Abstract
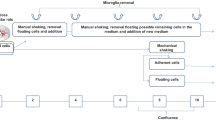
Astrocyte-rich primary cultures (APCs) are frequently used as a model system for the investigation of properties of brain astrocytes. However, as APCs contain a substantial number of microglial and oligodendroglial cells, biochemical parameters determined for such cultures may at least in part reflect also the presence of the contaminating cell types. To lower the potential contributions of microglial and oligodendroglial cells on properties of the astrocytes in APCs we prepared rat astrocyte-rich secondary cultures (ASCs) by subculturing of APCs and compared these ASCs with APCs regarding basal metabolic parameters, specific enzyme activities and the accumulation of iron oxide nanoparticles. Immunocytochemical characterization revealed that ASCs contained only minute amounts of microglial and oligodendroglial cells. ASCs and APCs did not significantly differ in their specific glucose consumption and lactate production rates, in their specific iron and glutathione contents, in their specific activities of various enzymes involved in glucose and glutathione metabolism nor in their accumulation of iron oxide nanoparticles. Thus, the absence or presence of some contaminating microglial and oligodendroglial cells appears not to substantially modulate the investigated metabolic parameters of astrocyte cultures.





Similar content being viewed by others
References
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119:7–35
Parpura V, Heneka MT, Montana V, Oliet SH, Schousboe A, Haydon PG, Stout RF Jr, Spray DC, Reichenbach A, Pannicke T, Pekny M, Pekna M, Zorec R, Verkhratsky A (2012) Glial cells in (patho)physiology. J Neurochem 121:4–27
Kettenmann H, Verkhratsky A (2011) Neuroglia—living nerve glue. Fortschr Neurol Psychiatr 79:588–597
Kimelberg HK (2010) Functions of mature mammalian astrocytes: a current view. Neuroscientist 16:79–106
Barros LF, Deitmer JW (2010) Glucose and lactate supply to the synapse. Brain Res Rev 63:149–159
Belanger M, Allaman I, Magistretti PJ (2011) Brain energy metabolism: focus on astrocyte-neuron metabolic cooperation. Cell Metab 14:724–738
Mangia S, Simpson IA, Vannucci SJ, Carruthers A (2009) The in vivo neuron-to-astrocyte lactate shuttle in human brain: evidence from modeling of measured lactate levels during visual stimulation. J Neurochem 109:55–62
Zielke HR, Zielke CL, Baab PJ, Tildon JT (2007) Effect of fluorocitrate on cerebral oxidation of lactate and glucose in freely moving rats. J Neurochem 101:9–16
Hirrlinger J, Dringen R (2010) The cytosolic redox state of astrocytes: maintenance, regulation and functional implications for metabolite trafficking. Brain Res Rev 63:177–188
Schmidt MM, Dringen R (2012) Glutathione (GSH) synthesis and metabolism. In: Choi IY, Gruetter R (ed) Neural metabolism in vivo, 4th volume, Springer, pp 1029–1050
Dringen R, Bishop GM, Koeppe M, Dang TN, Robinson SR (2007) The pivotal role of astrocytes in the metabolism of iron in the brain. Neurochem Res 32:1884–1890
Scheiber IF, Dringen R (2013) Astrocyte functions in the copper homeostasis of the brain. Neurochem Int 62:556–565
Hohnholt MC, Geppert M, Luther EM, Petters C, Bulcke F, Dringen R (2013) Handling of iron oxide and silver nanoparticles by astrocytes. Neurochem Res 38:227–239
Lange SC, Bak LK, Waagepetersen HS, Schousboe A, Norenberg MD (2012) Primary cultures of astrocytes: their value in understanding astrocytes in health and disease. Neurochem Res 37:2569–2588
de Vellis J, Cole R (2012) Preparation of mixed glial cultures from postnatal rat brain. Methods Mol Biol 814:49–59
