev Biochem 86:715–748. https://doi.org/10.1146/annurev-biochem-061516-045037
Acknowledgements
MRO receives a “Bolsa de Produtividade em Pesquisa 2 - PQ2” fellow from the Conselho Nacional de Pesquisa e Desenvolvimento Tecnológico (CNPq) (protocol number 301273/2018-9). This work was partially supported by CNPq (protocol numbers 400216/2016-7). RCBG receives a fellow from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11064_2020_3204_MOESM1_ESM.pdf
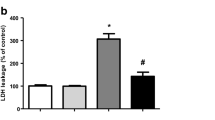
Figure S1 Mitochondrial isolation quality control. The activity of the cytoplasmic enzyme lactate dehydrogenase (LDH) was evaluated in the mitochondrial fraction obtained from the SH-SY5Y cells to check the purity of the samples.
11064_2020_3204_MOESM2_ESM.pdf
Figure S2 Sulforaphane (SFN) prevented the release of cytochrome c to the cytosol (A), the upregulation in the activity of the caspases-9 (B) and -3 (C), and the increase in the levels of cleaved PARP (D) and of DNA fragmentation (E) in methylglyoxal (MG)-treated SH-SY5Y cells. Cells were treated with SFN at 5 µM for 24 h before the administration of MG at 500 µM for additional 24 h. Data are presented as the mean ± SEM of three or five independent experiments each done in triplicate. One-way ANOVA followed by the post hoc Tukey’s test, *p < 0.01 vs the control group, #p < 0.01 vs MG-treated cells.
11064_2020_3204_MOESM3_ESM.pdf
Figure S3 Buthionine sulfoximine (BSO) suppressed the effect of a pretreatment with sulforaphane (SFN) on the levels of cytosolic cytochrome c and of malondialdehyde (MDA, a molecular marker of lipid peroxidation) in the membranes of mitochondria obtained from SH-SY5Y cells exposed to methylglyoxal (MG). Buthionine sulfoximine (BSO) at 400 µM was administrated to the cells for 24 h in the presence or absence of SFN at 5 µM. Then, the cells were exposed to MG for further 24 h. Data are presented as the mean ± SEM of three or five independent experiments each done in triplicate. One-way ANOVA followed by the post hoc Tukey’s test, *p < 0.01 vs the control group, #p < 0.01 vs MG-treated cells, º p < 0.01 vs SFN + MG-treated cells.
11064_2020_3204_MOESM4_ESM.pdf
Figure S4 The effects of silencing Nrf2 on the activity of this transcription factor. Data are presented as the mean ± SEM of three or five independent experiments each done in triplicate. One-way ANOVA followed by the post hoc Tukey’s test, a p < 0.01 vs the control cells transfected with negative control (NC) siRNA; b p < 0.01 vs the SFN-treated cells transfected with NC siRNA
Rights and permissions
About this article
Cite this article
Brasil, F.B., Gobbo, R.C.B., de Almeida, F.J.S. et al. The Isothiocyanate Sulforaphane Depends on the Nrf2/γ-GCL/GSH Axis to Prevent Mitochondrial Dysfunction in Cells Exposed to Methylglyoxal. Neurochem Res 46, 740–754 (2021). https://doi.org/10.1007/s11064-020-03204-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-020-03204-x