Crocker SJ, Frausto RF, Whitton JL, Milner R (2008) A novel method to establish microglia-free astrocyte cultures: comparison of matrix metalloproteinase expression profiles in pure cultures of astrocytes and microglia. Glia 56:1187–1198
Losciuto S, Dorban G, Gabel S, Gustin A, Hoenen C, Grandbarbe L, Heuschling P, Heurtaux T (2012) An efficient method to limit microglia-dependent effects in astroglial cultures. J Neurosci Methods 207:59–71
Saura J (2007) Microglial cells in astroglial cultures: a cautionary note. J Neuroinflammation 4:26
Geppert M, Hohnholt MC, Thiel K, Nurnberger S, Grunwald I, Rezwan K, Dringen R (2011) Uptake of dimercaptosuccinate-coated magnetic iron oxide nanoparticles by cultured brain astrocytes. Nanotechnology 22:145101
Dauth S, Schmidt MM, Rehders M, Dietz F, Kelm S, Dringen R, Brix K (2012) Characterisation and metabolism of astroglia-rich primary cultures from cathepsin K-deficient mice. Biol Chem 393:959–970
Hamprecht B, Loffler F (1985) Primary glial cultures as a model for studying hormone action. Methods Enzymol 109:341–345
Tulpule K, Hohnholt MC, Hirrlinger J, Dringen R (2014) Primary cultures of astrocytes and neurons as model systems to study the metabolism and metabolite export from brain cells. In: Hirrlinger J, Waagepetersen H (ed) Neuromethods: Brain energy metabolism, Springer, Heidelberg (in press)
Kaltenbach JP, Kaltenbach MH, Lyons WB (1958) Nigrosin as a dye for differentiating live and dead ascites cells. Exp Cell Res 15:112–117
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Geppert M, Hohnholt M, Gaetjen L, Grunwald I, Baumer M, Dringen R (2009) Accumulation of iron oxide nanoparticles by cultured brain astrocytes. J Biomed Nanotechnol 5:285–293
Riemer J, Hoepken HH, Czerwinska H, Robinson SR, Dringen R (2004) Colorimetric ferrozine-based assay for the quantitation of iron in cultured cells. Anal Biochem 331:370–375
Hirrlinger J, Dringen R (2005) Multidrug resistance protein 1-mediated export of glutathione and glutathione disulfide from brain astrocytes. Methods Enzymol 400:395–409
Dringen R, Gebhardt R, Hamprecht B (1993) Glycogen in astrocytes: possible function as lactate supply for neighboring cells. Brain Res 623:208–214
Schmidt MM, Dringen R (2009) Differential effects of iodoacetamide and iodoacetate on glycolysis and glutathione metabolism of cultured astrocytes. Front Neuroenergetics 1:1–10
Minich T, Yokota S, Dringen R (2003) Cytosolic and mitochondrial isoforms of NADP+-dependent isocitrate dehydrogenases are expressed in cultured rat neurons, astrocytes, oligodendrocytes and microglial cells. J Neurochem 86:605–614
Gutterer JM, Dringen R, Hirrlinger J, Hamprecht B (1999) Purification of glutathione reductase from bovine brain, generation of an antiserum, and immunocytochemical localization of the enzyme in neural cells. J Neurochem 73:1422–1430
Sola C, Casal C, Tusell JM, Serratosa J (2002) Astrocytes enhance lipopolysaccharide-induced nitric oxide production by microglial cells. Eur J Neurosci 16:1275–1283
Walker AG, Chapman J, Bruce CB, Rumsby MG (1984) Immunocytochemical characterisation of cell cultures grown from dissociated 1-2-day post-natal rat cerebral tissue. A developmental study. J Neuroimmunol 7:1–20
McCarthy KD, de Vellis J (1980) Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J Cell Biol 85:890–902
Richardson A, Hao C, Fedoroff S (1993) Microglia progenitor cells: a subpopulation in cultures of mouse neopallial astroglia. Glia 7:25–33
Knorpp T, Robinson SR, Crack PJ, Dringen R (2006) Glutathione peroxidase-1 contributes to the protection of glutamine synthetase in astrocytes during oxidative stress. J Neural Transm 113:1145–1155
Skytt DM, Madsen KK, Pajecka K, Schousboe A, Waagepetersen HS (2010) Characterization of primary and secondary cultures of astrocytes prepared from mouse cerebral cortex. Neurochem Res 35:2043–2052
Oberheim NA, Goldmann SA, Nedergaard M (2012) Heterogeneity of astrocytic form and function. Methods Mol Biol 814:23–45
Zhang Y, Barres BA (2010) Astrocyte heterogeneity: an underappreciated topic in neurobiology. Curr Opin Neurobiol 20:588–594
Du F, Qian ZM, Zhu L, Wu XM, Qian C, Chan R, Ke Y (2010) Purity, cell viability, expression of GFAP and bystin in astrocytes cultured by different procedures. J Cell Biochem 109:30–37
Vanella A, Avola R, Condorelli DF, Campisi A, Costa A, Guiffrida Stella AM, Perez-Polo JR (1989) Antioxidant enzymatic activities and resistance to oxidative stress in primary and subcultured rat astroglial cells. Int J Dev Neurosci 7:233–241
Codeluppi S, Gregory EN, Kjell J, Wigerblad G, Olson L, Svensson CI (2011) Influence of rat substrain and growth conditions on the characteristics of primary cultures of adult rat spinal cord astrocytes. J Neurosci Methods 197:118–127
Goetschy JF, Ulrich G, Aunis D, Ciesielski-Treska J (1986) The organization and solubility properties of intermediate filaments and microtubules of cortical astrocytes in culture. J Neurocytol 15:375–387
Sen E, Basu A, Willing LB, Uliasz TF, Myrkalo JL, Vannucci SJ, Hewett SJ, Levison SW (2011) Pre-conditioning induces the precocious differentiation of neonatal astrocytes to enhance their neuroprotective properties. ASN Neuro 3:e00062
Nakanishi M, Niidome T, Matsuda S, Akaike A, Kihara T, Sugimoto H (2007) Microglia-derived interleukin-6 and leukaemia inhibitory factor promote astrocytic differentiation of neural stem/progenitor cells. Eur J Neurosci 25(3):649–658
Tilleux S, Berger J, Hermans E (2007) Induction of astrogliosis by activated microglia is associated with a down-regulation of metabotropic glutamate receptor 5. J Neuroimmunol 189:23–30
Passaquin AC, Schreier WA, de Vellis J (1994) Gene expression in astrocytes is affected by subculture. Int J Dev Neurosci 12:363–372
Bolanos JP, Almeida A, Moncada S (2010) Glycolysis: a bioenergetic or a survival pathway? Trends Biochem Sci 35:145–149
Figley CR, Stroman PW (2011) The role(s) of astrocytes and astrocyte activity in neurometabolism, neurovascular coupling, and the production of functional neuroimaging signals. Eur J Neurosci 33:577–588
Hertz L, Peng L, Dienel GA (2007) Energy metabolism in astrocytes: high rate of oxidative metabolism and spatiotemporal dependence on glycolysis/glycogenolysis. J Cereb Blood Flow Metab 27:219–249
Tulpule K, Dringen R (2012) Formate generated by cellular oxidation of formaldehyde accelerates the glycolytic flux in cultured astrocytes. Glia 60:582–593
Cruz F, Wolf A (2001) Effects of the novel cyclosporine derivative PSC833 on glucose metabolism in rat primary cultures of neuronal and glial cells. Biochem Pharmacol 62:129–139
Zwingmann C, Richter-Landsberg C, Brand A, Leibfritz D (2000) NMR spectroscopic study on the metabolic fate of [3-13C]alanine in astrocytes, neurons, and cocultures: implications for glia-neuron interactions in neurotransmitter metabolism. Glia 32:286–303
Schmidt MM, Dringen R (2010) Fumaric acid diesters deprive cultured primary astrocytes rapidly of glutathione. Neurochem Int 57:460–467
Hazell AS, Desjardins P, Butterworth RF (1999) Increased expression of glyceraldehyde-3-phosphate dehydrogenase in cultured astrocytes following exposure to manganese. Neurochem Int 35:11–17
O’Brien J, Kla KM, Hopkins IB, Malecki EA, McKenna MC (2007) Kinetic parameters and lactate dehydrogenase isozyme activities support possible lactate utilization by neurons. Neurochem Res 32:597–607
Vasquez OL, Almeida A, Bolanos JP (2001) Depletion of glutathione up-regulates mitochondrial complex I expression in glial cells. J Neurochem 76:1593–1596
Dringen R, Pfeiffer B, Hamprecht B (1999) Synthesis of the antioxidant glutathione in neurons: supply by astrocytes of CysGly as precursor for neuronal glutathione. J Neurosci 19:562–569
Arend C, Brandmann M, Dringen R (2013) The antiretrovial protease inhibitor ritonavir accelerates glutathione export from cultured primary astrocytes. Neurochem Res 38:732–741
Scheiber IF, Dringen R (2011) Copper-treatment increases the cellular GSH content and accelerates GSH export from cultured rat astrocytes. Neurosci Lett 498:42–46
Dringen R, Gutterer JM (2002) Glutathione reductase from bovine brain. Methods Enzymol 348:281–288
Dringen R, Hoepken HH, Minich T, Ruedig C (2007) Pentose phosphate pathway and NADPH metabolism. In: Dienel G, Gibson G (eds) Handbook of neurochemistry and molecular neurobiology, 5th volume: neural energy utilization. Springer, Heidelberg, pp 41–62
Minich T, Riemer J, Schulz JB, Wielinga P, Wijnholds J, Dringen R (2006) The multidrug resistance protein 1 (Mrp1), but not Mrp5, mediates export of glutathione and glutathione disulfide from brain astrocytes. J Neurochem 97:373–384
Hirrlinger J, Schulz JB, Dringen R (2002) Glutathione release from cultured brain cells: multidrug resistance protein 1 mediates the release of GSH from rat astroglial cells. J Neurosci Res 69:318–326
Geppert M, Hohnholt MC, Nurnberger S, Dringen R (2012) Ferritin up-regulation and transient ROS production in cultured brain astrocytes after loading with iron oxide nanoparticles. Acta Biomater 8:3832–3839
Bishop GM, Dang TN, Dringen R, Robinson SR (2011) Accumulation of non-transferrin-bound iron by neurons, astrocytes, and microglia. Neurotox Res 19:443–451
Hoepken HH (2005) Untersuchungen zum Eisenstoffwechsel neuraler Zellen. Dissertation, University of Tuebingen, Germany
Thorburne SK, Juurlink BH (1996) Low glutathione and high iron govern the susceptibility of oligodendroglial precursors to oxidative stress. J Neurochem 67:1014–1022
Todorich B, Pasquini JM, Garcia CI, Paez PM, Connor JR (2009) Oligodendrocytes and myelination: the role of iron. Glia 57:467–478
Urrutia P, Aguirre P, Esparza A, Tapia V, Mena NP, Arredondo M, Gonzalez-Billault C, Nunez MT (2013) Inflammation alters the expression of DMT1, FPN1 and hepcidin, and it causes iron accumulation in central nervous system cells. J Neurochem 126:541–549
Lamkowsky MC, Geppert M, Schmidt MM, Dringen R (2012) Magnetic field-induced acceleration of the accumulation of magnetic iron oxide nanoparticles by cultured brain astrocytes. J Biomed Mater Res A 100A:323–334
Jenkins SI, Pickard MR, Furness DN, Yiu HH, Chari DM (2012) Differences in magnetic particle uptake by CNS neuroglial subclasses: implications for neural tissue engineering. Nanomedicine (Lond) 8:951–968
Pinkernelle J, Calatayud P, Goya GF, Fansa H, Keilhoff G (2012) Magnetic nanoparticles in primary neural cell cultures are mainly taken up by microglia. BMC Neurosci 13:32
Luther EM, Petters C, Bulcke F, Kaltz A, Thiel K, Bickmeyer U, Dringen R (2013) Endocytotic uptake of iron oxide nanoparticles by cultured brain microglial cells. Acta Biomater 9:8454–8465
Acknowledgments
The authors would like to thank Dr. Frank Dietz (University of Bremen) for providing the MAG- and anti-sheep-antibodies and Dr. Eva M. Luther for her help with the analysis of immunocytochemical stainings.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petters, C., Dringen, R. Comparison of Primary and Secondary Rat Astrocyte Cultures Regarding Glucose and Glutathione Metabolism and the Accumulation of Iron Oxide Nanoparticles. Neurochem Res 39, 46–58 (2014). https://doi.org/10.1007/s11064-013-1189-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-013-1189-7